Electrocatalytic Synthesis of 3-Hydroxycyclohexanone: A Scalable Green Route for Pharmaceutical Intermediates
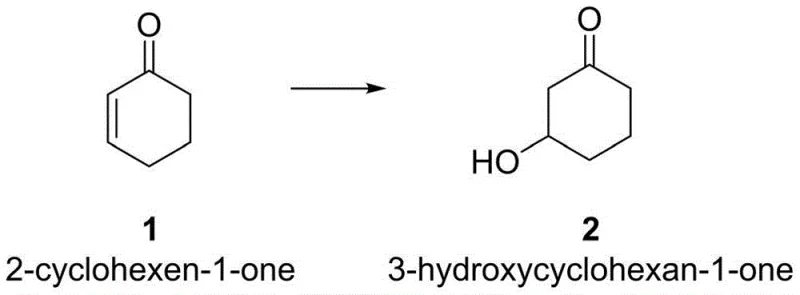
The landscape of fine chemical manufacturing is undergoing a paradigm shift towards sustainable electro-organic synthesis, a transition vividly exemplified by the technological breakthroughs detailed in patent CN111945180A. This intellectual property introduces a novel, environmentally benign methodology for the direct conversion of 2-cyclohexen-1-one into 3-hydroxycyclohexanone, a critical building block in the synthesis of various bioactive molecules and pharmaceutical intermediates. By leveraging the power of electrocatalysis, this process circumvents the need for stoichiometric oxidants and toxic heavy metal catalysts that have historically plagued the production of cyclic ketone derivatives. For R&D directors and procurement strategists alike, this represents a significant opportunity to streamline supply chains while adhering to increasingly stringent global environmental regulations. The core innovation lies in the utilization of water electrolysis to generate reactive radical species in situ, facilitating a clean addition reaction that proceeds under remarkably mild conditions. This report analyzes the technical merits and commercial viability of this route, positioning it as a superior alternative for the reliable supply of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of hydroxycyclohexanone derivatives has relied on chemically intensive pathways that pose substantial operational and environmental challenges for large-scale manufacturers. Traditional protocols often involve the selective oxidation of diols using reagents like sodium bromate and sodium bisulfite, or the use of cobalt acetate and N-hydroxyphthalimide (NHPI) systems which introduce heavy metal contamination risks into the final product stream. Furthermore, alternative routes utilizing 1,4-cyclohexanedione ethylene glycol monoketal require harsh hydrolysis conditions involving strong acids or bases at elevated temperatures to remove protecting groups. These legacy methods not only generate significant volumes of hazardous waste requiring costly treatment but also suffer from issues related to the instability of halogenated intermediates, leading to inconsistent batch quality and reduced overall process efficiency. The reliance on such aggressive reagents inevitably drives up the cost of goods sold (COGS) due to the expenses associated with waste disposal, specialized corrosion-resistant equipment, and extensive purification steps required to meet pharmacopeial standards.
The Novel Approach
In stark contrast, the electrocatalytic method disclosed in CN111945180A offers a streamlined, single-step transformation that directly functionalizes the carbon-carbon double bond of 2-cyclohexen-1-one. By employing simple carbonate salts such as potassium carbonate (K2CO3) as electrolytes in an aqueous or semi-aqueous medium, the process eliminates the need for external chemical oxidants entirely. The reaction is driven by electricity, utilizing a platinum cathode and a carbon anode to facilitate the generation of hydroxyl radicals from water, which then add across the alkene moiety with high regioselectivity. This approach operates at ambient temperature and pressure under inert gas protection, drastically reducing energy consumption compared to thermal reflux methods. The simplicity of the workup procedure, which typically involves standard extraction and column chromatography, further enhances the practicality of this method for industrial adoption. Consequently, this novel route addresses the critical pain points of traditional synthesis by offering a greener, safer, and potentially more cost-effective pathway for cost reduction in fine chemical manufacturing.
Mechanistic Insights into Electrocatalytic Radical Addition
The fundamental mechanism driving this transformation is rooted in the electrochemical oxidation of water at the anode surface, which generates highly reactive hydroxyl radicals (•OH) in the immediate vicinity of the electrode. In the presence of the carbonate electrolyte, the solution conductivity is maintained, allowing for a steady flow of current that sustains the radical flux without the accumulation of deleterious side products. These electro-generated radicals undergo a conjugate addition to the electron-deficient double bond of the 2-cyclohexen-1-one substrate, forming a transient carbon-centered radical intermediate. This intermediate subsequently abstracts a hydrogen atom or undergoes further electrochemical reduction to yield the saturated 3-hydroxycyclohexanone product. The use of mild carbonate bases helps to buffer the local pH near the electrode, preventing the degradation of the sensitive ketone functionality which might occur under strongly acidic or alkaline conditions typical of other oxidation methods. This precise control over the reaction environment ensures that the delicate balance between reactivity and selectivity is maintained, resulting in a cleaner impurity profile that simplifies downstream processing.
From an impurity control perspective, the absence of halogenating agents means that there is no risk of forming difficult-to-remove organohalide byproducts, which are often genotoxic impurities of concern in pharmaceutical synthesis. Additionally, the avoidance of transition metal catalysts like cobalt or copper eliminates the need for rigorous metal scavenging steps, which can otherwise lead to product loss and increased cycle times. The radical nature of the reaction is inherently self-limiting in this specific electrochemical setup, as the concentration of radicals is governed by the applied current density rather than the concentration of a chemical initiator. This allows for fine-tuning of the reaction rate simply by adjusting the voltage and current parameters, providing process engineers with a robust handle on reaction kinetics. Such mechanistic clarity is essential for scaling up the process from laboratory benchtop to pilot plant, ensuring that the high purity specifications required for API intermediates can be consistently met.
How to Synthesize 3-Hydroxycyclohexanone Efficiently
To implement this electrocatalytic protocol effectively, operators must adhere to specific parameters regarding electrolyte concentration and electrical settings to maximize yield and minimize side reactions. The process begins by dissolving the substrate 2-cyclohexen-1-one in a solvent system comprising water or a mixture of water with organic co-solvents like tetrahydrofuran (THF) or acetonitrile. An appropriate amount of electrolyte, such as K2CO3, is added to ensure sufficient ionic strength for efficient current passage. The detailed standardized synthesis steps, including specific molar ratios and electrical configurations validated by the patent data, are outlined below to guide technical teams in replicating this high-efficiency route.
- Prepare the reaction vessel with 2-cyclohexen-1-one as the substrate and add a carbonate electrolyte such as K2CO3, Na2CO3, or Cs2CO3 to facilitate conductivity and catalysis.
- Introduce the solvent system, preferably water or a mixture of water with THF or acetonitrile, ensuring the molar ratio of substrate to electrolyte is optimized between 1: 0.5 and 1:6.
- Apply a constant current and voltage (e.g., 7V, 10mA) using a Platinum cathode and Carbon anode under inert gas protection at room temperature for 6 to 8 hours to achieve radical addition.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this electrocatalytic technology translates into tangible strategic benefits that extend beyond mere technical novelty. The primary advantage lies in the drastic simplification of the raw material basket; by replacing expensive and hazardous oxidants with electricity and water, the variable costs associated with reagent procurement are significantly lowered. Furthermore, the elimination of heavy metals and halogenated solvents reduces the regulatory burden and the costs associated with environmental compliance and waste disposal, contributing to substantial cost savings over the lifecycle of the product. The mild reaction conditions also imply that standard stainless steel or glass-lined reactors can be utilized without the need for exotic alloys resistant to strong acids, thereby lowering capital expenditure (CAPEX) for new production lines. This operational simplicity enhances supply chain reliability by reducing the dependency on complex global supply chains for specialty reagents that are often subject to volatility and shortage.
- Cost Reduction in Manufacturing: The economic model of this process is fundamentally superior because it substitutes costly chemical oxidants with electrical energy, which is generally cheaper and more stable in price. By removing the need for stoichiometric amounts of oxidizing agents and the subsequent quenching reagents, the mass intensity of the process is improved, leading to less waste generation per kilogram of product. The simplified workup procedure, which avoids complex extraction sequences required to remove metal catalysts, reduces labor hours and solvent consumption during the isolation phase. These factors collectively drive down the manufacturing cost, allowing for more competitive pricing in the market for high-purity pharmaceutical intermediates while maintaining healthy profit margins.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as potassium carbonate and water, combined with the use of durable electrode materials like carbon and platinum, ensures that the supply chain is resilient against disruptions. Unlike processes dependent on bespoke catalysts or unstable intermediates that require cold chain logistics, this method utilizes stable, shelf-stable reagents that are readily available from multiple global suppliers. The robustness of the electrochemical setup means that production can be sustained with minimal downtime for maintenance or reagent replenishment. This stability is crucial for meeting the just-in-time delivery requirements of downstream pharmaceutical clients, effectively reducing lead time for high-purity intermediates and strengthening the overall security of supply.
- Scalability and Environmental Compliance: Electrochemical processes are inherently scalable, particularly when adapted to continuous flow reactors, which offer superior heat and mass transfer characteristics compared to batch processing. This scalability facilitates the commercial scale-up of complex cyclic ketones from kilogram to multi-ton quantities without the exponential increase in safety risks associated with thermal runaways in traditional oxidation reactions. Moreover, the green nature of the process, characterized by the use of water as a reactant and the absence of toxic heavy metal effluents, aligns perfectly with modern sustainability goals and regulatory frameworks. This environmental compatibility minimizes the risk of regulatory shutdowns and enhances the corporate social responsibility profile of the manufacturing entity, making it a preferred partner for eco-conscious multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrocatalytic synthesis route. These answers are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances is critical for technical teams evaluating the feasibility of integrating this technology into existing production workflows.
Q: What are the primary advantages of this electrocatalytic method over traditional halogenation routes?
A: Unlike traditional methods that require harsh halogenation followed by hydrolysis involving unstable intermediates and heavy metals, this electrocatalytic approach utilizes water electrolysis to generate radicals under mild, room-temperature conditions, significantly reducing environmental impact and safety hazards.
Q: Can this process be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the process uses simple electrode materials (Pt/C) and common electrolytes (carbonates) in aqueous or semi-aqueous solvents, making it highly amenable to scale-up in flow electrochemical reactors for commercial manufacturing without the need for complex anhydrous conditions.
Q: How does the choice of electrolyte affect the reaction efficiency?
A: The patent specifies that strong base-weak acid salts like K2CO3, Na2CO3, or Cs2CO3 act as effective electrolytes to improve reaction rates and catalytic efficiency, with K2CO3 in water being a preferred embodiment for achieving high yields.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Hydroxycyclohexanone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrocatalytic synthesis in modernizing the production of key pharmaceutical intermediates like 3-hydroxycyclohexanone. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of materials. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch meets the exacting standards required for drug substance manufacturing. We are committed to leveraging advanced technologies, such as the green electrochemical routes described herein, to deliver superior value to our global partners.
We invite you to collaborate with us to explore how this innovative synthesis method can optimize your specific supply chain requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating exactly how switching to this electrocatalytic route can improve your bottom line. Please contact us today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and cost-effective source for your critical intermediates.
