Advanced Two-Step Synthesis of Pyrrolidyl Di-Amino-Pyrimidine Oxide for Commercial Hair Loss Solutions
The pharmaceutical and cosmetic industries are constantly seeking efficient pathways to synthesize novel hair growth promoters, specifically targeting compounds that can effectively transition hair follicles from the telogen phase to the anagen phase. Patent CN103755645A introduces a groundbreaking synthetic process for 6-(pyrrolidin-1-yl)pyrimidine-2,4-diamine 3-oxide, a potent active ingredient known for its ability to maintain the anagen phase and prevent hair loss. This technology addresses a critical gap in the market where previous synthesis reports were virtually non-existent, offering a reliable solution for manufacturers. By leveraging a straightforward two-step sequence involving oxidation and nucleophilic substitution, this method achieves high yields while maintaining operational simplicity. For R&D directors and procurement specialists, this patent represents a significant opportunity to secure a stable supply chain for high-value cosmetic and pharmaceutical intermediates without the burden of complex, multi-step legacy processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex pyrimidine N-oxides has been plagued by inefficient methodologies that rely heavily on hazardous reagents and cumbersome purification techniques. Traditional routes often require multiple protection and de-protection steps to manage the reactivity of the amino groups, leading to substantial material loss and increased production time. Furthermore, conventional substitution reactions frequently necessitate the use of expensive polar aprotic solvents such as DMF or DMSO, which are difficult to remove and pose significant environmental disposal challenges. The reliance on transition metal catalysts in some older protocols introduces the risk of heavy metal contamination, requiring additional, costly purification stages to meet stringent pharmaceutical standards. These factors collectively drive up the cost of goods sold and create bottlenecks in the supply chain, making it difficult for manufacturers to scale production economically.
The Novel Approach
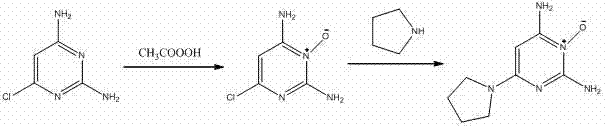
In stark contrast, the methodology outlined in CN103755645A utilizes a direct and atom-economical strategy that bypasses the need for protecting groups and exotic catalysts. The process initiates with the selective oxidation of 2,4-diamino-6-chloropyrimidine using peracetic acid, a reagent that is both cost-effective and readily available on an industrial scale. Following the formation of the N-oxide intermediate, the subsequent substitution reaction is remarkably performed in an aqueous medium, eliminating the need for toxic organic solvents in the critical coupling step. This shift to water not only drastically reduces solvent costs but also simplifies the isolation of the final product to a mere filtration and drying process. The result is a streamlined workflow that minimizes waste generation and maximizes throughput, providing a distinct competitive advantage for large-scale manufacturing.
Mechanistic Insights into N-Oxidation and Nucleophilic Substitution
The core of this synthetic success lies in the precise control of the N-oxidation mechanism, where peracetic acid acts as an electrophilic oxygen donor to the pyrimidine nitrogen. The reaction conditions are carefully optimized, with the oxidant added in batches to maintain a metastable concentration that prevents over-oxidation or decomposition of the sensitive amino groups. The presence of electron-donating amino groups at the 2 and 4 positions activates the pyrimidine ring, facilitating the attack on the nitrogen at the 3-position while the chlorine at the 6-position remains intact for the subsequent step. This selectivity is crucial, as it ensures that the chloro leaving group is preserved for the nucleophilic displacement, thereby maintaining the structural integrity required for the final biological activity. The use of alcoholic solvents in this first step further stabilizes the intermediate, allowing for high conversion rates without the formation of significant side products.
Following oxidation, the mechanism shifts to a classic nucleophilic aromatic substitution (SnAr) driven by the strong electron-withdrawing effect of the newly formed N-oxide moiety. The N-oxide group significantly activates the C-6 position towards nucleophilic attack by pyrrolidine (tetramethyleneimine), lowering the activation energy required for the displacement of the chloride ion. The addition of an alkali base, such as sodium hydroxide or triethylamine, serves to neutralize the hydrochloric acid byproduct, driving the equilibrium towards the formation of the desired product. Conducting this reaction in water is particularly advantageous because the high polarity of the solvent stabilizes the transition state and the ionic intermediates involved in the SnAr pathway. This mechanistic understanding allows for tight control over impurity profiles, ensuring that the final product meets the rigorous purity specifications demanded by the cosmetic and pharmaceutical sectors.
How to Synthesize 6-(Pyrrolidin-1-yl)pyrimidine-2,4-diamine 3-oxide Efficiently
To implement this synthesis effectively, manufacturers must adhere to the specific thermal and stoichiometric parameters defined in the patent to ensure optimal yield and safety. The process begins with the careful cooling of the reaction mixture during the addition of peracetic acid to manage the exotherm, followed by a prolonged stirring period at room temperature to ensure complete oxidation. Once the intermediate is isolated, it is redissolved in water for the substitution step, where temperature control between 60°C and 90°C is critical for driving the reaction to completion within a short timeframe. The detailed standardized synthesis steps, including specific mass ratios and reaction times, are provided in the guide below to assist technical teams in replicating this high-efficiency route.
- Oxidize 2,4-diamino-6-chloropyrimidine using peracetic acid in alcohol solvent at room temperature to form the N-oxide intermediate.
- Dissolve the N-oxide intermediate in water and add pyrrolidine (tetramethyleneimine) along with an alkali base.
- Heat the mixture to 60-90°C for 1-3 hours, then cool, filter, wash, and dry to obtain the final high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers transformative benefits that extend far beyond simple chemical yield improvements. By shifting the solvent system for the substitution step from expensive organic liquids to water, the process dramatically lowers the variable costs associated with solvent purchase, recovery, and disposal. This reduction in operational complexity translates directly into a more resilient supply chain, as the reliance on specialized, potentially regulated solvents is minimized. Furthermore, the use of commodity chemicals like peracetic acid and pyrrolidine ensures that raw material availability remains high, shielding the production schedule from the volatility often seen with niche reagents. The simplicity of the workup, which relies on filtration rather than column chromatography, significantly reduces the labor hours and equipment time required per batch, enhancing overall facility throughput.
- Cost Reduction in Manufacturing: The elimination of organic solvents in the second reaction step represents a massive opportunity for cost optimization, as water is essentially free compared to pharmaceutical-grade alcohols or aprotic solvents. Additionally, the avoidance of transition metal catalysts removes the necessity for expensive scavenging resins or activated carbon treatments, further stripping away hidden processing costs. The high overall yield of over 76% means that less starting material is wasted, maximizing the return on investment for every kilogram of 2,4-diamino-6-chloropyrimidine purchased. These cumulative efficiencies result in a substantially lower cost of goods sold, allowing for more competitive pricing in the final market.
- Enhanced Supply Chain Reliability: Sourcing stability is greatly improved because the key reagents, including peracetic acid and pyrrolidine, are produced globally in massive quantities for various industrial applications. Unlike proprietary catalysts that may have single-source suppliers, these commodities can be procured from multiple vendors, reducing the risk of supply disruptions. The robustness of the reaction conditions, which tolerate slight variations in temperature and mixing, ensures consistent batch-to-batch quality even when scaling up to multi-ton production. This reliability is essential for maintaining continuous manufacturing schedules and meeting the just-in-time delivery requirements of major pharmaceutical clients.
- Scalability and Environmental Compliance: The process is inherently green, generating minimal hazardous waste due to the use of water as the primary reaction medium and the absence of heavy metals. This aligns perfectly with increasingly strict environmental regulations, reducing the permitting burden and potential fines associated with volatile organic compound (VOC) emissions. The scalability is proven by the simple unit operations involved—mixing, heating, and filtering—which are easily replicated in large-scale reactors without the need for specialized equipment. Consequently, manufacturers can rapidly ramp up production capacity to meet surging demand for hair growth treatments without facing significant engineering bottlenecks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical decision-makers. Understanding these nuances is vital for assessing the feasibility of integrating this process into your existing manufacturing portfolio.
Q: What are the key advantages of this synthesis route over traditional methods?
A: This process eliminates the need for expensive organic solvents in the second step by utilizing water, significantly reducing raw material costs and environmental waste. Furthermore, the purification is simplified to direct filtration, avoiding complex chromatographic separations.
Q: What is the expected yield and purity profile for this intermediate?
A: The patent data indicates a robust overall yield exceeding 76.5%, with individual step yields of approximately 90% for oxidation and 85% for substitution. The simplicity of the workup ensures high product purity suitable for pharmaceutical applications.
Q: Is this process scalable for industrial manufacturing?
A: Yes, the process is explicitly designed for industrial suitability, utilizing stable reagents like peracetic acid and common bases like sodium hydroxide. The exothermic nature of the oxidation is managed via controlled addition, ensuring safety at large scales.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrrolidyl Di-Amino-Pyrimidine Oxide Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of 6-(pyrrolidin-1-yl)pyrimidine-2,4-diamine 3-oxide in the next generation of hair care formulations. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of this critical intermediate meets the highest global standards. We are committed to translating the theoretical advantages of patent CN103755645A into tangible commercial reality for our partners.
We invite you to collaborate with us to leverage this cost-effective and environmentally friendly synthesis technology for your product pipeline. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how we can become your strategic partner in delivering high-quality hair growth solutions to the global market.
