Advanced Iridium Catalysis for Scalable Synthesis of Chiral Tetrahydropyrrolo[1,2-a]pyrazine Intermediates
Introduction to Advanced Chiral Amine Synthesis
The synthesis of chiral heterocyclic compounds represents a cornerstone in modern medicinal chemistry, particularly for scaffolds exhibiting potent biological activities such as anti-aging, antiviral, and antiarrhythmic properties. Patent CN109956946B introduces a groundbreaking methodology for the asymmetric hydrogenation of dihydropyrrole and indole[1,2-a]pyrazine derivatives, utilizing a highly efficient iridium-based catalytic system. This technology addresses the critical industry demand for reliable pharmaceutical intermediate supplier capabilities by offering a route that combines exceptional enantioselectivity with operational simplicity. The core innovation lies in the use of an iridium metal precursor complexed with specialized chiral bisphosphine ligands, which facilitates the direct reduction of the C=N bond without the need for harsh activating agents. By leveraging this advanced catalytic approach, manufacturers can access high-purity tetrahydropyrrolo[1,2-a]pyrazine derivatives that serve as vital building blocks for next-generation therapeutic agents.
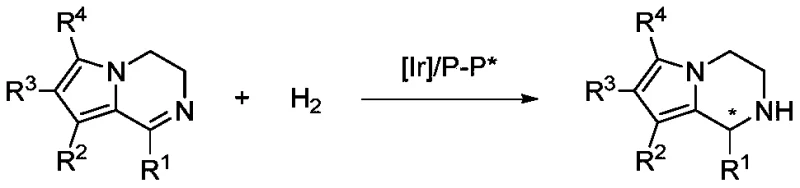
The significance of this patent extends beyond mere academic interest; it provides a practical solution for the cost reduction in chiral amine manufacturing by streamlining the synthetic pathway. Traditional methods often involve multi-step sequences or resolution processes that inherently limit yield and increase material costs. In contrast, this direct hydrogenation strategy maximizes atom economy and minimizes waste generation, aligning perfectly with green chemistry principles. The ability to tune the stereochemical outcome through ligand selection allows for the precise production of specific enantiomers required for biological efficacy. As we delve deeper into the technical specifics, it becomes evident that this methodology offers a robust platform for the commercial scale-up of complex heterocyclic intermediates, ensuring supply chain continuity for critical drug substances.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the tetrahydropyrrolo[1,2-a]pyrazine core has relied heavily on substrate-induced strategies or kinetic resolution techniques, both of which present significant drawbacks for industrial application. Substrate-induced approaches often necessitate the use of chiral starting materials that are expensive and difficult to source in bulk quantities, thereby inflating the overall cost of goods. Furthermore, kinetic resolution methods are intrinsically limited to a maximum theoretical yield of 50%, meaning that half of the valuable starting material is discarded as the unwanted enantiomer, creating substantial waste disposal challenges. Previous catalytic attempts, such as those reported by Zhou's group in 2014, required substrate activation strategies involving the formation of benzyl bromide salts and the addition of stoichiometric amounts of base to neutralize acid byproducts. This requirement for excess base not only complicates the workup procedure but also poses risks of product racemization if not meticulously controlled, leading to inconsistent quality and potential batch failures.
The Novel Approach
The methodology described in CN109956946B fundamentally shifts the paradigm by employing a direct asymmetric hydrogenation strategy that eliminates the need for substrate activation or stoichiometric additives. By utilizing a homogeneous iridium catalyst system paired with optimized chiral bisphosphine ligands, the reaction proceeds under mild conditions to deliver the target chiral amines with high efficiency. This novel approach bypasses the yield ceiling associated with kinetic resolution, allowing for near-quantitative conversion of the starting dihydropyrrole substrates into the desired tetrahydropyrrolo[1,2-a]pyrazine products. The elimination of base additives simplifies the downstream processing significantly, as there is no need for extensive washing steps to remove salt byproducts, thus reducing solvent consumption and processing time. Moreover, the reaction demonstrates remarkable tolerance to various functional groups, enabling the synthesis of a diverse library of derivatives without the need for protecting group manipulations, which further enhances the step economy of the overall process.
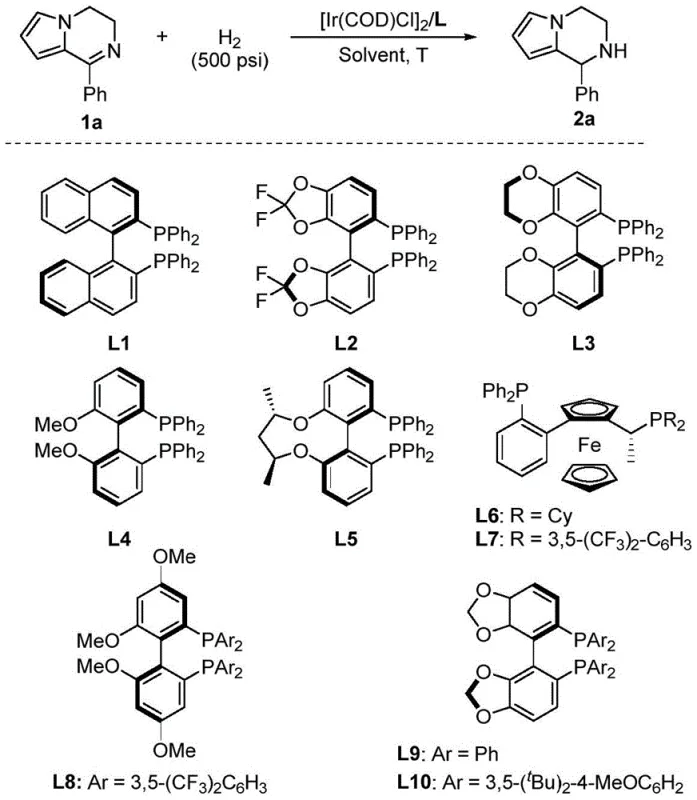
Mechanistic Insights into Iridium-Catalyzed Asymmetric Hydrogenation
The success of this transformation hinges on the precise interplay between the iridium metal center and the chiral bisphosphine ligand, which together create a highly defined stereogenic environment. The catalytic cycle initiates with the formation of an active cationic iridium species upon the interaction of the [Ir(COD)Cl]2 precursor with the chiral ligand in the presence of hydrogen. This active complex then coordinates with the nitrogen-containing heterocycle of the dihydropyrrole substrate, positioning the C=N double bond for hydride transfer. The bulky substituents on the phosphine ligands, such as those found in the Walphos family, exert significant steric pressure that forces the substrate to adopt a specific orientation relative to the metal center. This steric differentiation ensures that hydrogen delivery occurs exclusively from one face of the planar imine moiety, thereby dictating the absolute configuration of the newly formed chiral center with high fidelity.
Impurity control is inherently built into this mechanism due to the high chemoselectivity of the iridium catalyst towards the C=N bond over other potentially reducible functionalities. Unlike non-selective reducing agents like lithium aluminum hydride, which might reduce aromatic rings or other sensitive groups, this catalytic system targets the imine bond specifically. The mild reaction temperatures, typically ranging from 30°C to 60°C, further suppress side reactions such as polymerization or decomposition that are often observed under more vigorous conditions. Additionally, the stability of the iridium-ligand complex prevents the leaching of free metal ions that could catalyze unwanted background reactions, ensuring a clean reaction profile. This mechanistic robustness translates directly to a cleaner crude reaction mixture, facilitating easier purification and resulting in a final product with superior purity specifications that meet stringent regulatory requirements for pharmaceutical intermediates.
How to Synthesize Tetrahydropyrrolo[1,2-a]pyrazine Efficiently
The practical implementation of this synthesis route is designed for ease of operation, making it accessible for both laboratory-scale optimization and pilot-plant production. The process begins with the in situ generation of the catalyst, where the iridium precursor and ligand are stirred in a solvent like toluene under an inert atmosphere to ensure full complexation before substrate addition. Following this activation period, the substrate is introduced, and the vessel is pressurized with hydrogen gas to drive the reduction to completion. The reaction progress can be monitored via standard analytical techniques, and upon reaching full conversion, the workup involves a straightforward solvent removal and chromatographic purification.
- Prepare the catalyst by mixing an iridium metal precursor, such as [Ir(COD)Cl]2, with a chiral bisphosphine ligand like Walphos in an organic solvent under inert atmosphere.
- Transfer the catalyst solution to a reactor containing the dihydropyrrole or indole[1,2-a]pyrazine substrate dissolved in toluene or similar solvent.
- Pressurize the reactor with hydrogen gas (500-800 psi) and maintain the reaction temperature between 30-60°C for 24 to 48 hours to achieve high conversion and enantioselectivity.
- Upon completion, release hydrogen pressure, remove the solvent under reduced pressure, and purify the resulting chiral amine product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this iridium-catalyzed technology offers transformative benefits regarding cost structure and operational reliability. The primary driver for cost reduction in manufacturing stems from the high atom economy of the hydrogenation reaction, which utilizes molecular hydrogen as the reductant, a reagent that is both inexpensive and generates no stoichiometric waste. By avoiding the use of expensive chiral auxiliaries or resolving agents that are consumed in the process, the raw material costs are significantly lowered, improving the overall margin profile of the final API intermediate. Furthermore, the high yields reported, often exceeding 90%, mean that less starting material is required to produce a given amount of product, effectively stretching the purchasing power of the raw material budget and reducing the frequency of procurement cycles.
- Cost Reduction in Manufacturing: The elimination of stoichiometric bases and activating agents drastically simplifies the bill of materials, removing the costs associated with purchasing, handling, and disposing of these auxiliary chemicals. The mild reaction conditions also translate to lower energy consumption, as the process does not require extreme heating or cooling, thereby reducing utility costs over the lifecycle of the production campaign. Additionally, the high selectivity minimizes the formation of byproducts, which reduces the load on waste treatment facilities and lowers the environmental compliance costs associated with hazardous waste disposal. This streamlined chemical profile allows for a more predictable and stable cost of goods sold, shielding the organization from volatility in raw material pricing.
- Enhanced Supply Chain Reliability: The robustness of the catalyst system across a broad substrate scope ensures that supply chains are not disrupted by the need for highly specialized or custom-synthesized starting materials. Since the reaction tolerates various substituents including halogens and alkyl groups, manufacturers can source common, commodity-grade building blocks rather than relying on fragile supply lines for exotic reagents. The use of commercially available iridium precursors and ligands further de-risks the supply chain, as these components are produced by multiple global vendors, preventing single-source bottlenecks. This flexibility allows for rapid scaling of production volumes to meet market demand without the long lead times typically associated with developing new synthetic routes for each new derivative.
- Scalability and Environmental Compliance: The transition from bench scale to commercial production is facilitated by the homogeneous nature of the catalysis, which behaves predictably in larger reactors provided adequate mixing and gas-liquid mass transfer are maintained. The absence of hazardous reagents and the use of relatively benign solvents like toluene simplify the safety case for plant operations, accelerating regulatory approvals for new manufacturing sites. From an environmental perspective, the high efficiency and low waste generation align with corporate sustainability goals, reducing the carbon footprint of the manufacturing process. This compliance with green chemistry principles not only mitigates regulatory risk but also enhances the brand reputation of the end-product in markets that increasingly value environmentally responsible sourcing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric hydrogenation technology. These insights are derived directly from the experimental data and beneficial effects outlined in the patent documentation, providing a clear understanding of the process capabilities.
Q: What are the key advantages of this iridium-catalyzed method over traditional synthesis routes?
A: Unlike traditional methods that require substrate activation with stoichiometric bases or suffer from low yields due to kinetic resolution limits, this method utilizes direct asymmetric hydrogenation. It operates under mild conditions without the need for excess base, achieving high enantiomeric excess (up to 95%) and excellent yields, thereby simplifying the purification process and reducing waste.
Q: Which ligands provide the best enantioselectivity for this transformation?
A: The patent data indicates that chiral bisphosphine ligands, particularly Walphos variants (such as CAS 388079-60-5), demonstrate superior performance. These ligands create a specific chiral environment around the iridium center that effectively differentiates the prochiral faces of the substrate, leading to high optical purity in the final tetrahydropyrrolo[1,2-a]pyrazine products.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process is highly scalable. It uses commercially available catalyst precursors and operates at moderate temperatures and pressures. The robustness of the catalyst system across a wide range of substrates, including those with halogen and alkyl substituents, ensures consistent quality, making it ideal for the commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrahydropyrrolo[1,2-a]pyrazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing high-quality chiral intermediates to accelerate drug development timelines. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize state-of-the-art analytical instrumentation to verify enantiomeric excess and chemical purity. By partnering with us, you gain access to a supply chain that is resilient, compliant, and optimized for cost-effectiveness, allowing you to focus on your core competencies in drug discovery and clinical development.
We invite you to engage with our technical procurement team to discuss how this advanced iridium-catalyzed synthesis can be integrated into your specific project requirements. Request a Customized Cost-Saving Analysis today to understand the potential economic benefits of switching to this superior manufacturing route. Our experts are ready to provide specific COA data and route feasibility assessments tailored to your target molecules, ensuring that your supply chain is built on a foundation of scientific excellence and commercial viability.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
