Advanced Palladium-Catalyzed Synthesis of Polysubstituted Oxazoles for Commercial Scale-Up
Advanced Palladium-Catalyzed Synthesis of Polysubstituted Oxazoles for Commercial Scale-Up
The landscape of heterocyclic chemistry is constantly evolving, driven by the relentless demand for efficient routes to bioactive scaffolds found in modern therapeutics. A significant breakthrough in this domain is detailed in patent CN110156710B, which discloses a highly versatile preparation method for polysubstituted oxazole compounds. This technology addresses long-standing challenges in organic synthesis by leveraging a palladium-catalyzed coupling strategy that merges benzamide derivatives with propargyl carbonates. For R&D directors and process chemists seeking reliable pathways to complex nitrogen-containing heterocycles, this methodology represents a paradigm shift from traditional, often cumbersome, synthetic routes. The core innovation lies in its ability to construct the oxazole ring system under relatively mild thermal conditions while maintaining exceptional substrate tolerance, thereby opening new avenues for the rapid generation of diverse chemical libraries essential for drug discovery programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the oxazole core has relied heavily on the oxidation of oxazolines or various intramolecular and intermolecular cyclization strategies. While effective in specific contexts, these legacy methods frequently suffer from significant drawbacks that hinder their utility in large-scale manufacturing. A primary concern is the availability and stability of starting materials; many traditional precursors are not commercially abundant and require multi-step synthesis themselves, inflating both cost and lead time. Furthermore, conventional oxidative protocols often necessitate harsh reaction conditions, including strong oxidants and extreme temperatures, which can compromise sensitive functional groups and lead to complex impurity profiles. These factors collectively create bottlenecks in the supply chain, making it difficult for procurement teams to secure consistent quality and quantity of critical intermediates without incurring substantial overhead costs associated with purification and waste management.
The Novel Approach
In stark contrast, the methodology outlined in CN110156710B introduces a streamlined catalytic cycle that bypasses these historical inefficiencies. By utilizing readily accessible benzamide compounds and propargyl carbonates as building blocks, the process drastically simplifies the raw material sourcing landscape. The reaction proceeds smoothly in polar organic solvents such as N-methylpyrrolidone (NMP) or N,N-dimethylformamide (DMF) at temperatures ranging from 100°C to 140°C. This moderate thermal window is crucial for energy efficiency and equipment longevity in a plant setting. Moreover, the use of inexpensive inorganic bases like potassium carbonate or cesium carbonate eliminates the need for costly organometallic reagents. The result is a robust, operationally simple protocol that delivers high-purity polysubstituted oxazoles with minimal downstream processing, directly addressing the pain points of both cost reduction in pharmaceutical intermediate manufacturing and the need for scalable, green chemistry solutions.

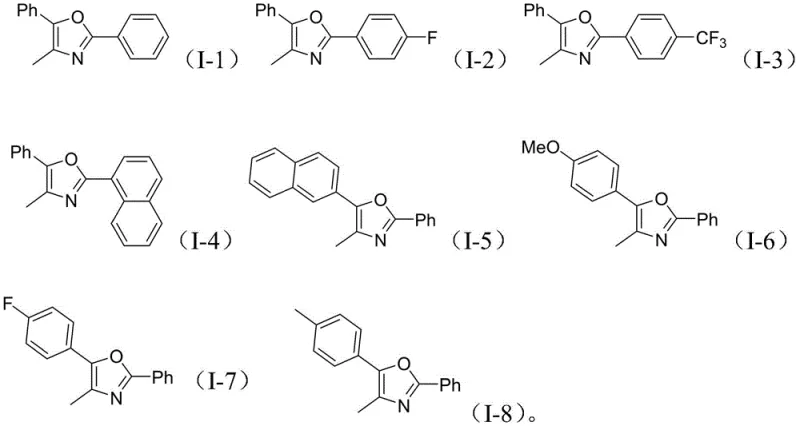
The versatility of this synthetic platform is further evidenced by its broad substrate scope, accommodating a wide array of electronic and steric environments. As illustrated in the structural diversity of the products, the system tolerates various substituents on the aromatic rings, including electron-withdrawing groups like fluorine and trifluoromethyl, as well as electron-donating moieties such as methoxy and methyl. This flexibility is paramount for medicinal chemists who require precise tuning of physicochemical properties during lead optimization. The ability to seamlessly integrate naphthyl groups and halogenated phenyl rings without compromising yield demonstrates the resilience of the catalytic system. Such adaptability ensures that this method is not merely a laboratory curiosity but a viable industrial tool capable of supporting the synthesis of complex API intermediates with high fidelity and reproducibility across different batches.
Mechanistic Insights into Pd-Catalyzed Cyclization
At the heart of this transformation lies a sophisticated palladium-catalyzed mechanism that orchestrates the formation of the C-O and C-N bonds requisite for the oxazole ring. The cycle initiates with the activation of the zero-valent palladium species, likely generated in situ from precursors such as tris(dibenzylideneacetone)dipalladium. This active catalyst coordinates with the alkyne functionality of the propargyl carbonate, facilitating an oxidative addition or insertion event that sets the stage for ring closure. The choice of ligand is critical here; bidentate phosphines like 4,6-bis(diphenylphosphino)dibenzofuran provide the necessary bite angle and electronic stabilization to support the palladium center through the high-energy transition states. This precise coordination environment minimizes catalyst deactivation and suppresses the formation of homocoupling byproducts, ensuring that the reaction pathway remains selective for the desired heterocyclic product.
Furthermore, the role of the base in this mechanistic framework cannot be overstated. Inorganic bases such as potassium carbonate serve a dual purpose: they neutralize the acidic byproducts generated during the reaction and potentially assist in the deprotonation steps required for the final aromatization of the oxazole ring. The interaction between the base, the solvent, and the catalyst creates a synergistic effect that drives the equilibrium towards product formation. From an impurity control perspective, this mechanism is advantageous because it avoids the radical pathways often associated with oxidative cyclizations, which are notorious for generating tarry, difficult-to-remove side products. Instead, the concerted nature of the palladium cycle promotes a cleaner reaction profile, simplifying the purification process and enhancing the overall mass balance of the synthesis, which is a key metric for process efficiency in commercial settings.
How to Synthesize Polysubstituted Oxazoles Efficiently
Implementing this synthesis in a practical setting requires careful attention to reagent stoichiometry and reaction parameters to maximize yield and purity. The patent data suggests a molar ratio where the benzamide is used in excess relative to the propargyl carbonate, typically in a range of 2:1 to 4:1, to drive the reaction to completion and compensate for any potential volatility or decomposition of the carbonate species. The catalyst loading is kept low, generally between 0.05 to 0.15 mmol per mmol of substrate, reflecting the high turnover number of the palladium system. Reaction times typically span from 10 to 20 hours, allowing sufficient time for the slow cyclization steps to occur without necessitating excessive thermal stress. Detailed standardized synthesis steps follow below to guide process implementation.
- Charge a reaction vessel with zero-valent palladium catalyst, phosphine ligand, benzamide compound, propargyl carbonate, and base in an organic solvent such as NMP or DMF.
- Heat the reaction mixture to a temperature range of 100-140°C and maintain stirring for 10 to 20 hours to ensure complete conversion.
- Upon completion, perform filtration and extraction, followed by silica gel column chromatography to isolate the pure polysubstituted oxazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology translates into tangible strategic benefits that extend beyond mere chemical yield. The primary advantage lies in the significant simplification of the raw material supply chain. Benzamides and propargyl carbonates are commodity chemicals available from multiple global vendors, reducing the risk of single-source dependency and price volatility. This abundance ensures a steady flow of inputs, which is critical for maintaining continuous production schedules and meeting tight delivery deadlines for downstream API manufacturers. Additionally, the elimination of exotic or hazardous reagents lowers the barrier for entry for contract manufacturing organizations, fostering a more competitive bidding environment that naturally drives down costs.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the convergence of several efficiency drivers. By utilizing inexpensive inorganic bases instead of costly organolithium or Grignard reagents, the direct material cost is substantially lowered. Furthermore, the simplified workup procedure, which involves standard filtration and extraction followed by column chromatography, reduces the consumption of solvents and stationary phases compared to more complex purification regimes. The high conversion rates observed in the patent data imply less unreacted starting material to recover and recycle, thereby minimizing waste disposal costs and maximizing the throughput of the manufacturing facility.
- Enhanced Supply Chain Reliability: Stability in the supply chain is bolstered by the robustness of the reaction conditions. The tolerance for polar aprotic solvents like DMF and NMP, which are widely produced and stocked, means that solvent shortages are unlikely to halt production. Moreover, the reaction's insensitivity to minor variations in temperature and atmosphere (as evidenced by the successful use of standard Schlenk techniques) makes it highly transferable between different manufacturing sites. This flexibility allows supply chain managers to diversify their manufacturing footprint, mitigating risks associated with geopolitical instability or localized logistical disruptions.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram quantities is facilitated by the homogeneous nature of the reaction mixture and the absence of gas-evolving steps that complicate reactor design. The use of established solvents and catalysts aligns well with existing environmental, health, and safety (EHS) protocols in most chemical plants. While palladium is a heavy metal, the low loading levels and the potential for recovery through standard scavenging resins ensure that the final product meets stringent regulatory limits for residual metals. This compliance reduces the regulatory burden on the quality assurance team and accelerates the timeline for regulatory filings.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxazole synthesis technology. These insights are derived directly from the experimental data and specifications provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers. Understanding these nuances is essential for evaluating the feasibility of integrating this route into your existing portfolio of synthetic methodologies.
Q: What are the primary advantages of this Pd-catalyzed method over traditional oxazole synthesis?
A: Unlike conventional methods that often require harsh oxidation conditions or difficult-to-prepare oxazoline precursors, this novel approach utilizes readily available benzamides and propargyl carbonates. It operates under milder thermal conditions (100-140°C) and offers a broader substrate scope, significantly simplifying the operational complexity for process chemists.
Q: Which catalysts and ligands are optimal for this transformation?
A: The patent specifies the use of zero-valent palladium sources, specifically tris(dibenzylideneacetone)dipalladium or bis(triphenylphosphine)palladium(II) dichloride. These are paired with bidentate phosphine ligands such as 4,6-bis(diphenylphosphino)dibenzofuran or bis(2-diphenylphosphino)phenyl ether to maximize catalytic efficiency and yield.
Q: Is this process suitable for large-scale manufacturing of API intermediates?
A: Yes, the process is designed for scalability. It employs common organic solvents like NMP and DMF, uses inexpensive inorganic bases like potassium carbonate, and features a straightforward workup procedure involving filtration and column chromatography, making it highly adaptable for commercial production environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Oxazole Supplier
As the demand for high-quality heterocyclic building blocks continues to surge, partnering with an experienced CDMO becomes a strategic imperative. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging deep expertise in transition metal catalysis to deliver superior results. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions seamlessly from the bench to the plant. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of polysubstituted oxazole meets the exacting standards required for pharmaceutical applications.
We invite you to collaborate with us to unlock the full potential of this innovative synthesis route. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. By engaging with us early in your development cycle, you can secure a reliable supply of high-purity intermediates while optimizing your overall manufacturing budget. Please contact us today to request specific COA data and comprehensive route feasibility assessments for your next project.
