Advanced Metal-Free Synthesis of Spiro-gamma-lactam Compounds for Pharmaceutical Applications
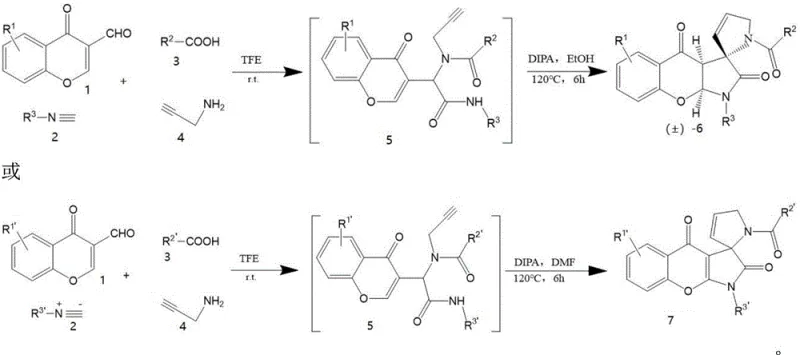
The pharmaceutical industry continuously seeks robust synthetic routes for complex heterocyclic scaffolds that serve as critical building blocks for next-generation therapeutics. Patent CN113024572B introduces a groundbreaking methodology for the construction of spiro-γ-lactam compounds, a structural motif renowned for its presence in bioactive alkaloids and potential anticancer agents. Unlike traditional approaches that heavily rely on transition metal catalysis, this invention leverages a catalyst-free Ugi multicomponent reaction strategy. By utilizing chromone-3-carbaldehyde derivatives, carboxylic acids, isocyanides, and propargylamine as starting materials, the process achieves high molecular complexity in a single operational sequence. This technological leap not only streamlines the synthetic pathway but also addresses critical purity concerns associated with metal residues in active pharmaceutical ingredients (APIs), positioning it as a highly attractive route for reliable pharmaceutical intermediate supplier partnerships focused on quality and compliance.
The development of efficient synthetic methodologies for spirocyclic structures has long been a challenging frontier in organic chemistry due to the steric hindrance associated with forming quaternary carbon centers. Historically, chemists have depended on transition metal catalysts to overcome these energy barriers, often resulting in complex reaction mixtures and stringent purification requirements. The limitations of these conventional methods are particularly pronounced when scaling up for commercial manufacturing, where catalyst cost and removal become significant bottlenecks. The novel approach detailed in this patent circumvents these issues by employing a tandem Ugi reaction followed by thermal cyclization. This metal-free protocol operates under mild conditions, significantly reducing the environmental footprint and operational complexity. By eliminating the need for exotic ligands or sensitive metal complexes, the process enhances the overall robustness of the supply chain for high-purity pharmaceutical intermediates, ensuring consistent quality without the variability introduced by catalyst degradation or batch-to-batch metal loading differences.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for constructing spiro-γ-lactam frameworks frequently involve transition metal-catalyzed reactions, such as copper-catalyzed trifluoromethylation or palladium-mediated coupling processes. These methods, while effective in academic settings, present substantial drawbacks for industrial application, primarily concerning cost and safety. The reliance on precious metals like palladium or copper necessitates the inclusion of additional downstream processing steps to ensure residual metal levels meet strict regulatory limits for pharmaceutical products. Furthermore, these catalysts are often sensitive to air and moisture, requiring inert atmosphere conditions that increase capital expenditure and operational costs. The use of stoichiometric oxidants or specialized reagents like Togni's reagent further complicates the waste management profile, generating hazardous byproducts that require careful disposal. Consequently, the overall process mass intensity (PMI) of these conventional routes is often suboptimal, leading to higher production costs and reduced sustainability metrics compared to newer, more direct methodologies.
The Novel Approach
In stark contrast, the patented method utilizes a four-component Ugi reaction that proceeds efficiently without any external catalyst, marking a significant paradigm shift in spirocycle synthesis. The reaction initiates at room temperature in tetrafluoroethylene, combining chromone-3-carbaldehyde, isocyanide, carboxylic acid, and propargylamine to form a key intermediate. This intermediate subsequently undergoes an intramolecular cyclization upon heating in a mixture of diisopropanolamine and ethanol or DMF. The absence of metal catalysts not only simplifies the reaction setup but also drastically reduces the impurity profile, as there are no metal-ligand complexes to separate. The mild reaction conditions allow for a broader substrate scope, tolerating various functional groups on the aromatic rings without the need for protective group strategies. This streamlined approach translates directly into cost reduction in pharmaceutical intermediates manufacturing by minimizing raw material costs, reducing solvent consumption, and shortening the overall production cycle time through fewer unit operations.
Mechanistic Insights into Catalyst-Free Ugi Cyclization
The mechanistic pathway of this transformation is a testament to the power of multicomponent reactions in building molecular complexity with atom economy. The process begins with the formation of an imine from the condensation of the aldehyde and amine components, followed by the nucleophilic attack of the isocyanide to generate a nitrilium ion intermediate. The carboxylate anion then traps this electrophilic species, leading to the formation of the Ugi adduct, which contains the necessary functionality for the subsequent cyclization. Upon heating, the propargyl amide moiety undergoes an intramolecular nucleophilic attack, likely facilitated by the basic environment provided by diisopropanolamine, to close the lactam ring and form the spiro center. This cascade sequence effectively constructs two new bonds and a quaternary stereocenter in a single pot. The elegance of this mechanism lies in its self-assembly nature, where the thermodynamic drive towards the stable spiro-γ-lactam structure dictates the reaction outcome, minimizing side reactions and maximizing yield without the need for kinetic control via metal catalysis.
Impurity control is inherently superior in this metal-free system due to the absence of transition metal-induced side reactions such as homocoupling or over-oxidation. In traditional metal-catalyzed processes, trace metals can catalyze decomposition pathways or promote the formation of regioisomers that are difficult to separate. Here, the primary impurities arise from unreacted starting materials or simple hydrolysis products, which are generally more polar and easier to remove via standard aqueous workups or crystallization. The use of diisopropanolamine serves a dual purpose as both a solvent component and a base, buffering the reaction medium to prevent acid-catalyzed degradation of the sensitive lactam ring. This controlled environment ensures that the final product maintains high chemical integrity, which is crucial for downstream biological testing and clinical applications. The ability to produce high-purity spiro-γ-lactam compounds with minimal chromatographic purification steps significantly enhances the commercial viability of this route for large-scale API intermediate production.
How to Synthesize Spiro-gamma-lactam Efficiently
The practical execution of this synthesis involves a straightforward two-stage protocol that is amenable to standard laboratory and pilot plant equipment. Initially, equimolar amounts of the four components are stirred at ambient temperature to ensure complete conversion to the Ugi adduct, monitored conveniently by thin-layer chromatography. Following the removal of the initial solvent, the crude residue is subjected to thermal cyclization in a sealed vessel, ensuring safety while maintaining the required temperature of 120°C. The detailed standardized synthesis steps see the guide below, which outlines the specific workup procedures involving ethyl acetate extraction and silica gel chromatography to isolate the final pure compounds. This operational simplicity reduces the training burden on technical staff and minimizes the risk of operator error, making it an ideal candidate for technology transfer between R&D and manufacturing sites.
- Mix chromone-3-carbaldehyde, isocyanide, carboxylic acid, and propargylamine in tetrafluoroethylene at room temperature for 5 hours to form the Ugi adduct intermediate.
- Seal the crude intermediate in a mixed solution of Diisopropanolamine (DIPA) and ethanol or DMF.
- Heat the mixture at 120°C for 6 hours to induce cyclization, then purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the elimination of transition metal catalysts represents a substantial opportunity for cost optimization and supply chain resilience. Precious metal prices are subject to significant market volatility, and securing a consistent supply of high-purity catalysts can be a logistical challenge. By adopting this metal-free methodology, manufacturers can decouple their production costs from the fluctuating commodities market, leading to more predictable budgeting and pricing stability for clients. Additionally, the simplified purification process reduces the consumption of specialized scavenger resins and silica gel, further lowering the bill of materials. The reduced waste generation also aligns with increasingly stringent environmental regulations, potentially lowering disposal fees and enhancing the company's sustainability profile, which is a key factor for modern pharmaceutical supply chains aiming for green chemistry certification.
- Cost Reduction in Manufacturing: The removal of expensive metal catalysts and the associated ligands directly lowers the raw material costs per kilogram of product. Furthermore, the simplified workup procedure eliminates the need for costly metal scavenging steps and reduces solvent usage during purification. This lean manufacturing approach results in significant cost savings that can be passed down to the customer or reinvested into process optimization. The high atom economy of the multicomponent reaction ensures that a larger proportion of the starting materials end up in the final product, minimizing waste disposal costs and maximizing resource efficiency throughout the production lifecycle.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals like carboxylic acids and amines rather than specialized catalysts mitigates the risk of supply disruptions. These starting materials are widely available from multiple global suppliers, ensuring continuity of supply even in volatile market conditions. The robustness of the reaction conditions means that production is less susceptible to delays caused by equipment failures related to inert gas systems or sensitive catalyst handling. This reliability is critical for meeting tight delivery schedules and maintaining the trust of downstream pharmaceutical partners who depend on just-in-time delivery models for their clinical trial materials and commercial launches.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by the successful synthesis of a wide range of derivatives with consistent yields. The use of common solvents like ethanol and DMF facilitates easy scale-up without requiring specialized high-pressure reactors or cryogenic cooling systems. From an environmental standpoint, the absence of heavy metals simplifies wastewater treatment and reduces the ecological footprint of the manufacturing process. This compliance with green chemistry principles not only satisfies regulatory requirements but also appeals to environmentally conscious stakeholders, positioning the manufacturer as a leader in sustainable chemical production practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthetic route. Understanding these details is essential for R&D teams evaluating the feasibility of integrating this chemistry into their existing pipelines. The answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, ensuring accuracy and relevance for decision-makers. These insights cover aspects ranging from reaction scalability to impurity profiles, providing a comprehensive overview of the technology's capabilities and limitations in a real-world manufacturing context.
Q: Does this synthesis method require transition metal catalysts?
A: No, the patented method (CN113024572B) utilizes a metal-free Ugi multicomponent reaction, eliminating the need for expensive copper or palladium catalysts and subsequent heavy metal removal steps.
Q: What are the biological activities of the synthesized compounds?
A: The synthesized spiro-gamma-lactam compounds exhibit significant antitumor activity, specifically showing inhibition effects on PANC pancreatic cancer cells and U87 glioma cells comparable to Paclitaxel in experimental data.
Q: Is the process suitable for large-scale manufacturing?
A: Yes, the reaction conditions are mild (room temperature for the first step, 120°C for cyclization) and use common solvents like ethanol and DMF, making the process highly scalable for commercial production without complex pressure requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Spiro-gamma-lactam Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this catalyst-free synthesis technology for the development of novel anticancer therapeutics. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of spiro-γ-lactam intermediate meets the highest international standards. We understand that consistency and quality are paramount in the pharmaceutical industry, and our dedicated technical team is committed to delivering products that support your regulatory filings and clinical success.
We invite you to collaborate with us to leverage this advanced chemistry for your drug development programs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can accelerate your timeline and optimize your budget. Let us be your trusted partner in bringing these promising spiro-γ-lactam compounds from the laboratory to the patients who need them most.
