Advanced Synthesis of High-Purity Nintedanib Ethanesulfonate for Commercial API Production
The pharmaceutical industry continuously demands robust, scalable, and high-purity synthetic routes for critical kinase inhibitors like Nintedanib Ethanesulfonate. Patent CN111848490A introduces a significant technological advancement in this domain, addressing long-standing challenges related to impurity control and process efficiency. This proprietary methodology outlines a refined four-step synthesis that leverages a DMAP-catalyzed one-pot reaction for the initial intermediate, followed by a strategic coupling sequence and a novel stage crystallization protocol. For R&D directors and procurement specialists, this patent represents a viable pathway to secure a reliable pharmaceutical intermediates supplier capable of delivering material with purity exceeding 99.5%. The core innovation lies not just in the chemical transformations but in the meticulous control of crystallization dynamics, which effectively suppresses the formation of the difficult-to-remove impurity IMP01, thereby ensuring a cleaner profile for downstream API manufacturing.
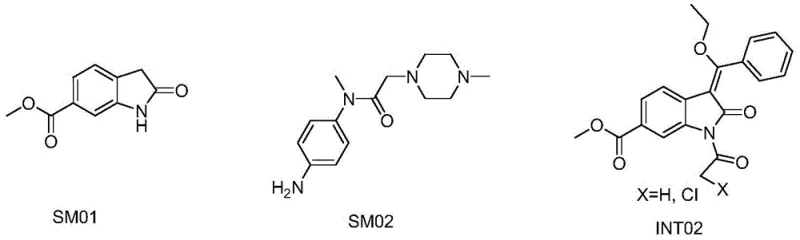
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Nintedanib Ethanesulfonate has been plagued by issues regarding impurity profiles and operational complexity. Many existing patents describe one-pot methods that attempt to streamline the process by skipping intermediate isolation; however, this often results in the accumulation of side products that are difficult to purge in later stages. A critical flaw in several prior art routes involves the deprotection of the amino group on the indole scaffold before coupling with the piperazine-containing fragment (SM02). This premature exposure of the imino functionality creates a competitive nucleophilic environment, leading to unwanted side reactions and a complex mixture of by-products. Furthermore, traditional salification processes often rely on isopropanol as a crystallization solvent. While effective for precipitation, isopropanol can react with ethanesulfonic acid to form isopropyl ethanesulfonate, a genotoxic impurity that poses severe regulatory risks and requires costly additional purification steps to mitigate.
The Novel Approach
The methodology disclosed in CN111848490A fundamentally restructures the synthetic sequence to overcome these deficits. Instead of immediate deprotection, the process stabilizes the intermediate INT02 through a DMAP-catalyzed condensation in toluene, achieving high purity (>99.0%) before proceeding. Crucially, the coupling with SM02 is performed on the protected intermediate INT02 to form INT03, avoiding the competitive side reactions associated with free imino groups. The most distinct advantage, however, is the final purification strategy. By employing a stage crystallization technique using a specific blend of methyl tert-butyl ether (MTBE) and isopropyl ether, the process exploits polarity differences to selectively exclude impurity IMP01. This approach eliminates the risk of genotoxic ester formation associated with isopropanol and ensures that any single impurity remains below 0.1%, setting a new benchmark for quality in high-purity kinase inhibitor intermediates.
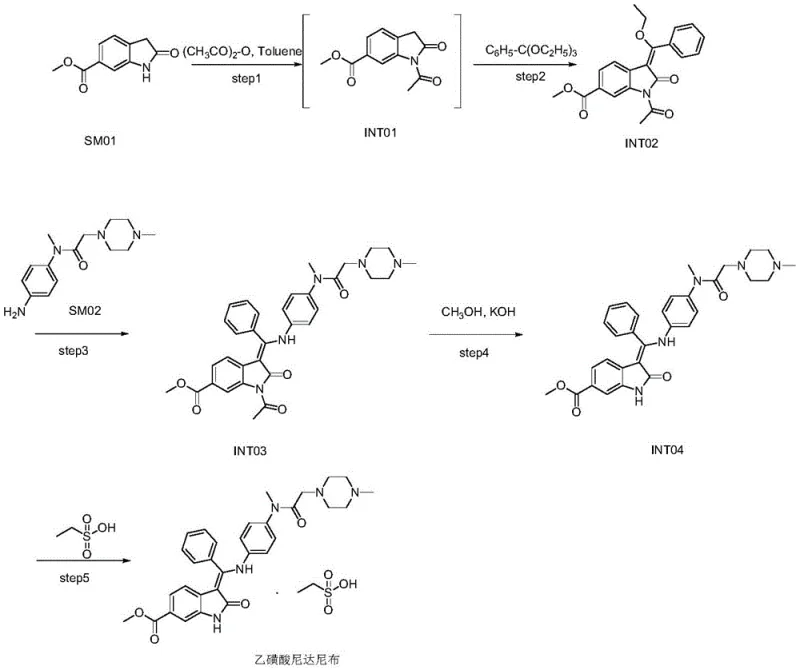
Mechanistic Insights into DMAP-Catalyzed Condensation and Stage Crystallization
The chemical efficacy of this route is anchored in the strategic use of 4-dimethylaminopyridine (DMAP) during the formation of intermediate INT02. In the reaction of SM01 with triethyl orthobenzoate and acetic anhydride, DMAP acts as a potent nucleophilic catalyst. It facilitates the acylation of the indole nitrogen and promotes the condensation reaction at the 3-position of the indole ring. The mechanism involves the formation of a highly reactive acyl-pyridinium intermediate, which significantly lowers the activation energy for the substitution reaction. This catalytic cycle allows the reaction to proceed efficiently at reflux temperatures of 104-110°C in toluene. Moreover, the process incorporates a dynamic solvent removal step where 2 to 5 times the weight of the starting material in solvent is evaporated during the reaction. This shift in equilibrium drives the condensation forward, minimizing reverse reactions and ensuring high conversion rates, which is critical for maintaining the purity of the intermediate before it enters the coupling phase.
Beyond the reaction kinetics, the control of solid-state properties through stage crystallization is a masterclass in impurity management. The final step involves reacting intermediate INT04 with ethanesulfonic acid in methanol. The novelty arises in the antisolvent addition sequence. Initially, methyl tert-butyl ether is added at 40-45°C to induce primary nucleation. Subsequently, isopropyl ether is introduced as the temperature is lowered to 0-10°C. Isopropyl ether possesses a higher polarity compared to MTBE, which alters the solvation shell around the dissolved species. This specific solvent gradient is thermodynamically tuned to keep the polar impurity IMP01 (a methylated piperazine derivative) in the mother liquor while forcing the less polar Nintedanib Ethanesulfonate salt to precipitate. This physical separation mechanism is far more effective than simple washing, providing a robust barrier against impurity carryover and ensuring the final product meets stringent pharmacopeial standards without requiring recrystallization.
How to Synthesize Nintedanib Ethanesulfonate Efficiently
The synthesis of this complex kinase inhibitor intermediate requires precise adherence to thermal and stoichiometric parameters to replicate the high yields and purity reported in the patent. The process is divided into four distinct operational units: the catalytic formation of the indole scaffold, the coupling with the piperazine side chain, the hydrolytic deprotection, and the final salification with controlled crystallization. Each step builds upon the purity of the previous one, emphasizing the importance of intermediate isolation. For process chemists aiming to implement this route, attention must be paid to the solvent ratios and the specific timing of antisolvent addition, as these are the critical process parameters (CPPs) that dictate the final crystal habit and purity profile. The detailed standardized synthesis steps are provided in the guide below to facilitate technology transfer and scale-up.
- Prepare intermediate INT02 via one-pot reaction of SM01, triethyl orthobenzoate, and acetic anhydride in toluene with DMAP catalyst at reflux (104-110°C), followed by solvent evaporation and crystallization.
- React INT02 with SM02 in DMF at 60-65°C to form intermediate INT03, ensuring controlled temperature to prevent premature deacetylation.
- Perform alkaline hydrolysis of INT03 using KOH in methanol at 50-55°C to yield intermediate INT04, followed by cooling and filtration.
- Salt formation with ethanesulfonic acid in methanol, followed by stage crystallization using methyl tert-butyl ether and isopropyl ether to remove impurity IMP01 and isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers tangible strategic benefits beyond mere chemical elegance. The primary value proposition lies in the substantial cost savings achieved through process intensification and waste reduction. By utilizing a DMAP-catalyzed system that operates with high efficiency, the overall reaction time is significantly reduced compared to conventional non-catalyzed methods. This reduction in cycle time translates directly to increased throughput in multipurpose reactors, allowing for better asset utilization and lower fixed cost allocation per kilogram of product. Furthermore, the ability to recover and reuse toluene, a common and relatively inexpensive solvent, contributes to a more sustainable and economically viable manufacturing model, driving cost reduction in API manufacturing without compromising on quality standards.
- Cost Reduction in Manufacturing: The elimination of complex purification sequences typically required to remove genotoxic impurities represents a major financial advantage. Conventional routes often necessitate extensive chromatography or multiple recrystallizations to meet safety specifications, which are both time-consuming and expensive in terms of solvent and labor consumption. By preventing the formation of isopropyl ethanesulfonate through the use of MTBE and isopropyl ether, this process removes the need for these costly remediation steps. Additionally, the high yield of the intermediate steps minimizes the loss of valuable starting materials like SM01 and SM02, ensuring that the raw material cost per unit of final product is optimized for commercial competitiveness.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply chain continuity by reducing the risk of batch failures. The strict control over impurity IMP01 ensures that batches consistently meet release specifications, minimizing the likelihood of quarantine or rejection. This reliability is crucial for maintaining the production schedules of downstream API manufacturers who depend on a steady flow of high-quality intermediates. Moreover, the starting materials employed in this route are commercially available and do not rely on exotic or restricted reagents, mitigating the risk of supply disruptions caused by raw material shortages and ensuring a stable supply for long-term contracts.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, the process is designed for seamless transition from pilot to commercial scale. The use of standard solvents like toluene, DMF, and methanol simplifies solvent recovery and waste treatment protocols, aligning with modern green chemistry principles. The stage crystallization technique is inherently scalable, as it relies on thermodynamic principles rather than delicate kinetic controls that are hard to reproduce in large vessels. This ease of commercial scale-up of complex pharmaceutical intermediates ensures that production volumes can be ramped up quickly to meet market demand without the need for specialized equipment or extensive process re-validation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the comparative data and experimental examples provided in the patent documentation, offering clarity on why this specific route outperforms prior art. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this method for their own production lines or for procurement teams assessing the quality claims of potential suppliers.
Q: How does this process control the genotoxic impurity IMP01?
A: The process utilizes a specific stage crystallization technique in the final step. By first adding methyl tert-butyl ether and subsequently adding isopropyl ether at lower temperatures (0-10°C), the polarity of the solvent system is optimized to selectively precipitate the product while keeping the polar impurity IMP01 (methylated piperazine derivative) in solution, reducing its content to below 0.1%.
Q: Why is DMAP used in the preparation of intermediate INT02?
A: DMAP (4-dimethylaminopyridine) acts as a highly efficient nucleophilic catalyst in the acylation and condensation steps. Its inclusion significantly accelerates the reaction rate, shortening the reaction time from the conventional 10-15 hours down to 6-10 hours, while simultaneously improving the purity of intermediate INT02 to greater than 99.0%.
Q: What are the advantages of isolating intermediate INT03 before deprotection?
A: Isolating INT03 prevents the exposure of the imino group prior to coupling with SM02. In conventional routes where deprotection occurs first, the exposed imino group competes with the amino group of SM02, leading to increased by-product formation. This step-wise approach ensures higher selectivity and overall product purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nintedanib Ethanesulfonate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of purity and consistency in the production of oncology and fibrosis therapeutics. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our facilities are equipped with rigorous QC labs and advanced analytical capabilities to verify stringent purity specifications, including the detection of trace impurities like IMP01 at levels below 0.1%. We are committed to delivering high-purity kinase inhibitor intermediates that empower your drug development programs and support your commercial manufacturing goals with unwavering quality assurance.
We invite you to collaborate with us to leverage this advanced synthetic technology for your supply chain. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. Please contact us to request specific COA data from our recent batches and to discuss route feasibility assessments for your projects. By partnering with NINGBO INNO PHARMCHEM, you secure not just a supplier, but a strategic ally dedicated to optimizing your reducing lead time for high-purity API intermediates and ensuring the success of your pharmaceutical products in the global market.
