Revolutionizing Pyrazolo[1,3,5]triazine Production: A Catalyst-Free One-Pot Strategy for Commercial Scale
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access privileged scaffolds that mimic biological motifs. Patent CN113801119B introduces a groundbreaking synthetic methodology for constructing pyrazolo[1,3,5]triazine derivatives, a class of compounds recognized as potent electronic isosteres of purines. These structures are critical in medicinal chemistry for targeting purine signaling receptors and enzymes, offering therapeutic potential as kinase inhibitors and receptor antagonists. The disclosed technology represents a significant leap forward by enabling the direct assembly of these complex heterocycles through a simple, one-pot series reaction. Unlike traditional multi-step sequences that often suffer from low overall yields and cumbersome isolation procedures, this novel approach utilizes readily available aromatic aldehydes, 3-aminopyrazoles, and ammonium iodide. The process is driven by oxidative cyclization under mild thermal conditions, eliminating the need for expensive transition metal catalysts. This innovation not only streamlines the synthetic route but also aligns perfectly with modern green chemistry principles, making it an attractive candidate for industrial adoption.
![General reaction scheme for synthesizing pyrazolo[1,3,5]triazine compounds from aromatic aldehydes and 3-aminopyrazoles](/insights/img/pyrazolo-triazine-synthesis-pharma-intermediate-supplier-20260304060737-01.webp)
Historically, the construction of the pyrazolo[1,3,5]triazine core has relied heavily on the condensation of 3-aminopyrazoles with pre-prepared nitrogen-containing 1,3-dielectrophiles or through the heterocyclization of N-pyrazolyl amidines. While these conventional methods are chemically sound, they present substantial logistical and economic challenges for large-scale manufacturing. The requirement for pre-synthesized, often unstable electrophilic reagents increases the step count, thereby reducing the overall atom economy and increasing waste generation. Furthermore, many existing protocols necessitate the use of harsh reaction conditions or specialized catalysts that require rigorous removal steps to meet pharmaceutical purity standards. The presence of metal residues, in particular, poses a significant regulatory hurdle for API intermediates, often demanding additional purification technologies like scavenging resins or recrystallization, which drive up production costs and extend lead times. These limitations have historically restricted the widespread application of pyrazolo[1,3,5]triazine scaffolds in drug discovery pipelines due to supply chain bottlenecks and high material costs.
In stark contrast, the novel approach detailed in CN113801119B dismantles these barriers by employing a direct oxidative coupling strategy. By leveraging ammonium iodide as a nitrogen source and promoter alongside common oxidants like di-tert-butyl peroxide (DTBP) or even molecular oxygen, the reaction achieves high efficiency without external metal catalysts. This catalyst-free design is a game-changer for process chemistry, as it inherently removes the risk of heavy metal contamination, simplifying the downstream processing significantly. The reaction proceeds smoothly in high-boiling solvents such as o-dichlorobenzene or chlorobenzene at temperatures ranging from 130°C to 150°C. The robustness of this method is evidenced by its tolerance to a wide array of functional groups, allowing for the late-stage diversification of the scaffold. This operational simplicity translates directly into reduced operational expenditure (OpEx) and capital expenditure (CapEx) for manufacturing facilities, as it avoids the need for specialized inert atmosphere equipment when air is used as the oxidant.
Mechanistic Insights into Oxidative Cyclization
The mechanistic pathway of this transformation involves a fascinating cascade of condensation and oxidation events that construct the triazine ring system with high precision. Initially, the aromatic aldehyde reacts with the 3-aminopyrazole and ammonium iodide to form an intermediate imine or hemiaminal species. The presence of the oxidant is crucial here, as it facilitates the dehydrogenative aromatization required to close the triazine ring. The iodide ion likely plays a dual role, acting both as a nucleophilic catalyst to activate the aldehyde and as a redox mediator in the oxidation cycle. This intricate interplay ensures that the reaction proceeds with high selectivity, minimizing the formation of side products such as over-oxidized species or polymerization byproducts. The ability to control the oxidation state precisely without metal catalysts suggests a radical or ionic mechanism that is highly dependent on the thermal energy provided. Understanding this mechanism is vital for R&D teams aiming to optimize the process further, as it highlights the importance of oxidant stoichiometry and temperature control in maximizing yield and purity.
From an impurity control perspective, the absence of transition metals drastically simplifies the impurity profile of the final product. In traditional catalytic systems, metal-ligand complexes can sometimes lead to difficult-to-remove organometallic impurities that persist through multiple purification steps. Here, the primary byproducts are likely derived from the decomposition of the oxidant or minor condensation variants, which are typically organic in nature and easier to separate via standard crystallization or chromatography. The patent data indicates that simple aqueous workups followed by silica gel chromatography are sufficient to achieve high purity levels, as demonstrated by the clean NMR spectra and high-resolution mass spectrometry data provided for compounds like 3a and 3b. This clean reaction profile is particularly advantageous for GMP manufacturing, where controlling genotoxic impurities and elemental impurities is paramount. The robustness of the reaction against moisture and air (in some embodiments) further enhances its reliability, ensuring consistent batch-to-batch quality which is essential for regulatory filings.
How to Synthesize Pyrazolo[1,3,5]triazine Efficiently
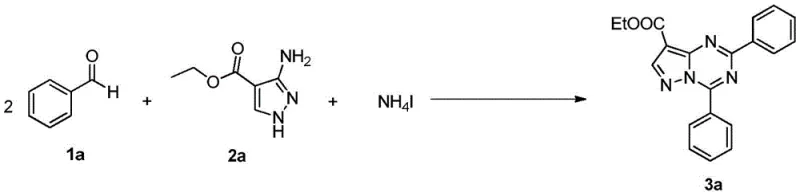
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent ratios and thermal management to ensure optimal conversion. The general protocol involves dissolving the aromatic aldehyde, 3-aminopyrazole derivative, and ammonium iodide in a suitable high-boiling solvent. The choice of oxidant can be tailored based on availability and safety considerations, with options ranging from organic peroxides to gaseous oxygen. The reaction mixture is then heated to the specified temperature range, where the oxidative cyclization takes place over a period of approximately 8 hours. Following the reaction, a straightforward quenching procedure with water allows for the extraction of the product into an organic phase. This operational simplicity makes the technology highly transferable from bench scale to commercial production, reducing the learning curve for process engineers. For a detailed visual representation of a specific successful run, refer to the reaction scheme for compound 3a below.
- Dissolve aromatic aldehyde, 3-aminopyrazole, and ammonium iodide in a high-boiling solvent like o-dichlorobenzene.
- Add an oxidant such as di-tert-butyl peroxide (DTBP) or utilize air oxygen under open conditions.
- Heat the mixture to 130-150°C for approximately 8 hours, then quench with water and extract with ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible strategic advantages that go beyond mere chemical elegance. The primary benefit lies in the drastic simplification of the raw material supply chain. Since the method utilizes commodity chemicals like aromatic aldehydes and ammonium iodide, sourcing is straightforward and less susceptible to the volatility associated with specialized catalysts or custom-synthesized intermediates. This reliance on bulk chemicals ensures a stable supply base, reducing the risk of production stoppages due to material shortages. Furthermore, the elimination of catalyst removal steps translates into significant time savings during the manufacturing cycle. In a commercial context, shorter cycle times mean higher throughput and better asset utilization, allowing manufacturers to respond more agilely to market demand fluctuations without needing to expand physical capacity.
- Cost Reduction in Manufacturing: The economic impact of removing transition metal catalysts cannot be overstated. Precious metal catalysts are not only expensive to purchase but also incur hidden costs related to recovery, recycling, or disposal. By operating without these metals, the process eliminates the need for expensive scavenging resins and the associated validation testing for residual metals. Additionally, the one-pot nature of the reaction reduces solvent consumption and energy usage compared to multi-step sequences that require isolation and drying of intermediates. These cumulative efficiencies lead to a substantially lower cost of goods sold (COGS), providing a competitive pricing advantage in the global marketplace for pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the reaction conditions. The ability to use air as an oxidant in open vessels removes the dependency on pressurized gas lines or specialized oxygen delivery systems, simplifying the infrastructure requirements for production facilities. Moreover, the wide substrate scope means that if a specific aldehyde becomes unavailable, alternative substituted aldehydes can often be used to generate analogues without re-optimizing the entire process. This flexibility is crucial for maintaining continuity in the supply of diverse API intermediates, ensuring that downstream drug development programs are not delayed by raw material constraints.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges regarding heat transfer and safety, but this exothermic oxidative cyclization is well-suited for scale-up due to its moderate temperature requirements. The use of high-boiling solvents like o-dichlorobenzene allows for safe operation at atmospheric pressure, reducing the engineering controls needed for high-pressure reactors. From an environmental standpoint, the reduction in waste generation aligns with increasingly stringent global regulations on chemical manufacturing. The absence of heavy metal waste streams simplifies wastewater treatment and disposal, lowering environmental compliance costs and enhancing the sustainability profile of the manufacturing site, which is a key metric for modern corporate social responsibility goals.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating this technology for their specific applications, we have compiled answers to common questions regarding the practical implementation of this synthetic method. These insights are derived directly from the experimental data and scope defined in the patent documentation, addressing concerns about reaction specificity, safety, and adaptability. Understanding these nuances is essential for integrating this chemistry into existing production workflows effectively.
Q: Is a transition metal catalyst required for this pyrazolo[1,3,5]triazine synthesis?
A: No, the patented method (CN113801119B) specifically operates without transition metal catalysts or additives, utilizing ammonium iodide and an oxidant instead, which significantly simplifies purification.
Q: What is the substrate scope for the aldehyde component in this reaction?
A: The method demonstrates broad compatibility with various aromatic aldehydes, including those with electron-donating groups (methyl, methoxy), electron-withdrawing groups (halogens, nitro, cyano), and heterocyclic aldehydes like thiophene and pyridine.
Q: What are the optimal reaction conditions for maximum yield?
A: Optimal yields (up to 95%) are achieved using o-dichlorobenzene as the solvent at temperatures between 130-150°C, with di-tert-butyl peroxide or air serving as effective oxidants.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrazolo[1,3,5]triazine Supplier
The technological advancements described in CN113801119B highlight the immense potential of pyrazolo[1,3,5]triazine derivatives in next-generation therapeutics. At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating such innovative academic and patent research into reliable commercial reality. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from gram-scale optimization to full-scale manufacturing. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of pyrazolo[1,3,5]triazine intermediate delivered meets the highest international standards for pharmaceutical use.
We invite you to leverage our technical expertise to accelerate your drug development timeline. Our team is ready to conduct a Customized Cost-Saving Analysis tailored to your specific target molecule, identifying opportunities to further optimize the synthetic route for maximum economic efficiency. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a supply chain that is not only cost-effective but also resilient and compliant, positioning your organization for success in the competitive pharmaceutical landscape.
