Advanced Synthesis of Biimidazole Phosphine Ligands for Industrial Catalysis
The chemical landscape of organophosphorus ligands has been significantly advanced by the innovations detailed in patent CN101676293B, which introduces a robust methodology for synthesizing biimidazole organic phosphine compounds. This patent addresses a critical gap in the availability of functionalized ligands that combine the stability of the biimidazole scaffold with the versatile coordination chemistry of phosphorus. For research and development teams focused on catalyst design, this technology offers a pathway to access novel metal-organic complexes that were previously difficult to procure. The core innovation lies in a multi-step indirect synthetic strategy that bypasses the thermodynamic pitfalls of direct alkylation, ensuring high structural fidelity and purity. By leveraging this proprietary knowledge, industrial partners can secure a reliable biimidazole phosphine supplier capable of delivering complex intermediates essential for next-generation catalytic systems and separation technologies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-substituted biimidazole derivatives relied heavily on direct alkylation strategies using dihaloalkanes, a method documented in earlier literature such as J. Org. Chem. (1989). However, this conventional approach suffers from severe intrinsic limitations when applied to the formation of long-chain phosphine precursors. The primary issue arises from the high nucleophilicity of the imidazole nitrogen atoms, which tend to undergo intramolecular nucleophilic substitution reactions when exposed to dihaloalkanes. Instead of forming the desired linear bis-alkylated product, the reaction frequently results in cyclization or polymerization, yielding imidazolium salts or other unwanted byproducts. This side reaction drastically reduces the yield of the target intermediate and complicates the purification process, making it economically unviable for large-scale production. Furthermore, the harsh conditions often required for direct alkylation can degrade sensitive functional groups, limiting the scope of substituents that can be introduced onto the biimidazole core.
The Novel Approach
In stark contrast to the failed direct methods, the technology disclosed in CN101676293B employs a sophisticated indirect synthetic route that effectively circumvents these cyclization issues. The process begins with the preparation of 1,1'-bis(alkyl propionate)-2,2'-biimidazole, a stable ester precursor that serves as the foundation for the carbon chain extension. By utilizing a stepwise transformation involving reduction, halogenation, and finally nucleophilic substitution, the synthesis maintains control over the reaction trajectory at every stage. This method allows for the precise installation of the propyl linker arms without triggering the premature intramolecular attacks that plague direct alkylation. The result is a high-yielding, scalable process capable of producing 1,1'-bis(3-chloropropyl)-2,2'-biimidazole and its subsequent phosphine derivatives with exceptional purity. This strategic shift from direct to indirect synthesis represents a paradigm change in how complex biimidazole ligands are manufactured, offering a viable solution for cost reduction in catalyst intermediate manufacturing.
Mechanistic Insights into Stepwise Functionalization and Phosphination
The mechanistic elegance of this synthesis lies in its sequential manipulation of oxidation states and functional groups to build molecular complexity. The process initiates with the reduction of the diester precursor using powerful reducing agents such as sodium borohydride (NaBH4) in ethanol or lithium aluminum hydride (LiAlH4) in tetrahydrofuran. This step converts the ester moieties into primary alcohol groups, yielding 1,1'-bis(3-hydroxypropyl)-2,2'-biimidazole. Following this, the hydroxyl groups are activated through a halogenation reaction with thionyl chloride (SOCl2), transforming them into excellent leaving groups (chlorides) to form 1,1'-bis(3-chloropropyl)-2,2'-biimidazole. The final and most critical step involves a nucleophilic substitution reaction where the chloro-intermediate reacts with lithium diphenylphosphide (LiPPh2) under inert atmosphere. This reaction replaces the chlorine atoms with diphenylphosphino groups, successfully installing the phosphorus centers onto the biimidazole scaffold. The resulting trivalent phosphine can then be further derivatized via oxidation or sulfurization to tune its electronic properties for specific metal coordination tasks.
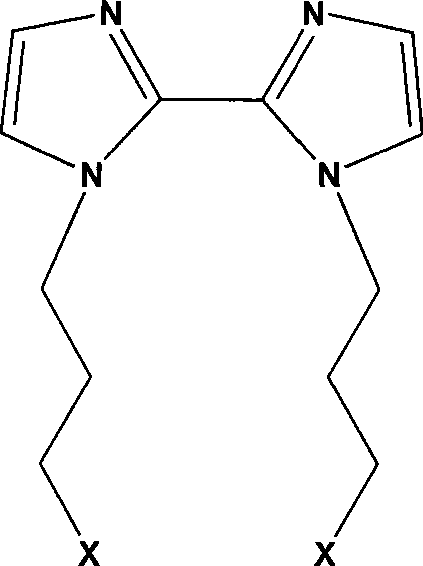
Beyond the primary phosphine synthesis, the patent details mechanisms for further functionalization that enhance the utility of these compounds. The trivalent phosphine group (-PPh2) is susceptible to oxidation by hydrogen peroxide or organic peracids to form phosphine oxides [-P(O)Ph2], or sulfurization by elemental sulfur to form phosphine sulfides [-P(S)Ph2]. These modifications alter the hard-soft acid-base characteristics of the ligand, allowing chemists to tailor the compound for binding specific metal ions, such as lanthanides for rare earth extraction or transition metals for homogeneous catalysis. The ability to systematically vary the 'X' group on the side chain while maintaining the integrity of the biimidazole core provides a versatile platform for material science applications. This level of mechanistic control ensures that the final products meet stringent purity specifications required for high-performance catalytic cycles, minimizing the risk of catalyst deactivation due to impurities.
How to Synthesize Biimidazole Phosphine Efficiently
The practical execution of this synthesis requires careful attention to reaction conditions, particularly regarding moisture and oxygen sensitivity during the phosphination step. The standardized protocol involves dissolving the chloro-intermediate in anhydrous ether solvents and slowly adding the lithium diphenylphosphide reagent under a nitrogen or argon blanket to prevent oxidation of the phosphine. Detailed standard operating procedures for each reaction stage, including specific molar ratios, temperature controls, and workup techniques, are essential for reproducibility. For a comprehensive guide on the exact experimental parameters and safety precautions required to execute this synthesis in a GMP-compliant environment, please refer to the technical documentation below.
- Reduce 1,1'-bis(alkyl propionate)-2,2'-biimidazole using NaBH4 or LiAlH4 to form the diol intermediate.
- Convert the diol to 1,1'-bis(3-chloropropyl)-2,2'-biimidazole via reaction with thionyl chloride.
- Perform nucleophilic substitution with lithium diphenylphosphide to yield the target biimidazole phosphine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this indirect synthetic route offers substantial strategic benefits beyond mere technical feasibility. The primary advantage lies in the use of readily available, commodity-grade starting materials such as alkyl propionates and common reducing agents, which stabilizes the supply chain against raw material volatility. Unlike processes relying on exotic or scarce reagents, this method utilizes chemicals that are produced on a massive global scale, ensuring consistent availability and predictable pricing structures. Furthermore, the elimination of complex purification steps associated with removing cyclization byproducts significantly streamlines the manufacturing workflow. This efficiency translates directly into reduced processing time and lower energy consumption per kilogram of finished product, driving down the overall cost of goods sold without compromising on quality standards.
- Cost Reduction in Manufacturing: The indirect synthesis route eliminates the need for extensive chromatographic purification often required to separate cyclic byproducts from linear targets in direct alkylation methods. By avoiding these resource-intensive purification steps, manufacturers can achieve significant operational savings in solvent usage and labor hours. Additionally, the high selectivity of the nucleophilic substitution step ensures that the majority of the starting material is converted into the desired product, maximizing atom economy. This efficient utilization of raw materials reduces waste generation and lowers the cost burden associated with waste disposal and environmental compliance, contributing to a more sustainable and economically viable production model.
- Enhanced Supply Chain Reliability: The reliance on stable ester intermediates rather than reactive dihaloalkanes mitigates the risks associated with handling hazardous materials during storage and transport. Ester precursors generally possess better shelf-life stability and are less prone to degradation, allowing for larger batch production and inventory buffering. This robustness ensures that production schedules can be maintained even during fluctuations in upstream supply, providing customers with greater certainty regarding lead times. The scalability of the process from laboratory benchtop to industrial reactor volumes has been demonstrated, confirming that supply continuity can be maintained as demand for high-purity biimidazole phosphines grows in the specialty chemicals sector.
- Scalability and Environmental Compliance: The reaction conditions employed in this patent, such as the use of ethanol or tetrahydrofuran as solvents, align well with established industrial safety protocols and waste management practices. The process avoids the generation of heavy metal waste streams often associated with transition-metal catalyzed coupling reactions, simplifying the environmental permitting process for manufacturing facilities. Moreover, the ability to recycle solvents and recover byproducts like lithium salts further enhances the green chemistry profile of the synthesis. This alignment with environmental, social, and governance (ESG) goals makes the technology attractive for multinational corporations seeking to reduce the carbon footprint of their chemical supply chains while maintaining high production throughput.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of biimidazole phosphine compounds. These answers are derived directly from the experimental data and claims presented in the patent literature, ensuring accuracy and relevance for industry professionals. Understanding these nuances is critical for evaluating the feasibility of integrating these ligands into your specific catalytic or material science applications.
Q: Why is the indirect synthesis route preferred over direct alkylation for biimidazole phosphines?
A: Direct alkylation of biimidazole with dihaloalkanes often fails due to intramolecular nucleophilic substitution, leading to cyclization rather than the desired linear chain extension. The indirect route via ester reduction avoids this side reaction.
Q: What are the primary applications of these biimidazole phosphine derivatives?
A: These compounds serve as excellent ligands for transition metals and lanthanides, finding use in organic catalysis, rare earth ion extraction, and as precursors for coordination-functionalized ionic liquids.
Q: Can the phosphine group be further modified after synthesis?
A: Yes, the trivalent phosphine group can be oxidized to phosphine oxides or sulfurized to phosphine sulfides, expanding the range of coordination properties and potential applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Biimidazole Phosphine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance ligands play in advancing catalytic technologies and material science. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the complex synthesis routes described in CN101676293B can be translated into reliable industrial supply. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to verify the structural integrity of every batch. Whether you require the trivalent phosphine, its oxide, or sulfide derivatives, our commitment to quality assurance guarantees that you receive materials that perform consistently in your downstream processes.
We invite you to engage with our technical procurement team to discuss your specific requirements for functionalized biimidazole compounds. By requesting a Customized Cost-Saving Analysis, you can explore how our optimized manufacturing processes can reduce your overall project costs. We encourage potential partners to contact us for specific COA data and route feasibility assessments tailored to your unique application needs, ensuring a seamless integration of our high-purity intermediates into your supply chain.
