Scalable One-Pot Hydrogenation Route for High-Purity 4-Dimethylaminopiperidine Hydrochloride
Scalable One-Pot Hydrogenation Route for High-Purity 4-Dimethylaminopiperidine Hydrochloride
The pharmaceutical industry constantly seeks robust, scalable, and cost-effective pathways for critical intermediates, and the synthesis of 4-dimethylaminopiperidine hydrochloride stands as a prime example where process innovation drives commercial value. Based on the technological breakthroughs detailed in patent CN101531628B, a novel one-pot reductive amination strategy has been established that fundamentally alters the production landscape for this vital piperidine derivative. This method bypasses the complex multi-step sequences of legacy protocols by directly coupling 4-piperidone hydrochloride with dimethylamine under catalytic hydrogenation conditions. For R&D directors and procurement specialists alike, this represents a shift towards streamlined manufacturing that minimizes unit operations while maximizing yield and purity profiles. The ability to produce this key building block with high efficiency addresses the growing demand for reliable pharmaceutical intermediate suppliers who can guarantee supply chain continuity without compromising on quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the manufacturing of 4-dimethylaminopiperidine derivatives has been plagued by inefficient synthetic routes that impose significant burdens on both cost and environmental compliance. The first conventional approach involves the use of N-protected 4-piperidone reacted with dimethylamine followed by reduction with sodium cyanoborohydride; this method is notoriously sluggish, requiring extended reaction times and generating toxic boron waste streams that complicate downstream processing. A second traditional pathway attempts the direct methylation of 4-aminopiperidine, yet this route suffers from poor stability of intermediates and creates a nightmare scenario for separation and purification, making industrial scale-up nearly impossible due to the formation of inseparable by-products. Furthermore, a third method involving the hydrogenation of 4-dimethylaminopyridine (DMAP) using precious metal catalysts like palladium carbon or platinum carbon often results in poor selectivity for the target piperidine ring saturation, leading to low transformation efficiency and inconsistent batch quality. These legacy issues collectively drive up the cost of goods sold and introduce unacceptable risks for supply chain managers seeking consistent API intermediate supplies.
The Novel Approach
In stark contrast to these cumbersome historical methods, the innovative process disclosed in the patent utilizes a direct, one-pot reductive amination of 4-piperidone hydrochloride with dimethylamine. This approach elegantly combines the imine formation and subsequent reduction steps into a single operational sequence within an autoclave, drastically reducing the number of isolation steps and solvent exchanges required. By employing a heterogeneous palladium on carbon (Pd/C) catalyst under controlled hydrogen pressure ranging from 0.3 to 1.5 MPa, the reaction achieves high conversion rates at moderate temperatures between 30 and 100°C. The simplicity of this workflow means that the crude product can be obtained merely by filtration and solvent evaporation, followed by a straightforward recrystallization to achieve purity levels exceeding 98%. This technological leap not only simplifies the operator's workload but also aligns perfectly with green chemistry principles by minimizing waste generation and energy consumption, thereby offering a compelling value proposition for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Pd/C-Catalyzed Reductive Amination
The core of this synthetic success lies in the efficient mechanistic pathway of catalytic reductive amination, which proceeds through a well-defined sequence of condensation and hydrogenation events. Initially, the nucleophilic attack of dimethylamine on the carbonyl carbon of 4-piperidone hydrochloride leads to the formation of a hemiaminal intermediate, which subsequently undergoes dehydration to generate an reactive iminium or enamine species in situ. This unsaturated intermediate is then immediately subjected to catalytic hydrogenation on the surface of the Pd/C catalyst, where molecular hydrogen is activated and transferred to the carbon-nitrogen double bond system. The use of a heterogeneous catalyst is particularly advantageous here, as it ensures that the active metal sites are accessible for the reduction while remaining easily separable from the reaction mixture via simple filtration post-reaction. This mechanism avoids the accumulation of unstable intermediates that plague other routes, ensuring a smooth progression to the saturated piperidine ring structure with high stereochemical integrity and minimal side reactions.
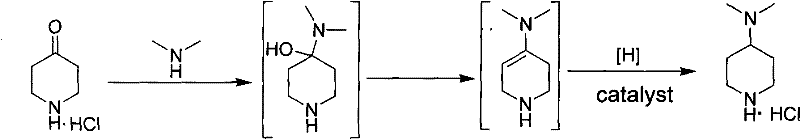
Controlling the impurity profile in this synthesis is critically managed through the optimization of reaction parameters and the final purification strategy. The patent specifies that maintaining the hydrogen pressure and temperature within the defined windows prevents over-reduction or the formation of polymeric by-products that could arise from uncontrolled exotherms. Furthermore, the choice of solvent system plays a pivotal role in impurity management; solvents such as methanol, ethanol, or mixtures with ethyl acetate are selected not only for their solubility properties but also for their ability to facilitate the crystallization of the target hydrochloride salt. The final recrystallization step acts as a powerful purification gate, leveraging the differential solubility of the product versus potential organic impurities or trace metal residues. This rigorous control over the chemical environment ensures that the final 4-dimethylaminopiperidine hydrochloride meets the stringent purity specifications required for medical production, providing R&D teams with confidence in the material's performance during downstream API synthesis.
How to Synthesize 4-Dimethylaminopiperidine Hydrochloride Efficiently
Executing this synthesis requires precise adherence to the patented protocol to ensure safety and optimal yield, beginning with the careful charging of raw materials into a pressure-rated reactor. The process initiates by mixing 4-piperidone hydrochloride with a solution of dimethylamine in a suitable alcoholic solvent, allowing the condensation to proceed under ambient stirring before the introduction of the catalyst. Once the catalyst is added and the system is purged, hydrogen is introduced to the specified pressure, and the mixture is heated to initiate the reduction phase, which continues until hydrogen uptake ceases indicating complete conversion. The detailed standardized synthesis steps, including specific molar ratios, catalyst loading percentages, and workup procedures, are outlined below to guide technical teams in replicating this high-efficiency route.
- Charge 4-piperidone hydrochloride and dimethylamine solution into an autoclave with a suitable alcoholic solvent and stir at room temperature to form the imine intermediate.
- Add Palladium on Carbon (Pd/C) catalyst, purge with hydrogen, and pressurize the system to 0.3-1.5 MPa while heating to 30-100°C.
- Maintain hydrogen pressure until uptake ceases, cool the reaction, filter off the catalyst, and purify the crude product via recrystallization to achieve >98% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route translates directly into tangible operational efficiencies and risk mitigation strategies. The elimination of complex protection-deprotection sequences and the avoidance of hazardous reducing agents like sodium cyanoborohydride significantly streamline the supply chain, reducing the dependency on specialized reagents that may face market volatility. By consolidating the reaction into a single pot, manufacturers can drastically reduce cycle times and labor costs associated with multiple isolation and drying steps, leading to a more agile production schedule that can respond quickly to fluctuating market demands. Furthermore, the use of common solvents and a recoverable heterogeneous catalyst enhances the overall sustainability profile of the manufacturing process, aligning with increasingly strict environmental regulations and corporate responsibility goals without sacrificing economic viability.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of the unit operations and the use of readily available, commodity-grade raw materials. By removing the need for expensive N-protecting groups and stoichiometric reducing agents, the direct material costs are substantially lowered, while the one-pot nature of the reaction reduces utility consumption and waste disposal fees. The ability to recycle the Pd/C catalyst or utilize it at low loadings further contributes to margin improvement, making the final product highly competitive in the global marketplace for fine chemical intermediates.
- Enhanced Supply Chain Reliability: Sourcing reliability is greatly improved because the key starting material, 4-piperidone hydrochloride, is a stable and widely produced chemical, unlike some specialized precursors required by older methods. The robustness of the reaction conditions, which tolerate a range of temperatures and pressures, ensures consistent batch-to-batch quality, minimizing the risk of production delays caused by failed runs or out-of-specification material. This stability allows supply chain planners to forecast inventory needs with greater accuracy and maintain safer stock levels of this critical intermediate.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing standard hydrogenation autoclaves found in most multipurpose chemical plants, which eliminates the need for costly capital investment in new equipment. From an environmental perspective, the absence of heavy metal waste streams associated with borohydride reductions and the ease of catalyst filtration simplify wastewater treatment and solid waste management. This compliance readiness ensures uninterrupted production even as regulatory scrutiny on chemical manufacturing intensifies, securing the long-term viability of the supply source.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 4-dimethylaminopiperidine hydrochloride, derived directly from the specific advantages and data points presented in the patent literature. Understanding these nuances helps stakeholders make informed decisions about integrating this material into their broader manufacturing workflows and quality assurance protocols. The answers reflect the consensus on best practices for handling, storing, and utilizing this intermediate to maximize its potential in drug synthesis.
Q: What are the primary advantages of this synthesis method over traditional N-protection routes?
A: Unlike traditional methods requiring N-protection and sodium cyanoborohydride reduction which suffer from long reaction times and difficult purification, this patented one-pot reductive amination utilizes direct hydrogenation with Pd/C, significantly simplifying the workflow and improving yield.
Q: How is high purity (>98%) achieved in the final product?
A: High purity is secured through a robust recrystallization step using mixed solvents such as methanol/ethyl acetate or ethanol/ethyl acetate, which effectively removes trace impurities and catalyst residues from the crude yellow solid.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for industrial amplification, utilizing standard autoclave equipment, readily available raw materials like 4-piperidone hydrochloride, and heterogeneous catalysis that allows for easy catalyst recovery and filtration.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Dimethylaminopiperidine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory patent to commercial reality requires a partner with deep technical expertise and unwavering commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising metrics of this one-pot hydrogenation route are fully realized in large-scale manufacturing. We operate with stringent purity specifications and utilize rigorous QC labs to verify that every batch of 4-dimethylaminopiperidine hydrochloride meets the exacting standards required for pharmaceutical applications, providing you with a secure and compliant supply chain foundation.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic advantages of switching to this method for your production needs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our capability as a trusted partner in delivering high-value chemical solutions.
