Advanced Oxidative Synthesis of N-Monosubstituted-Alpha-Carbonylamides for Pharmaceutical Intermediates
Introduction to Patent CN102153433B Technology
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing complex molecular scaffolds, particularly those containing the alpha-carbonyl amide motif which is prevalent in numerous bioactive natural products and synthetic drugs. Patent CN102153433B introduces a highly efficient and environmentally benign preparation method for N-monosubstituted-alpha-carbonylamide compounds, addressing critical bottlenecks in existing synthetic routes. This technology leverages a novel aerobic oxidation strategy that transforms readily available phenylacetamide derivatives into valuable alpha-keto amides using molecular oxygen from air as the terminal oxidant. The significance of this innovation lies in its ability to bypass the use of hazardous heavy metal oxidants and expensive transition metal catalysts, thereby offering a sustainable pathway for producing key intermediates used in the synthesis of antifungal agents, protease inhibitors, and other therapeutic candidates. By establishing a reliable supply chain for these high-purity pharmaceutical intermediates, manufacturers can significantly streamline their production workflows while adhering to increasingly stringent environmental regulations.
The core breakthrough described in this patent involves a phase-transfer catalyzed oxidation system that operates under mild reflux conditions in dioxane. Unlike traditional methods that often require stoichiometric amounts of toxic reagents or harsh reaction environments, this approach utilizes a combination of inorganic bases such as sodium bicarbonate or cesium carbonate alongside tetrabutylammonium bisulfate to facilitate the activation of molecular oxygen. The result is a versatile synthetic platform capable of accommodating a wide range of substituents on both the aromatic ring and the nitrogen atom, ensuring broad applicability across diverse drug discovery programs. For R&D directors and procurement managers alike, this represents a strategic opportunity to reduce dependency on volatile supply chains for specialized oxidizing agents while achieving superior cost efficiency through the use of commodity chemicals. The reported yields, reaching as high as 93% for specific substrates, underscore the practical viability of this method for commercial scale-up.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-monosubstituted-alpha-carbonylamides has been plagued by significant operational and safety challenges associated with conventional oxidation protocols. One of the most common traditional approaches involves the use of selenium dioxide (SeO2) to directly oxidize the alpha-methylene group of N-substituted amides, a process that necessitates the handling of highly toxic selenium compounds and generates substantial amounts of hazardous waste that require complex disposal procedures. 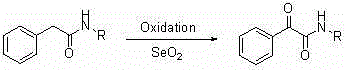 Another widely used method relies on photooxygenation techniques employing sensitizers like Rose Bengal or Tetraphenylporphyrin, which demand specialized photoreactors and precise control of light sources, thereby increasing capital expenditure and limiting scalability. Furthermore, alternative routes such as the Ugi reaction or transition metal-catalyzed carbonylation often suffer from the need for expensive ligands, toxic solvents like benzene, or dangerous carbon monoxide gas, all of which contribute to elevated production costs and heightened safety risks in a manufacturing setting. These limitations collectively hinder the ability of chemical suppliers to provide consistent, high-volume supplies of these critical intermediates at competitive prices.
Another widely used method relies on photooxygenation techniques employing sensitizers like Rose Bengal or Tetraphenylporphyrin, which demand specialized photoreactors and precise control of light sources, thereby increasing capital expenditure and limiting scalability. Furthermore, alternative routes such as the Ugi reaction or transition metal-catalyzed carbonylation often suffer from the need for expensive ligands, toxic solvents like benzene, or dangerous carbon monoxide gas, all of which contribute to elevated production costs and heightened safety risks in a manufacturing setting. These limitations collectively hinder the ability of chemical suppliers to provide consistent, high-volume supplies of these critical intermediates at competitive prices.
The Novel Approach
In stark contrast to these legacy technologies, the method disclosed in CN102153433B offers a paradigm shift by utilizing atmospheric oxygen as a clean and abundant oxidant within a simple phase-transfer catalytic system. This novel approach eliminates the requirement for stoichiometric toxic oxidants and precious metal catalysts, replacing them with inexpensive inorganic bases and quaternary ammonium salts that are easily sourced and handled. The reaction proceeds smoothly under reflux in dioxane with simple exposure to air, removing the need for complex pressurized equipment or specialized irradiation setups. This simplification of the reaction infrastructure translates directly into substantial cost reduction in pharmaceutical intermediate manufacturing, as it lowers both the raw material input costs and the overhead associated with waste management and safety compliance. Moreover, the mild nature of the reaction conditions preserves sensitive functional groups on the substrate, leading to cleaner reaction profiles and higher purity products that require less intensive downstream purification efforts.
Mechanistic Insights into Aerobic Oxidative Amidation
The mechanistic foundation of this synthesis relies on the synergistic interaction between the phase transfer catalyst and the base to activate the alpha-position of the phenylacetamide towards oxidation by molecular oxygen. 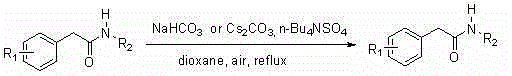 The tetrabutylammonium bisulfate facilitates the transport of the basic anions into the organic phase, where they deprotonate the acidic alpha-hydrogen of the amide substrate to generate a reactive enolate or carbanion species. This activated intermediate then reacts with dissolved oxygen to form a hydroperoxide species, which subsequently undergoes decomposition and rearrangement to yield the desired alpha-keto carbonyl functionality. The elegance of this mechanism lies in its self-sustaining nature, where the catalyst is regenerated and the only byproduct is water, aligning perfectly with green chemistry principles. For technical teams, understanding this pathway is crucial for optimizing reaction parameters such as stirring speed and aeration rates to ensure maximum oxygen mass transfer, which is often the rate-limiting step in gas-liquid heterogeneous reactions.
The tetrabutylammonium bisulfate facilitates the transport of the basic anions into the organic phase, where they deprotonate the acidic alpha-hydrogen of the amide substrate to generate a reactive enolate or carbanion species. This activated intermediate then reacts with dissolved oxygen to form a hydroperoxide species, which subsequently undergoes decomposition and rearrangement to yield the desired alpha-keto carbonyl functionality. The elegance of this mechanism lies in its self-sustaining nature, where the catalyst is regenerated and the only byproduct is water, aligning perfectly with green chemistry principles. For technical teams, understanding this pathway is crucial for optimizing reaction parameters such as stirring speed and aeration rates to ensure maximum oxygen mass transfer, which is often the rate-limiting step in gas-liquid heterogeneous reactions.
Beyond the primary transformation, the utility of the resulting N-monosubstituted-alpha-carbonylamides is demonstrated by their versatility as synthetic building blocks for further derivatization.  For instance, these alpha-keto amides can serve as precursors for the synthesis of alpha-hydroxy amides through stereoselective reduction, a key step in the production of potent antifungal agents and protease inhibitors. The presence of the adjacent carbonyl group enhances the electrophilicity of the molecule, allowing for facile nucleophilic attacks and cyclization reactions that construct complex heterocyclic systems found in drugs targeting Cathepsin K or HIV-1. From an impurity control perspective, the mild oxidative conditions minimize the formation of over-oxidized byproducts or degradation fragments that are common with harsher oxidants like chromic acid or permanganate. This results in a cleaner crude product profile, simplifying the isolation process and ensuring that the final active pharmaceutical ingredient meets stringent regulatory specifications for residual impurities and heavy metals.
For instance, these alpha-keto amides can serve as precursors for the synthesis of alpha-hydroxy amides through stereoselective reduction, a key step in the production of potent antifungal agents and protease inhibitors. The presence of the adjacent carbonyl group enhances the electrophilicity of the molecule, allowing for facile nucleophilic attacks and cyclization reactions that construct complex heterocyclic systems found in drugs targeting Cathepsin K or HIV-1. From an impurity control perspective, the mild oxidative conditions minimize the formation of over-oxidized byproducts or degradation fragments that are common with harsher oxidants like chromic acid or permanganate. This results in a cleaner crude product profile, simplifying the isolation process and ensuring that the final active pharmaceutical ingredient meets stringent regulatory specifications for residual impurities and heavy metals.
How to Synthesize N-Monosubstituted-Alpha-Carbonylamides Efficiently
The implementation of this oxidative protocol in a laboratory or pilot plant setting follows a straightforward operational sequence designed to maximize safety and yield. The process begins with the precise charging of the phenylacetamide starting material, the selected inorganic base, and the phase transfer catalyst into a reaction vessel containing dioxane solvent. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and optimal performance across different scales of operation.
- Charge phenylacetamide derivative, sodium bicarbonate or cesium carbonate, and tetrabutylammonium bisulfate into dioxane solvent.
- Stir the mixture under reflux conditions with exposure to air until the starting material is fully consumed.
- Filter the reaction mixture, remove solvent under reduced pressure, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers compelling economic and logistical benefits that extend far beyond simple yield improvements. The elimination of expensive and toxic reagents like selenium dioxide or transition metal complexes drastically simplifies the sourcing strategy, allowing companies to rely on commodity chemicals that are available from multiple global suppliers. This diversification of the supply base mitigates the risk of production delays caused by shortages of specialized reagents, ensuring a more resilient and continuous manufacturing operation. Furthermore, the use of air as the oxidant removes the cost and safety burden associated with purchasing, storing, and disposing of hazardous oxidizing agents, leading to significant operational expenditure savings over the lifecycle of the product.
- Cost Reduction in Manufacturing: The replacement of stoichiometric toxic oxidants with molecular oxygen fundamentally alters the cost structure of the synthesis by removing high-cost line items from the bill of materials. Since air is freely available and the catalyst loading is minimal, the variable cost per kilogram of product is substantially lowered compared to traditional methods. Additionally, the simplified workup procedure involving simple filtration and solvent removal reduces labor hours and energy consumption associated with complex extraction and washing steps. These cumulative efficiencies translate into a more competitive pricing model for the final intermediate, enabling better margin protection for downstream drug manufacturers.
- Enhanced Supply Chain Reliability: By utilizing robust and widely available starting materials such as substituted phenylacetamides and common inorganic bases, this method reduces dependency on single-source suppliers for exotic reagents. The stability of the reaction conditions also means that the process is less susceptible to variations in raw material quality, ensuring consistent output even when sourcing from different vendors. This reliability is critical for maintaining just-in-time inventory levels and meeting tight delivery schedules for clinical trial materials or commercial drug launches without the risk of batch failures due to reagent incompatibility.
- Scalability and Environmental Compliance: The absence of heavy metals and toxic byproducts simplifies the environmental permitting process and reduces the cost of wastewater treatment and solid waste disposal. Scaling this reaction from grams to tons does not require significant changes in equipment design, as standard glass-lined or stainless steel reactors equipped with reflux condensers are sufficient. This ease of scale-up accelerates the time-to-market for new drug candidates by shortening the process development phase and facilitating a smoother transition from pilot plant to full commercial production facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxidative synthesis technology. These answers are derived directly from the experimental data and scope defined in the patent documentation to provide accurate guidance for potential partners.
Q: What are the advantages of this aerobic oxidation method over traditional SeO2 oxidation?
A: This method eliminates the need for toxic and stoichiometric selenium dioxide (SeO2), utilizing molecular oxygen from air instead. This significantly reduces hazardous waste generation and raw material costs while maintaining high yields up to 93%.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process uses conventional solvents like dioxane and simple reflux conditions without requiring specialized high-pressure equipment or cryogenic temperatures, making it highly scalable for industrial manufacturing.
Q: What types of substrates are compatible with this synthesis route?
A: The method demonstrates broad substrate scope, tolerating various substituents on the aromatic ring including electron-donating groups like methoxy and methyl, as well as electron-withdrawing groups like chloro and nitro.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Monosubstituted-Alpha-Carbonylamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of modern drug development programs. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of N-monosubstituted-alpha-carbonylamide delivered meets the highest industry standards for potency and impurity profiles. We are committed to leveraging advanced synthetic technologies like the one described in CN102153433B to provide our clients with sustainable and cost-effective solutions for their complex chemical requirements.
We invite you to engage with our technical procurement team to discuss how this innovative oxidative method can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this greener synthesis route for your supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that will demonstrate our capability to support your long-term manufacturing goals with reliability and excellence.
