Scalable Synthesis of 5-Azaspiro[2.4]heptane-6-Carboxylic Acid for HCV Inhibitor Manufacturing
The pharmaceutical industry's relentless pursuit of effective Hepatitis C virus (HCV) treatments has placed immense pressure on the supply chains of critical intermediates, specifically complex spirocyclic amino acids. Patent CN103687489A introduces a transformative methodology for the preparation of 5-azaspiro[2.4]heptane-6-carboxylic acid and its derivatives, which serve as pivotal building blocks in the synthesis of NS5A inhibitors. This intellectual property addresses long-standing bottlenecks in the manufacturing of these high-value compounds by replacing hazardous, low-yielding protocols with a robust, scalable chemical sequence. For R&D directors and procurement strategists, understanding this shift is vital, as it promises not only enhanced safety profiles but also significant improvements in process reliability and cost-efficiency for reliable pharmaceutical intermediate supplier networks globally.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the spirocyclopropyl moiety found in 4-spirocyclopropyl proline derivatives has relied heavily on the Simmons-Smith reaction or its variants. These traditional pathways typically employ diethylzinc (Et2Zn) in combination with diiodomethane or chloroiodomethane to generate metal carbenoids. While chemically effective on a small laboratory scale, these methods suffer from severe drawbacks when translated to industrial manufacturing. Diethylzinc is notoriously pyrophoric, igniting spontaneously upon contact with air, which necessitates specialized handling equipment, inert atmosphere protocols, and rigorous safety measures that drive up operational costs. Furthermore, these reactions often experience incomplete conversion, leading to difficult purification challenges and suboptimal yields that erode profit margins. The generation of zinc waste streams also complicates environmental compliance, making these legacy routes increasingly unattractive for modern cost reduction in API manufacturing initiatives.
The Novel Approach
The methodology disclosed in the patent circumvents these hazards by utilizing a dihalocarbene addition strategy followed by reductive hydrodehalogenation. Instead of unstable organozinc reagents, the process employs thermally stable trihaloacetic acid salts, such as sodium tribromoacetate, to generate the necessary carbene species in situ. This shift eliminates the fire risks associated with pyrophoric materials and allows for reactions to be conducted under much more manageable conditions. The subsequent removal of the halogen atoms via radical-mediated reduction ensures high fidelity in forming the final spirocyclic ring system. By decoupling the cyclopropanation from sensitive metal carbenoids, this approach offers a streamlined pathway that is inherently safer and more amenable to commercial scale-up of complex spiro compounds.
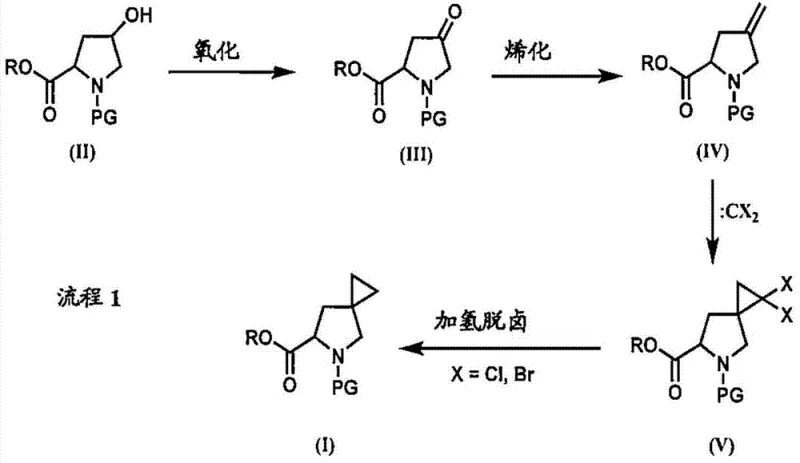
Mechanistic Insights into Dihalocarbene Cyclopropanation and Radical Reduction
The core innovation lies in the precise orchestration of four distinct chemical transformations that convert readily available 4-hydroxy-proline derivatives into the target spirocyclic architecture. The sequence initiates with the oxidation of the hydroxyl group to a ketone, typically achieved using TEMPO-mediated catalytic systems which offer excellent chemoselectivity without over-oxidizing the sensitive amine functionality. Following this, the ketone undergoes olefination, often via a Wittig reaction using methyltriphenylphosphonium bromide, to install an exocyclic methylene group. This alkene serves as the dipolarophile for the critical cyclopropanation step, where dihalocarbene adds across the double bond to form a dihalocyclopropane intermediate. The final transformation involves a reductive hydrodehalogenation, where radical initiators like AIBN配合 silanes or hypophosphorous acid facilitate the clean removal of halogen atoms, locking in the spiro-geometry with high stereochemical integrity.
From an impurity control perspective, this mechanism offers distinct advantages over metal-carbene routes. The use of defined radical precursors minimizes the formation of polymeric byproducts often seen with zinc carbenoids. Additionally, the intermediate dihalocyclopropane species is sufficiently stable to allow for isolation or in-process monitoring, providing quality control checkpoints that are absent in one-pot Simmons-Smith reactions. This level of control is essential for producing high-purity HCV inhibitor intermediate batches that meet stringent regulatory specifications. The ability to tune the reduction step—choosing between tin hydrides, silanes, or phosphorus-based reductants—further allows process chemists to optimize the removal of halogenated impurities, ensuring a clean final product profile.
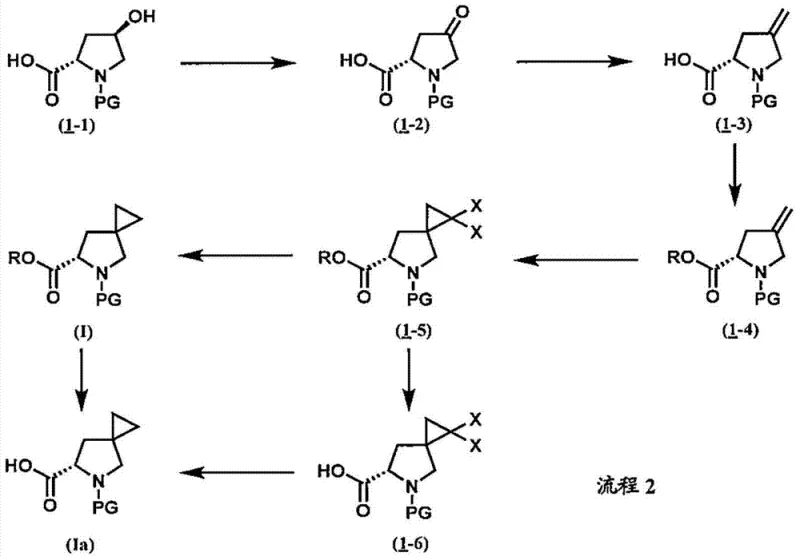
How to Synthesize 5-Azaspiro[2.4]heptane-6-Carboxylic Acid Efficiently
Implementing this synthesis requires careful attention to reaction parameters, particularly during the thermal generation of dihalocarbene and the subsequent radical reduction. The process begins with the protection and oxidation of the proline scaffold, followed by methylenation to create the reactive alkene. The cyclopropanation step typically utilizes phase transfer catalysts to enhance the efficiency of carbene transfer from the trihaloacetate salt. Finally, the hydrodehalogenation is conducted under reflux conditions with appropriate radical initiators to ensure complete conversion. For detailed standard operating procedures and specific stoichiometric ratios validated by experimental data, please refer to the technical guide below.
- Oxidize 4-hydroxy-proline derivatives to the corresponding ketone using TEMPO/NaClO systems.
- Convert the ketone to an exocyclic alkene via Wittig olefination or Tebbe reagent treatment.
- Perform cyclopropanation using dihalocarbene generated from trihaloacetic acid salts, followed by radical-mediated hydrodehalogenation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this patented process represents a strategic opportunity to de-risk the sourcing of critical antiviral intermediates. By eliminating the dependency on hazardous, air-sensitive reagents like diethylzinc, manufacturers can significantly reduce the overhead costs associated with specialized storage, handling, and waste disposal. The inherent stability of the reagents used in this new route translates directly into improved batch-to-batch consistency and reduced downtime caused by safety incidents or purification failures. This reliability is crucial for maintaining continuous supply lines in the fast-paced antiviral market, where delays can have cascading effects on drug availability.
- Cost Reduction in Manufacturing: The replacement of expensive and hazardous organozinc reagents with commodity chemicals like sodium tribromoacetate and common radical initiators leads to substantial raw material cost savings. Furthermore, the simplified workup procedures and higher yields reduce the consumption of solvents and chromatography media, driving down the overall cost of goods sold. The avoidance of complex metal removal steps also lowers the burden on downstream processing infrastructure, contributing to a leaner manufacturing model.
- Enhanced Supply Chain Reliability: Sourcing pyrophoric reagents often involves long lead times and strict regulatory compliance checks, creating bottlenecks in the supply chain. In contrast, the reagents required for this novel process are widely available from multiple global suppliers, ensuring a resilient supply base. This diversification mitigates the risk of single-source failures and allows for more flexible inventory management, ultimately reducing lead time for high-purity amino acids and intermediates.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that are easily transferred from pilot plants to multi-ton production facilities. The reduction in hazardous waste generation, particularly zinc-containing effluents, simplifies environmental permitting and reduces the ecological footprint of the manufacturing site. This alignment with green chemistry principles not only satisfies regulatory requirements but also enhances the corporate sustainability profile of the supply chain partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of adopting this technology for their specific production needs.
Q: Why is the new process safer than the traditional Simmons-Smith reaction?
A: The traditional method relies on diethylzinc (Et2Zn), which is pyrophoric and poses significant fire hazards. The patented process utilizes stable trihaloacetic acid salts and radical reagents, drastically improving operational safety.
Q: What represents the key intermediate in this synthesis route?
A: The key intermediate is the dihalocyclopropane derivative (Formula V), which is formed via the addition of dihalocarbene to the exocyclic double bond before undergoing reductive removal of the halogens.
Q: Can this process be scaled for commercial API production?
A: Yes, the elimination of air-sensitive reagents and the use of robust radical reduction conditions make this pathway highly suitable for large-scale commercial manufacturing of HCV inhibitor intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Azaspiro[2.4]heptane-6-Carboxylic Acid Supplier
As the demand for next-generation HCV therapies continues to evolve, having a manufacturing partner with deep expertise in complex spirocyclic chemistry is indispensable. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from development to market. Our state-of-the-art facilities are equipped to handle the specific requirements of radical chemistry and sensitive intermediates, adhering to stringent purity specifications and rigorous QC labs to guarantee product quality that exceeds industry standards.
We invite you to collaborate with our technical team to explore how this advanced synthesis route can optimize your supply chain. Contact us today to request a Customized Cost-Saving Analysis tailored to your volume requirements. Our experts are ready to provide specific COA data and route feasibility assessments, helping you secure a competitive advantage in the global pharmaceutical market through our dedicated technical procurement team.
