Scalable Electrochemical Synthesis of Beta-Trifluoromethyl Amides for Pharma
Introduction to Advanced Electrochemical Fluorination Technology
The pharmaceutical and agrochemical industries are constantly seeking robust methodologies to introduce trifluoromethyl groups into organic scaffolds, given the profound impact of the CF3 moiety on metabolic stability and lipophilicity. Patent CN109972165B discloses a groundbreaking electrochemical preparation method for beta-trifluoromethyl amide compounds, representing a significant leap forward in green synthetic chemistry. This technology utilizes aryl vinyl compounds and accessible trifluoromethyl sources, such as sodium trifluoromethanesulfinate (Langlois reagent), under electrochemical anodic oxidation conditions. Unlike traditional methods that rely on harsh chemical oxidants or precious metal catalysts, this approach leverages electricity as a clean reagent to drive the transformation. The process is characterized by its operational simplicity, mild reaction conditions typically ranging from 25°C to 40°C, and exceptional atom economy. For R&D directors and process chemists, this patent offers a compelling alternative to conventional trifluoromethylation strategies, promising a streamlined pathway to high-value intermediates essential for modern drug discovery and development.
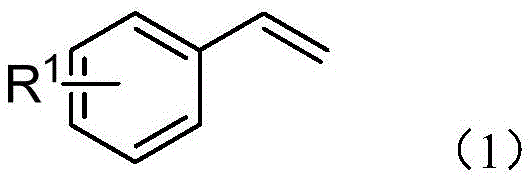
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the vicinal difunctionalization of alkenes, particularly amino-trifluoromethylation, has relied heavily on transition metal catalysis or photoredox systems. These conventional approaches often necessitate the use of expensive electrophilic trifluoromethylating reagents like Togni or Umemoto reagents, which are not only costly to synthesize but also generate significant stoichiometric waste. Furthermore, photoredox catalysis frequently requires specialized equipment and expensive iridium or ruthenium-based catalysts, creating bottlenecks in cost reduction for API manufacturing. The reliance on strong chemical oxidants in some protocols also raises safety concerns and complicates the impurity profile, requiring rigorous downstream purification to meet stringent pharmaceutical standards. These factors collectively hinder the widespread adoption of such methods for the commercial scale-up of complex pharmaceutical intermediates, driving the industry to seek more sustainable and economically viable alternatives.
The Novel Approach
The electrochemical method described in the patent fundamentally shifts the paradigm by replacing chemical oxidants with electrons. By employing a simple undivided cell setup with a carbon rod anode and a platinum cathode, the system generates trifluoromethyl radicals directly from inexpensive precursors via single-electron oxidation. This innovation effectively bypasses the need for noble metal catalysts and external oxidants, drastically simplifying the reaction mixture. As illustrated in the general reaction scheme below, the process seamlessly integrates the trifluoromethyl group and an amide functionality derived from the solvent (acetonitrile) onto the alkene substrate. This dual functionalization occurs under mild conditions with high functional group tolerance, accommodating various substituents on the aromatic ring. The result is a highly efficient synthesis that aligns perfectly with the principles of green chemistry while delivering superior economic benefits for procurement teams.
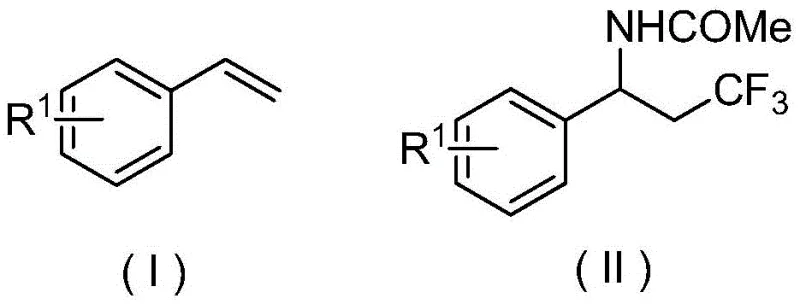
Mechanistic Insights into Electrochemical Anodic Oxidation
The core of this technology lies in the electrochemical anodic oxidation mechanism, which facilitates the generation of reactive radical species without the accumulation of hazardous byproducts. In this system, the trifluoromethyl source, typically sodium trifluoromethanesulfinate, undergoes anodic oxidation at the electrode surface to release a trifluoromethyl radical and sulfur dioxide. Simultaneously, the aryl vinyl substrate interacts with these radicals, initiating a cascade that leads to the formation of a carbon-centered radical intermediate. This intermediate is subsequently trapped by the nitrile group of the acetonitrile solvent, which acts as both the reaction medium and the nitrogen source, followed by hydrolysis during the workup to yield the final amide. This elegant mechanism ensures that the reaction proceeds with high selectivity, minimizing the formation of side products that often plague radical chemistry. The use of supporting electrolytes such as tetra-n-butylammonium hexafluorophosphate further enhances conductivity and stability, ensuring consistent performance across different substrate scopes.
From an impurity control perspective, the absence of transition metals is a critical advantage for pharmaceutical applications. Traditional metal-catalyzed routes often leave trace amounts of heavy metals in the final product, necessitating expensive scavenging steps to comply with ICH Q3D guidelines. In contrast, this electrochemical protocol produces a cleaner crude reaction mixture, primarily containing organic byproducts that are easier to separate via standard extraction and chromatography. The mild temperature range of 25°C to 40°C also prevents thermal degradation of sensitive functional groups, preserving the integrity of complex molecular architectures. This high level of purity and selectivity reduces the burden on quality control laboratories and accelerates the timeline from bench-scale optimization to pilot plant production, making it an attractive option for reliable pharmaceutical intermediate suppliers aiming to deliver high-purity materials.
How to Synthesize Beta-Trifluoromethyl Amides Efficiently
Implementing this electrochemical synthesis requires careful attention to reaction parameters to maximize yield and reproducibility. The patent outlines a robust procedure where aryl vinyl compounds are reacted with trifluoromethyl sources in a mixed solvent system of acetonitrile and dichloromethane. The process operates at a constant direct current, typically between 5 mA and 20 mA, depending on the specific substrate and scale. Detailed standardized synthesis steps, including precise molar ratios and workup procedures, are provided in the technical documentation to ensure successful replication. Following the reaction, a straightforward quenching and extraction protocol involving ethyl acetate and saturated brine allows for the isolation of the target beta-trifluoromethyl amide with high efficiency.
- Prepare the reaction system using aryl vinyl compounds and a trifluoromethyl source such as sodium trifluoromethanesulfinate in an acetonitrile-based solvent.
- Apply electrochemical anodic oxidation using a carbon rod anode and platinum cathode at a constant current of 5-20 mA at room temperature.
- Quench the reaction with ethyl acetate, perform aqueous workup with saturated brine, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this electrochemical methodology offers tangible strategic benefits beyond mere technical novelty. The primary advantage lies in the substantial cost savings achieved by eliminating expensive catalysts and oxidants from the bill of materials. By utilizing commodity chemicals like sodium trifluoromethanesulfinate and common solvents, the raw material costs are significantly reduced compared to processes relying on proprietary fluorinating reagents. Additionally, the simplified workup procedure reduces solvent consumption and waste disposal costs, contributing to a lower overall cost of goods sold (COGS). This economic efficiency is crucial for maintaining competitiveness in the global market for fine chemical intermediates, where margin pressure is constant.
- Cost Reduction in Manufacturing: The elimination of noble metal catalysts such as iridium or ruthenium removes a major cost driver associated with traditional photoredox methods. Furthermore, the avoidance of stoichiometric chemical oxidants reduces the expense of reagents and the subsequent cost of treating oxidative waste streams. This qualitative shift in the cost structure allows for more competitive pricing strategies without compromising on quality, enabling manufacturers to offer better value to their clients in the pharmaceutical sector.
- Enhanced Supply Chain Reliability: The reagents required for this electrochemical process, including aryl vinyl compounds and Langlois reagent, are commercially available from multiple global suppliers, reducing the risk of single-source dependency. The robustness of the reaction conditions, which tolerate ambient temperatures and do not require cryogenic cooling or high-pressure equipment, ensures that production can proceed without interruption due to utility failures. This reliability is essential for securing long-term supply contracts and meeting the just-in-time delivery expectations of major pharmaceutical companies.
- Scalability and Environmental Compliance: Electrochemical synthesis is inherently scalable, as increasing production capacity often involves adding more electrode surface area or running reactors in parallel rather than dealing with the heat transfer limitations of exothermic chemical oxidations. The green nature of the process, characterized by minimal waste generation and the absence of toxic heavy metals, aligns with increasingly stringent environmental regulations. This compliance reduces the regulatory burden and facilitates smoother audits, ensuring uninterrupted supply continuity for critical drug intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the electrochemical preparation of beta-trifluoromethyl amides. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the technology's capabilities. Understanding these aspects helps stakeholders make informed decisions about integrating this method into their supply chains.
Q: What are the advantages of this electrochemical method over traditional photoredox catalysis?
A: This method eliminates the need for expensive photocatalysts and stoichiometric oxidants, significantly reducing raw material costs and simplifying the purification process by avoiding heavy metal residues.
Q: What is the typical yield range for this synthesis?
A: According to patent data, the process achieves high yields ranging from 85% to 94% across various substituted styrene substrates, demonstrating excellent efficiency.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the use of mild reaction conditions (room temperature), inexpensive electrolytes, and simple workup procedures makes this electrochemical route highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Trifluoromethyl Amide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in modern drug development. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like this are successfully translated into industrial reality. Our state-of-the-art facilities are equipped with advanced electrochemical reactors and stringent purity specifications are maintained through our rigorous QC labs, guaranteeing that every batch meets the highest international standards. We are committed to leveraging cutting-edge technologies to provide our partners with superior chemical solutions that drive their projects forward efficiently.
We invite you to collaborate with us to explore the full potential of this electrochemical route for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your target molecule, demonstrating how this method can optimize your budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a stable, cost-effective supply of high-quality intermediates for your pipeline.
