Advanced Asymmetric Mannich Catalysis for High-Purity Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking more efficient, environmentally benign pathways to access chiral building blocks, particularly for the synthesis of complex active pharmaceutical ingredients (APIs). A groundbreaking approach detailed in patent CN101835745A introduces a novel method for preparing optically active 1,1'-diaryl-2,2'-disulfonic acid compounds, which serve as potent asymmetric Mannich catalysts. Unlike traditional synthetic routes that rely on hazardous brominating agents or toxic heavy metal oxidants, this innovation leverages pressurized oxygen oxidation to convert dithiol precursors directly into high-purity disulfonate salts. This technological leap not only addresses critical environmental compliance issues but also establishes a robust foundation for the reliable production of beta-amino carbonyl derivatives, which are indispensable scaffolds in medicinal chemistry. For R&D directors and procurement specialists alike, this represents a significant opportunity to streamline supply chains for high-value chiral intermediates while drastically reducing the ecological footprint of manufacturing processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,1'-dinaphthalene-2,2'-disulfonic acid has been fraught with reproducibility challenges and safety concerns. Prior art, such as that disclosed in Japanese Patent Document 1, typically employs N-bromosuccinimide (NBS) to introduce sulfonyl groups, a process that generates stoichiometric amounts of succinimide waste and requires rigorous purification steps to remove bromine residues. Furthermore, alternative oxidation strategies using chromic acid, periodate, or Oxone have proven largely unsuccessful for dithiol substrates due to the high propensity for forming intramolecular disulfide byproducts rather than the desired sulfonic acids. These conventional methods often suffer from low yields, inconsistent enantiomeric retention, and the generation of hazardous liquid waste streams containing heavy metals like chromium or ruthenium. For a commercial manufacturer, these factors translate into elevated operational costs, complex waste management protocols, and potential supply chain disruptions caused by the volatility of specialized reagent availability.
The Novel Approach
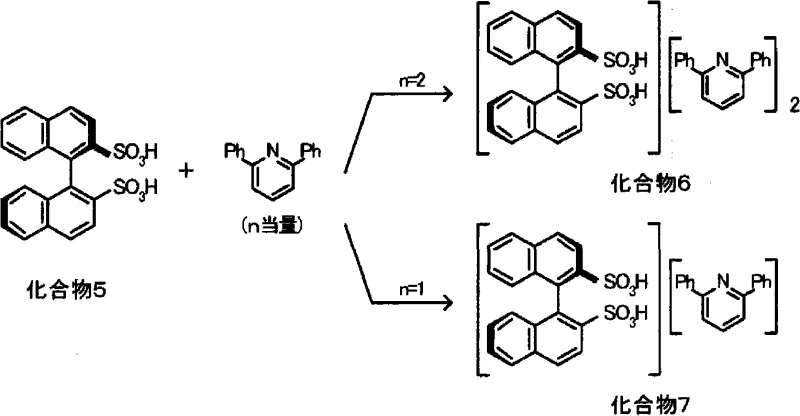
The patented methodology revolutionizes this landscape by utilizing molecular oxygen as the sole oxidant in the presence of a strong base like potassium hydroxide and a polar aprotic solvent such as hexamethylphosphoramide (HMPA). By conducting the reaction under pressurized oxygen conditions, specifically at pressures ranging from 5 to 7 atmospheres and temperatures around 80°C, the process achieves high conversion rates without the formation of disulfide impurities. This direct oxidation pathway allows for the quantitative recovery of the disulfonic acid after simple ion-exchange chromatography. The resulting chiral acid can then be effortlessly converted into an active catalyst by mixing with 2,6-disubstituted pyridines. This modular approach enables rapid tuning of the catalyst's steric and electronic properties simply by varying the pyridine substituent, offering a level of flexibility that covalent modification of the naphthalene backbone cannot match. Consequently, this method provides a scalable, cost-effective, and environmentally superior route to accessing high-performance asymmetric catalysts.
Mechanistic Insights into Oxygen-Mediated Disulfonic Acid Synthesis
The core mechanistic advantage of this invention lies in the unique ability of the alkaline oxygen system to overcome the kinetic barrier of sulfonic acid formation while suppressing thermodynamic sinks like disulfide dimerization. In the presence of excess potassium hydroxide (preferably 3 to 6 equivalents), the thiol protons of the 1,1'-dinaphthyl-2,2'-dithiol are deprotonated to form thiolate anions. Under pressurized oxygen, these anions undergo a stepwise oxidation where the sulfur atoms are sequentially transformed into sulfinate and finally sulfonate species. The use of HMPA as a solvent is critical, as its high polarity and coordinating ability stabilize the charged intermediates and facilitate the dissolution of oxygen, thereby accelerating the reaction kinetics. This careful balance of reaction parameters ensures that the chiral axis of the binaphthyl skeleton remains intact throughout the harsh oxidative conditions, preserving the optical integrity required for subsequent asymmetric induction. The result is a potassium disulfonate salt that can be isolated in high yield, serving as a stable precursor for the final catalytic species.
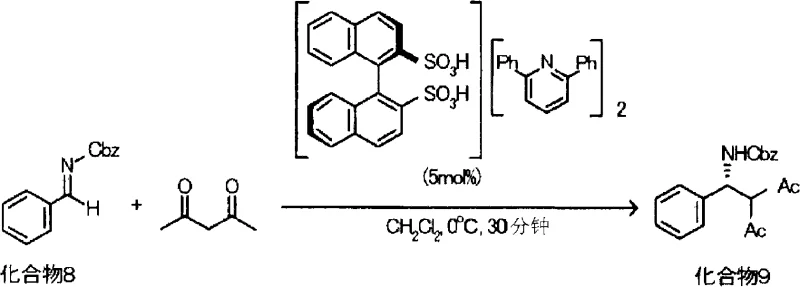
Once the disulfonic acid is generated, it forms a chiral ion-pair complex with 2,6-disubstituted pyridines, creating a confined chiral environment essential for high stereoselectivity. In the subsequent Mannich reaction, this Brønsted acid catalyst activates the aldimine substrate through hydrogen bonding or protonation, increasing its electrophilicity towards the nucleophilic attack by the 1,3-dicarbonyl compound. The bulky aryl groups on the pyridine ring, combined with the rigid binaphthyl backbone, create a sterically demanding pocket that effectively differentiates between the enantiotopic faces of the reacting species. This precise spatial arrangement directs the formation of the new carbon-carbon bond with exceptional stereocontrol, often achieving enantiomeric excess (ee) values exceeding 90%. The mechanism avoids the need for transition metals, thereby eliminating the risk of metal contamination in the final pharmaceutical product, a crucial consideration for regulatory compliance in API manufacturing.
How to Synthesize (R)-1,1'-Binaphthyl-2,2'-disulfonic Acid Efficiently
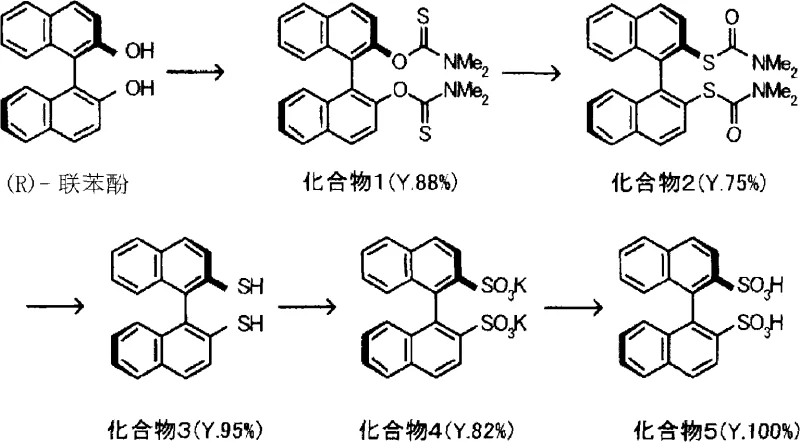
The synthesis of this high-value chiral catalyst precursor involves a streamlined sequence starting from commercially available (R)-binaphthol. The process begins with the conversion of the hydroxyl groups to thiocarbamates, followed by thermal rearrangement to install the sulfur atoms, and finally, the critical oxygen oxidation step. Detailed operational parameters, including specific solvent ratios, temperature gradients, and workup procedures, are essential for maximizing yield and optical purity. The following guide outlines the standardized protocol derived from the patent examples to ensure reproducible results in a laboratory or pilot plant setting.
- Oxidize optically active 1,1'-diaryl-2,2'-dithiol compounds using pressurized oxygen (5-7 atm) in the presence of potassium hydroxide and HMPA at 80°C to form the disulfonate salt.
- Convert the salt to the free disulfonic acid using an ion-exchange resin, then mix with 2,6-disubstituted pyridine (molar ratio 1: 0.75 to 3) to form the active catalyst complex.
- React the catalyst with N-protected aldimine and a 1,3-dicarbonyl compound in dichloromethane or acetonitrile at 0°C to 30°C to obtain the beta-amino carbonyl derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this oxygen-based oxidation technology offers profound strategic benefits beyond mere technical performance. The elimination of expensive and regulated reagents like N-bromosuccinimide or heavy metal oxidants significantly simplifies the raw material sourcing strategy. By relying on commodity chemicals such as oxygen gas, potassium hydroxide, and standard organic solvents, manufacturers can insulate their production costs from the volatility associated with specialty reagent markets. Furthermore, the removal of heavy metals from the process flow drastically reduces the complexity and cost of wastewater treatment and hazardous waste disposal. This aligns perfectly with modern green chemistry initiatives and corporate sustainability goals, potentially lowering insurance premiums and regulatory compliance burdens. The robustness of the reaction conditions also implies a lower risk of batch failures, ensuring a more consistent and reliable supply of critical intermediates for downstream drug synthesis.
- Cost Reduction in Manufacturing: The substitution of stoichiometric oxidants with catalytic or reagent-grade oxygen represents a fundamental shift in cost structure. Traditional methods often require multiple equivalents of expensive oxidizing agents, the byproducts of which add mass and complexity to the purification stage. By contrast, the oxygen oxidation method generates water as the primary byproduct, minimizing the load on downstream separation units like chromatography columns or crystallizers. This reduction in material consumption and waste processing volume translates directly into substantial cost savings per kilogram of product. Additionally, the avoidance of transition metal catalysts removes the need for costly metal scavenging steps, which are often mandatory to meet strict ppm limits in pharmaceutical grades, further enhancing the overall economic efficiency of the manufacturing process.
- Enhanced Supply Chain Reliability: Dependence on single-source suppliers for complex chiral ligands or specialized oxidants poses a significant risk to supply chain continuity. The reagents required for this novel synthesis—oxygen, base, and simple solvents—are globally available commodities with mature supply networks. This diversification of input materials mitigates the risk of production stoppages due to vendor shortages or logistical bottlenecks. Moreover, the stability of the intermediate potassium salt allows for flexible inventory management; the salt can be produced in bulk and stored, then converted to the free acid or catalyst on demand. This decoupling of production stages provides supply chain planners with greater agility to respond to fluctuating market demands without compromising on lead times or product quality.
- Scalability and Environmental Compliance: Scaling chemical processes often exposes hidden safety and environmental hazards, but this oxidation method is inherently designed for scalability. The use of pressurized oxygen is a well-understood unit operation in the fine chemical industry, and the reaction temperatures are moderate, reducing energy consumption and thermal runaway risks. From an environmental perspective, the process generates minimal hazardous waste, avoiding the discharge of heavy metals like chromium or ruthenium into the ecosystem. This clean profile facilitates easier permitting for new production facilities and ensures long-term operational viability in regions with stringent environmental regulations. The ability to produce high-purity intermediates with a reduced environmental footprint positions manufacturers favorably in an increasingly eco-conscious global marketplace.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this asymmetric catalytic system. These insights are derived directly from the experimental data and scope analysis provided in the patent documentation, offering practical guidance for chemists looking to integrate this technology into their existing workflows. Understanding the nuances of solvent effects, substrate scope, and catalyst loading is key to unlocking the full potential of this methodology for diverse synthetic applications.
Q: What are the advantages of using oxygen oxidation over traditional methods for disulfonic acid synthesis?
A: Traditional methods often rely on hazardous reagents like N-bromosuccinimide (NBS) or heavy metal oxidants which generate toxic waste and are difficult to reproduce. The patented oxygen oxidation method uses eco-friendly pressurized oxygen, eliminating heavy metal contamination and simplifying purification while maintaining high optical purity.
Q: How does the molar ratio of pyridine affect the enantiomeric excess (ee) in this Mannich reaction?
A: The patent data indicates that the molar ratio of 2,6-disubstituted pyridine to the disulfonic acid is critical. Ratios between 0.75 and 3 are effective, with optimal results often observed around 1:2. Ratios below 0.75 significantly reduce ee values, while excessive amine can lower acidity and yield.
Q: Can this catalyst system be scaled for industrial production of API intermediates?
A: Yes, the process utilizes standard industrial reagents like potassium hydroxide and oxygen gas under moderate pressure (5-7 atm) and temperature (80°C). The absence of sensitive transition metal catalysts and the use of robust solvents like dichloromethane or acetonitrile make it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R)-1,1'-Binaphthyl-2,2'-disulfonic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this advanced catalytic technology for the next generation of chiral pharmaceuticals. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate these innovative laboratory protocols into robust, commercial-scale manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from R&D to market is seamless and efficient. We maintain stringent purity specifications across all our product lines, supported by rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify enantiomeric excess and chemical purity. Whether you require custom synthesis of the disulfonic acid precursor or the fully formulated catalyst system, our facility is ready to support your volume requirements with unwavering consistency.
We invite you to collaborate with us to explore how this green oxidation technology can optimize your specific synthetic routes. Our technical sales team is prepared to provide a Customized Cost-Saving Analysis tailored to your current production metrics, highlighting exactly where efficiencies can be gained. Please contact our technical procurement team today to request specific COA data for our chiral intermediates and to discuss route feasibility assessments for your upcoming projects. Let us be your partner in driving innovation and efficiency in your supply chain.
