Advanced Green Synthesis of 3-Aryl-2H-benzo[b][1,4]oxazin-2-one Derivatives for Pharmaceutical Applications
Advanced Green Synthesis of 3-Aryl-2H-benzo[b][1,4]oxazin-2-one Derivatives for Pharmaceutical Applications
In the rapidly evolving landscape of pharmaceutical intermediate manufacturing, the demand for sustainable, high-efficiency synthetic routes has never been more critical. Patent CN111100085A introduces a groundbreaking preparation method for 3-aryl-2H-benzo[β][1,4]benzoxazine-2-one compounds, a class of heterocyclic structures renowned for their significant photophysical properties and potential antitumor activities. This technology represents a paradigm shift from traditional catalytic systems to a novel deep eutectic solvent (DES) approach, addressing long-standing challenges regarding environmental toxicity and operational complexity. By leveraging a eutectic mixture of choline chloride and urea, this method achieves exceptional reaction efficiency under mild thermal conditions, offering a compelling value proposition for R&D directors seeking robust pathways for complex heterocycle synthesis. The elimination of heavy metal catalysts not only streamlines the purification process but also aligns perfectly with the stringent regulatory requirements facing modern reliable pharmaceutical intermediate suppliers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzoxazine derivatives has relied heavily on conventional organic solvents and transition metal catalysts, which present substantial drawbacks in both economic and environmental contexts. Traditional protocols often necessitate the use of hazardous reagents that require rigorous safety handling and generate significant volumes of toxic waste, complicating the disposal process and inflating operational costs. Furthermore, many existing catalytic systems suffer from limited substrate tolerance, failing to accommodate diverse electronic environments on the aromatic rings without significant loss in yield or selectivity. The reliance on expensive noble metals or corrosive acids also introduces the risk of metal contamination in the final product, a critical failure point for high-purity pharmaceutical intermediates destined for clinical applications. These legacy methods frequently demand harsh reaction conditions, such as elevated temperatures or prolonged reaction times, which can lead to the degradation of sensitive functional groups and the formation of difficult-to-remove impurities.
The Novel Approach
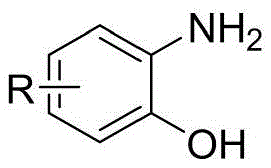
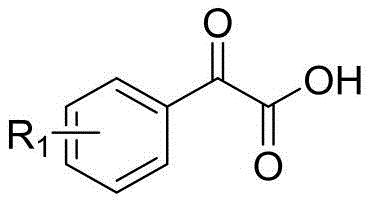
The methodology disclosed in CN111100085A fundamentally reimagines the reaction environment by replacing volatile organic solvents with a biodegradable deep eutectic solvent composed of choline chloride and urea. This innovative solvent system acts not merely as a medium but as a promoter of the reaction, facilitating the condensation between 2-aminophenol compounds and keto acids through a unique hydrogen-bonding network. The process operates under remarkably mild conditions, typically requiring magnetic stirring in an oil bath at just 80°C, which significantly reduces energy consumption compared to high-temperature reflux methods. This approach drastically simplifies the post-reaction workup, as the target 3-aryl-2H-benzo[β][1,4]benzoxazine-2-one compounds can be isolated with high purity through standard column chromatography and low-temperature vacuum drying. By removing the dependency on toxic catalysts, this novel route ensures a cleaner impurity profile and enhances the overall sustainability of the manufacturing process, making it an ideal candidate for cost reduction in API manufacturing.
Mechanistic Insights into Deep Eutectic Solvent-Promoted Cyclization
The efficacy of this synthesis lies in the physicochemical properties of the deep eutectic solvent, which creates a highly organized supramolecular structure capable of stabilizing transition states. The choline chloride and urea mixture, formed in a 1:2 molar ratio, generates a liquid phase with a melting point significantly lower than either individual component, providing a fluid medium that enhances mass transfer between the solid reactants. Mechanistically, the hydroxyl and amine groups of the 2-aminophenol engage in hydrogen bonding with the urea molecules, activating the nucleophilic sites for attack on the carbonyl group of the keto acid. This activation lowers the energy barrier for the initial condensation step, allowing the reaction to proceed rapidly at 80°C without the need for external acidic or basic catalysts. The subsequent intramolecular cyclization is facilitated by the solvent's ability to stabilize the developing charge separation in the transition state, leading to the formation of the stable benzoxazine ring system with high regioselectivity.
From an impurity control perspective, the mild thermal profile of this reaction is instrumental in minimizing side reactions such as polymerization or oxidative degradation, which are common pitfalls in high-temperature syntheses. The absence of transition metals eliminates the risk of metal-catalyzed side reactions that often generate complex byproduct mixtures difficult to separate. Furthermore, the polar nature of the deep eutectic solvent helps to solubilize polar intermediates while allowing the less polar final product to be easily extracted or precipitated during workup. This inherent selectivity ensures that the crude product obtained after the reaction contains a high percentage of the desired 3-aryl-2H-benzo[β][1,4]benzoxazine-2-one, reducing the burden on downstream purification units. For process chemists, this translates to a more predictable and controllable reaction profile, essential for maintaining consistent quality in commercial scale-up of complex pharmaceutical intermediates.
![Reaction scheme for the synthesis of 3-phenyl-2H-benzo[b][1,4]oxazin-2-one using deep eutectic solvent](/insights/img/3-aryl-benzoxazinone-synthesis-pharma-supplier-20260302230639-03.webp)
How to Synthesize 3-Aryl-2H-benzo[b][1,4]oxazin-2-one Efficiently
The practical implementation of this green chemistry protocol involves a straightforward sequence of mixing, heating, and purification steps that can be easily adapted for both laboratory and pilot-scale operations. The process begins with the in situ preparation of the deep eutectic solvent, followed by the addition of stoichiometric amounts of the 2-aminophenol and keto acid substrates. The reaction mixture is then subjected to controlled heating and agitation to ensure homogeneity and optimal reaction kinetics. Detailed standardized synthetic procedures, including specific molar ratios, temperature ramping rates, and purification parameters, are outlined in the technical guide below to ensure reproducibility and safety.
- Prepare the deep eutectic solvent by mixing choline chloride and urea in a 1: 2 molar ratio and heating to 80°C until a uniform transparent liquid forms.
- Add the 2-aminophenol compound and the keto acid compound to the solvent in a 1: 2 molar ratio and stir magnetically under an oil bath at 80°C.
- After the reaction completes (typically 2 hours), perform post-treatment including column chromatography purification and vacuum drying at 30°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this deep eutectic solvent technology offers transformative benefits that extend far beyond simple yield improvements. The primary economic driver is the drastic reduction in raw material costs associated with the elimination of expensive transition metal catalysts and specialized ligands, which are often subject to volatile market pricing and supply constraints. By utilizing commodity chemicals like choline chloride and urea, manufacturers can secure a stable and low-cost supply of reaction media, insulating production budgets from the fluctuations typical of the fine chemical market. Additionally, the simplified workup procedure reduces the consumption of organic solvents during extraction and washing phases, leading to significant savings in solvent procurement and waste disposal fees. This holistic reduction in material intensity directly contributes to a leaner cost structure, enabling more competitive pricing strategies for downstream clients.
- Cost Reduction in Manufacturing: The removal of precious metal catalysts eliminates the need for costly scavenging resins or complex filtration steps required to meet strict residual metal specifications. This streamlining of the purification train reduces both the time and capital expenditure associated with downstream processing. Furthermore, the recyclability potential of deep eutectic solvents offers an additional avenue for cost optimization, as the solvent matrix can potentially be recovered and reused in subsequent batches, further driving down the variable cost per kilogram of the final product.
- Enhanced Supply Chain Reliability: The reliance on widely available, bulk-grade starting materials ensures a robust supply chain that is less susceptible to disruptions caused by the scarcity of specialized reagents. Choline chloride and urea are produced on a massive industrial scale for other sectors, guaranteeing consistent availability and short lead times for procurement teams. This reliability is crucial for maintaining continuous production schedules and meeting the just-in-time delivery expectations of global pharmaceutical partners, thereby reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The benign nature of the reagents and solvents simplifies the regulatory approval process for new manufacturing sites, as the environmental, health, and safety (EHS) profile is significantly improved compared to traditional methods. The process generates minimal hazardous waste, reducing the liability and cost associated with waste treatment and disposal. This alignment with green chemistry principles facilitates easier permitting and expansion of production capacity, supporting the commercial scale-up required to meet growing market demand without compromising on sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this deep eutectic solvent technology. These insights are derived directly from the experimental data and beneficial effects described in the patent literature, providing a clear understanding of the method's capabilities and limitations for potential adopters.
Q: What are the advantages of using deep eutectic solvents over traditional catalysts for benzoxazine synthesis?
A: Deep eutectic solvents (DES) eliminate the need for toxic transition metal catalysts and volatile organic solvents. They provide a greener reaction medium that simplifies downstream processing, reduces environmental impact, and often results in higher yields due to the unique hydrogen-bonding network that stabilizes intermediates.
Q: What is the substrate scope for this synthesis method?
A: The method demonstrates excellent tolerance for various substituents. It works effectively with 2-aminophenols containing electron-withdrawing groups (like halogens) and electron-donating groups (like methyl). Similarly, keto acids with fluoro, chloro, bromo, methyl, and methoxy substituents are compatible, providing a versatile route for diverse derivatives.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is highly scalable. The use of inexpensive, non-toxic components (choline chloride and urea) and mild reaction conditions (80°C) makes it economically viable for industrial scale-up. The simple workup procedure involving column chromatography and vacuum drying further supports commercial feasibility.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Aryl-2H-benzo[b][1,4]oxazin-2-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of adopting next-generation synthetic methodologies to maintain a competitive edge in the global pharmaceutical market. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify the absence of residual solvents and metal impurities. Our infrastructure is designed to support the rapid deployment of green chemistry solutions like the deep eutectic solvent method, allowing us to offer our partners a sustainable and cost-effective supply of critical heterocyclic intermediates.
We invite you to collaborate with us to leverage this innovative technology for your specific project needs. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this greener route can optimize your bill of materials. Please contact our technical procurement team today to request specific COA data for our benzoxazine derivatives and to discuss route feasibility assessments for your custom synthesis projects. Together, we can drive efficiency and sustainability in the production of high-value pharmaceutical ingredients.
