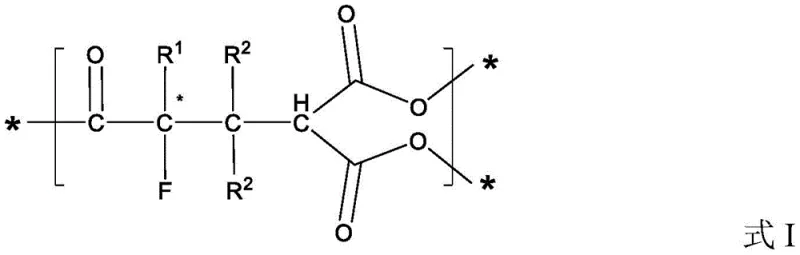
Advanced NHC-Catalyzed Kinetic Resolution for High-Purity Chiral Alpha-Fluoroketones
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to access complex chiral building blocks, particularly those containing fluorine atoms which are pivotal for enhancing metabolic stability and binding affinity in drug candidates. Patent CN112279765B introduces a groundbreaking preparation method for chiral alpha-fluoroketone compounds, addressing a significant gap in the synthesis of tetra-substituted chiral alpha-fluoro-chain ketones. This technology leverages a sophisticated kinetic resolution strategy mediated by chiral N-heterocyclic carbene (NHC) catalysts, transforming racemic alpha-fluoroketone compounds and alkenal compounds into valuable chiral intermediates. The significance of this innovation lies in its ability to overcome the limitations of traditional fluorination techniques, which often struggle with stereocontrol or require hazardous reagents. By utilizing a metal-free organocatalytic approach, this method not only ensures high enantioselectivity but also aligns with modern green chemistry principles, making it an attractive option for the production of high-purity pharmaceutical intermediates.
For R&D directors evaluating synthetic routes, understanding the mechanistic superiority of this novel approach over conventional methods is critical. Historically, the preparation of chiral alpha-fluoroketones has been fraught with challenges, particularly when targeting tetra-substituted structures. Conventional methods often rely on stoichiometric chiral auxiliaries or expensive transition metal catalysts that leave behind toxic residues, necessitating complex purification steps that erode overall yield. Furthermore, many existing protocols are limited to trisubstituted ketones or cyclic systems, failing to provide a general solution for acyclic tetra-substituted variants which are increasingly relevant in medicinal chemistry. The scarcity of direct and facile methods for these specific scaffolds has long hindered the rapid development of fluorinated drug candidates, forcing chemists to resort to multi-step sequences that are inefficient and costly.
In stark contrast, the novel approach detailed in the patent utilizes a chiral NHC catalyst to facilitate a dynamic kinetic resolution process that is both operationally simple and highly efficient. As illustrated in the reaction scheme below, the method employs readily available racemic alpha-fluoroketones and alkenals as starting materials, reacting them under mild conditions to yield the desired chiral core skeleton with excellent stereocontrol. The use of an oxidant, such as a diphenoquinone derivative, in conjunction with a base and specific additives like lithium chloride, creates a synergistic environment that promotes the selective transformation of one enantiomer while leaving the other intact or converting it into a separable byproduct. This strategy eliminates the need for cryogenic temperatures or inert atmospheres beyond standard Schlenk techniques, significantly lowering the barrier for implementation in both laboratory and pilot plant settings.
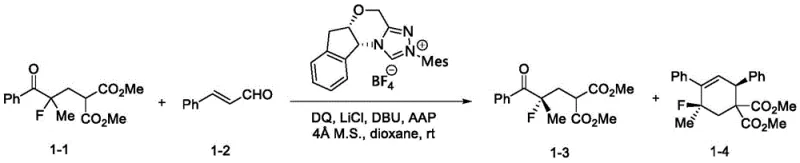
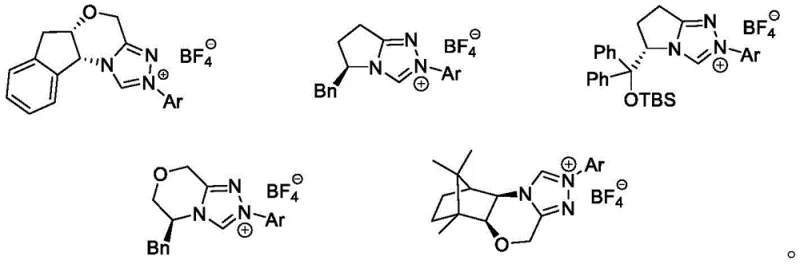
Mechanistically, the success of this transformation hinges on the unique reactivity of the N-heterocyclic carbene catalyst, which acts as a nucleophilic activator for the alkenal substrate. The catalyst forms a reactive Breslow intermediate or related acyl azolium species upon interaction with the aldehyde, which then engages with the alpha-fluoroketone in a stereoselective manner. The presence of the fluorine atom at the alpha-position introduces significant electronic effects that influence the acidity of adjacent protons and the stability of transition states, requiring a catalyst system capable of precise discrimination between enantiomers. The chiral environment provided by the NHC ligand, often featuring bulky substituents and rigid backbones as shown in the catalyst structures, ensures that the reaction proceeds through a favored transition state, leading to high enantiomeric excess (ee) values. Additionally, the inclusion of lithium chloride as an additive plays a crucial role in modulating the Lewis acidity of the system and stabilizing key intermediates, thereby enhancing both the reaction rate and the selectivity of the kinetic resolution.
Impurity control is another paramount concern for pharmaceutical manufacturing, and this method offers distinct advantages in managing the impurity profile. The kinetic resolution inherently separates the desired enantiomer from the undesired one, effectively acting as a purification step during the synthesis itself. Unlike methods that generate complex mixtures of regioisomers or over-fluorinated byproducts, the NHC-catalyzed pathway is highly specific, primarily yielding the target chiral ketone and a distinct cyclized byproduct that can be easily removed via standard silica gel chromatography. The mild reaction conditions, typically ranging from 0°C to 50°C, minimize thermal degradation and side reactions such as polymerization or elimination, which are common pitfalls in fluoro-organic synthesis. This results in a cleaner crude reaction mixture, reducing the burden on downstream purification processes and ensuring that the final product meets stringent purity specifications required for clinical applications.
How to Synthesize Chiral Alpha-Fluoroketone Efficiently
Implementing this synthesis route requires careful attention to reagent quality and reaction parameters to maximize yield and enantioselectivity. The process begins with the preparation of a reaction mixture containing the chiral NHC catalyst, an oxidant like 3,3',5,5'-tetra-tert-butyl-4,4'-diphenoquinone, and a lithium salt additive in an ether solvent such as dioxane. Molecular sieves are often added to sequester moisture, which can deactivate the sensitive carbene catalyst. The racemic alpha-fluoroketone is introduced as a solution, followed by the addition of the alkenal and a combination of organic bases such as DBU and AAP. The reaction is allowed to proceed at room temperature with stirring for a period of 4 to 12 hours, after which the solvent is removed under reduced pressure. The resulting residue is purified using column chromatography to isolate the chiral alpha-fluoroketone product. For detailed operational parameters and specific molar ratios, please refer to the standardized protocol below.
- Prepare the reaction mixture by combining the chiral NHC catalyst, oxidant (such as DQ), lithium chloride additive, and molecular sieves in a reaction vessel under an inert atmosphere.
- Add the solution of racemic alpha-fluoroketone in dioxane, followed by the alkenal compound, base (DBU/AAP), and ensure thorough mixing.
- Stir the reaction at room temperature for 4 to 12 hours, then concentrate under reduced pressure and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this NHC-catalyzed kinetic resolution method presents substantial opportunities for cost optimization and risk mitigation. Traditional routes to chiral fluorinated ketones often depend on precious metal catalysts or specialized fluorinating agents that are subject to volatile market prices and supply constraints. By shifting to an organocatalytic system based on nitrogen-containing heterocycles, manufacturers can decouple their production costs from the fluctuations of the precious metals market. The catalysts used in this process are synthesized from relatively inexpensive precursors and can often be recovered or used at low loadings, contributing to a significant reduction in the overall cost of goods sold. Furthermore, the elimination of heavy metals simplifies the regulatory compliance landscape, removing the need for expensive and time-consuming metal scavenging steps that are mandatory for API production.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the use of cheap and commercially available starting materials combined with a catalyst system that operates efficiently at low loadings. The avoidance of cryogenic conditions means that energy consumption for cooling is drastically minimized compared to traditional asymmetric syntheses that require temperatures below -78°C. Additionally, the simplified workup procedure, which involves basic concentration and chromatography rather than complex extractions or distillations, reduces labor costs and solvent usage. The high selectivity of the reaction minimizes waste generation, leading to better atom economy and lower disposal costs for hazardous chemical waste, thereby enhancing the overall profitability of the manufacturing process.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reliance on commodity chemicals such as dioxane, common organic bases, and simple quinone oxidants, all of which have robust global supply networks. Unlike specialized chiral ligands that may have single-source suppliers and long lead times, the NHC catalysts described can be synthesized in-house or sourced from multiple vendors, reducing the risk of supply disruption. The operational simplicity of the reaction, which tolerates a range of temperatures and does not require ultra-dry conditions beyond standard molecular sieve drying, makes it adaptable to various manufacturing facilities without the need for specialized equipment upgrades. This flexibility ensures consistent production schedules and reliable delivery timelines for downstream customers.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram or ton scale is facilitated by the homogeneous nature of the reaction and the absence of exothermic hazards associated with strong fluorinating agents. The mild conditions allow for the use of standard stainless steel reactors, avoiding the corrosion issues often encountered with aggressive fluorine chemistry. From an environmental standpoint, the metal-free nature of the catalyst aligns with increasing regulatory pressures to reduce heavy metal discharge in pharmaceutical effluents. The process generates fewer byproducts and utilizes solvents that can be readily recycled, supporting sustainability goals and helping companies meet stringent environmental, social, and governance (ESG) criteria without compromising on production efficiency or product quality.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology for their specific projects, we have compiled answers to common inquiries regarding the reaction scope, catalyst stability, and purification requirements. These insights are derived directly from the experimental data and embodiments provided in the patent documentation, ensuring accuracy and relevance for process development scientists. Understanding these nuances is essential for successful technology transfer and scale-up.
Q: What are the advantages of using NHC catalysis for alpha-fluoroketone synthesis?
A: NHC catalysis allows for mild reaction conditions, avoids the use of toxic transition metals, and enables efficient kinetic resolution of racemic mixtures to produce high-enantiomeric excess products.
Q: Can this method be scaled for industrial production?
A: Yes, the process utilizes readily available reagents, operates at room temperature, and involves simple workup procedures like filtration and chromatography, making it highly suitable for commercial scale-up.
Q: What is the role of the oxidant in this reaction system?
A: The oxidant, typically a diphenoquinone derivative, is crucial for regenerating the active catalytic species and driving the oxidative esterification or resolution process forward efficiently.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Alpha-Fluoroketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral intermediates play in the development of next-generation therapeutics. Our team of expert chemists has extensively evaluated the NHC-catalyzed kinetic resolution technology described in CN112279765B and possesses the technical expertise to implement this route effectively. We offer comprehensive CDMO services tailored to the unique challenges of fluorinated chemistry, ensuring that your project moves seamlessly from bench-scale optimization to commercial production. Our facilities are equipped with state-of-the-art analytical instruments and rigorous QC labs capable of verifying stringent purity specifications, including enantiomeric excess determination via chiral HPLC, to guarantee that every batch meets your exacting standards.
We invite you to leverage our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to accelerate your drug development timeline. By partnering with us, you gain access to a Customized Cost-Saving Analysis that identifies specific opportunities to optimize your supply chain and reduce manufacturing expenses without sacrificing quality. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments for your target chiral alpha-fluoroketone compounds, and let us demonstrate how our advanced synthetic capabilities can become a strategic asset for your organization.
