Advanced Iodide-Catalyzed Synthesis of Alpha-Cyanoamines for Commercial Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and safe methodologies for constructing complex nitrogen-containing scaffolds. Patent CN103274964B introduces a groundbreaking approach for the preparation of alpha-cyanoamines, a class of compounds with significant physiological activity and widespread application as key structural units in biochemistry and drug discovery. This technology represents a paradigm shift from traditional, hazardous cyanation protocols to a greener, radical exchange reaction mediated by inexpensive iodide catalysts. By leveraging tert-butyl hydroperoxide (TBHP) as an oxidant in a mixed solvent system of water and hydrophilic organic solvents, this method achieves high target product yields under mild conditions. For R&D directors and procurement specialists, this patent offers a compelling alternative that addresses both safety concerns and cost efficiency, positioning it as a critical asset for the reliable supply of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alpha-cyanoamines has relied heavily on oxidative cyanation strategies that pose significant safety and environmental challenges. Prominent prior art, such as the work by Murahashi, utilized sodium cyanide (NaCN) or hydrogen cyanide (HCN) as cyanide sources in the presence of ruthenium trichloride (RuCl3) catalysts. While effective, these methods involve extremely toxic reagents that require stringent safety protocols and specialized waste treatment facilities, drastically increasing operational costs. Furthermore, the reliance on noble metal catalysts like ruthenium introduces issues regarding metal residue removal, which is critical for pharmaceutical grade materials. Other approaches employing trimethylsilyl cyanide (TMSCN) with iron or gold catalysts also suffer from the high cost and environmental burden associated with metal catalyst preparation and disposal. These conventional pathways often limit the commercial viability of alpha-cyanoamine production due to narrow substrate scope and complex post-treatment requirements.
The Novel Approach
In stark contrast, the methodology disclosed in CN103274964B utilizes a metal-free catalytic system based on iodides, such as tetrabutylammonium iodide (Bu4NI), coupled with phenylacetonitrile as the cyanide source. This innovative route eliminates the need for toxic inorganic cyanides and expensive transition metals, thereby simplifying the regulatory compliance landscape for manufacturing. The reaction proceeds via a free radical exchange mechanism in a benign solvent mixture of water and tert-butanol, typically at temperatures ranging from 40°C to 80°C. This mild thermal profile not only enhances energy efficiency but also minimizes the formation of thermal degradation byproducts. The simplicity of the workup procedure, involving standard quenching with sodium sulfite followed by extraction and chromatography, ensures that the process is amenable to cost reduction in pharma manufacturing while maintaining high atom economy and product purity.
Mechanistic Insights into Iodide-Catalyzed Radical Exchange
The core of this technological advancement lies in the efficient generation of radical species facilitated by the iodide catalyst. The reaction initiates with the interaction between the iodide anion and the oxidant, tert-butyl hydroperoxide (TBHP), generating reactive radical intermediates. These species abstract a hydrogen atom from the alpha-position of the tertiary amine substrate, creating an alpha-amino radical. Simultaneously, the phenylacetonitrile undergoes activation to form a benzylic radical. The subsequent coupling of these two radical species leads to the formation of the desired alpha-cyanoamine bond. This mechanism avoids the harsh conditions typically required for nucleophilic substitution or electrophilic cyanation, allowing for the preservation of sensitive functional groups on the amine scaffold. The use of iodide is particularly advantageous due to its redox properties, which enable rapid turnover without the accumulation of stable metal-complex impurities that often plague transition-metal catalyzed reactions.
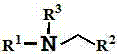
Furthermore, the impurity profile of this reaction is exceptionally clean compared to traditional methods. The mild oxidative conditions prevent over-oxidation of the amine to amides or nitriles, which are common side reactions in aggressive cyanation protocols. The solvent system, comprising water and tert-butanol, plays a crucial role in stabilizing the radical intermediates and facilitating phase transfer, ensuring consistent reaction kinetics across different substrate classes. For quality control teams, this translates to a more predictable impurity spectrum that is easier to characterize and remove during purification. The broad tolerance of the catalytic system towards various substituents on the aromatic ring of the amine, including electron-donating and electron-withdrawing groups, underscores its versatility for synthesizing diverse libraries of bioactive molecules.
How to Synthesize Alpha-Cyanoamine Efficiently
The practical implementation of this synthesis route is straightforward and designed for reproducibility in both laboratory and pilot plant settings. The process begins by charging a reaction vessel with the iodide catalyst, typically at a loading of 20 mol%, along with the tertiary amine and phenylacetonitrile in a molar ratio optimized for maximum conversion. The addition of the oxidant TBHP is carefully controlled to manage the exotherm, although the reaction temperature is maintained moderately between 40°C and 80°C. Detailed standardized synthetic steps see the guide below.
- Mix iodide catalyst (e.g., Bu4NI, 20 mol%), tertiary amine, phenylacetonitrile, and oxidant (TBHP) in a water and tert-butanol solvent mixture.
- Heat the reaction mixture to a temperature between 40°C and 80°C (optimally 60°C) and stir for 12 to 48 hours to facilitate the radical exchange reaction.
- Quench the reaction with saturated sodium sulfite, perform aqueous workup including washing and extraction with ethyl acetate, and purify the crude product via flash column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this iodide-catalyzed technology offers substantial benefits that directly impact the bottom line and supply chain resilience. The elimination of precious metal catalysts removes a major cost driver and mitigates the risk associated with the volatility of metal prices. Additionally, the use of phenylacetonitrile as a cyanide source circumvents the strict regulatory controls and security measures required for handling inorganic cyanides, streamlining the procurement process and reducing administrative overhead. The simplified workup procedure reduces solvent consumption and waste generation, aligning with modern sustainability goals and lowering disposal costs. These factors collectively contribute to a more robust and economically viable supply chain for critical pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The replacement of expensive noble metal catalysts like ruthenium or gold with commodity iodide salts results in significant raw material cost savings. Moreover, the absence of heavy metal residues eliminates the need for costly scavenging steps or specialized purification technologies to meet strict residual metal specifications. The mild reaction conditions also reduce energy consumption compared to high-temperature processes, further enhancing the overall cost efficiency of the manufacturing campaign.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including various tertiary amines and phenylacetonitrile, are commercially available in bulk quantities from multiple global suppliers. This diversity in sourcing options reduces dependency on single-source vendors and mitigates the risk of supply disruptions. The stability of the reagents and the robustness of the reaction conditions ensure consistent batch-to-batch quality, which is essential for maintaining uninterrupted production schedules for downstream API synthesis.
- Scalability and Environmental Compliance: The use of a water-containing solvent system significantly reduces the environmental footprint of the process by minimizing the volume of volatile organic compounds (VOCs) emitted. The mild thermal profile allows for safer scale-up in large reactors without the need for complex cooling systems to manage extreme exotherms. This ease of scale-up facilitates the commercial production of complex intermediates, ensuring that supply can meet demand fluctuations without compromising on safety or environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical decision-makers evaluating this route for their specific applications.
Q: What are the safety advantages of this iodide-catalyzed method compared to traditional cyanation?
A: Unlike traditional methods utilizing highly toxic sodium cyanide (NaCN) or hydrogen cyanide (HCN) with noble metal catalysts, this process employs phenylacetonitrile as a safer cyanide source and inexpensive iodide salts, significantly reducing hazardous waste and operational risks.
Q: Can this synthesis method accommodate diverse tertiary amine substrates?
A: Yes, the patent demonstrates broad substrate scope, successfully converting various tertiary amines including those with different alkyl chains (methyl, ethyl, propyl, butyl) and substituted anilines (halogenated, methoxy) into corresponding alpha-cyanoamines with good yields.
Q: Is this process suitable for large-scale industrial production?
A: The method utilizes mild reaction conditions (40-80°C), readily available raw materials, and a simple aqueous-organic solvent system, making it highly scalable and cost-effective for commercial manufacturing without requiring complex equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Cyanoamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this iodide-catalyzed synthesis for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of alpha-cyanoamine delivered meets the highest industry standards for quality and consistency.
We invite you to collaborate with our technical team to explore how this cost-effective and safe methodology can be integrated into your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your next development milestone.
