Scalable Synthesis of Indole-Based Na-Phos Ligands for Commercial Asymmetric Catalysis
The landscape of asymmetric catalysis is continually evolving, driven by the demand for more efficient and accessible chiral ligands that can streamline the production of high-value pharmaceutical intermediates. A significant advancement in this field is documented in patent CN112679540B, which discloses a novel class of chiral sulfinamide phosphine compounds known as Na-Phos, built upon a versatile indole ring skeleton. This technology represents a paradigm shift from traditional ligand synthesis, which often suffers from prohibitive costs and lengthy multi-step sequences, by offering a streamlined pathway to access optically pure compounds. The core innovation lies in the strategic combination of an indole scaffold with chiral sulfinamide auxiliaries, enabling the precise construction of multiple stereocenters through a concise series of condensation, substitution, and addition reactions. For R&D directors and procurement specialists alike, this development signals a new opportunity to secure reliable chiral ligand supplier partnerships that can deliver high-purity pharmaceutical intermediates without the traditional bottlenecks associated with complex chiral pool synthesis. The ability to access all four stereoisomers—(R, RS), (S, RS), (R, SS), and (S, SS)—from a common set of precursors further enhances the utility of this platform for diverse asymmetric transformations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral phosphine ligands has been plagued by significant economic and technical hurdles that impede their widespread adoption in industrial manufacturing processes. Traditional routes frequently rely on expensive, naturally derived chiral starting materials that are subject to supply chain volatility and price fluctuations, creating uncertainty for long-term production planning. Furthermore, many established methods require extensive synthetic sequences involving multiple protection and deprotection steps, which not only reduce overall atom economy but also accumulate impurities that are difficult to remove in later stages. The reliance on precious metal catalysts or specialized reagents in older methodologies often necessitates rigorous purification protocols to meet the stringent purity specifications required for active pharmaceutical ingredients, thereby driving up operational expenditures. Additionally, the inability to easily access different stereoisomers from a single synthetic route limits the flexibility of process chemists when optimizing enantioselectivity for specific substrate classes. These cumulative inefficiencies result in prolonged lead times and elevated costs, making the commercial scale-up of complex chiral ligands a challenging endeavor for many fine chemical manufacturers seeking cost reduction in asymmetric catalysis manufacturing.
The Novel Approach
In stark contrast to these legacy challenges, the methodology outlined in the patent introduces a robust and modular synthetic strategy that leverages commercially available and inexpensive raw materials to construct the Na-Phos framework. By utilizing 2-methylindole-3-carboxaldehyde as a foundational building block, the process bypasses the need for rare natural products, ensuring a stable and predictable supply chain for the core skeleton. The synthetic route is remarkably concise, achieving the target complexity through just four primary transformation steps that are amenable to optimization and scaling. A critical advantage of this approach is the divergent synthesis capability, where the stereochemical outcome is controlled by the choice of chiral sulfinamide and the subsequent nucleophilic addition reagent, allowing for the systematic generation of all possible stereoisomers without the need for chiral resolution. This modularity not only accelerates the screening process for optimal catalytic performance but also simplifies inventory management for suppliers. The use of standard organic solvents and reagents further lowers the barrier to entry for manufacturing, facilitating the commercial scale-up of complex chiral ligands with significantly reduced environmental impact and operational risk compared to conventional techniques.
Mechanistic Insights into Indole-Based Phosphine Construction
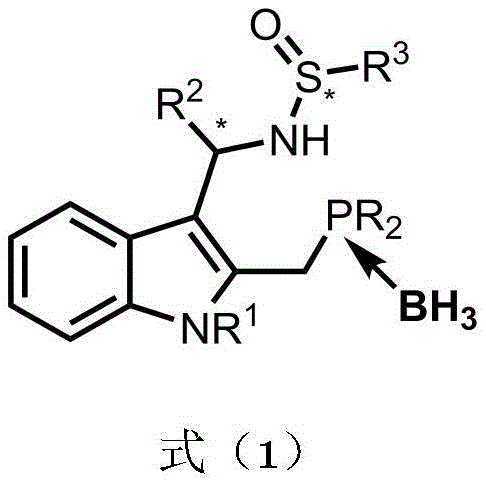
The chemical elegance of the Na-Phos synthesis lies in its precise control over stereochemistry through a well-defined sequence of functional group manipulations centered on the indole nucleus. The process initiates with the protection of the indole nitrogen, which is crucial for preventing side reactions and directing subsequent substitutions to the desired positions on the heterocyclic ring. Following this, a condensation reaction with a chiral sulfinamide establishes the first key stereocenter, leveraging the steric bulk of the tert-butyl group to induce diastereoselectivity during the formation of the sulfinylimine intermediate. The subsequent lithiation of the methyl group at the 2-position of the indole ring is a pivotal step, generating a reactive carbanion that undergoes nucleophilic substitution with diaryl or dialkyl chlorophosphines. This step is performed in a one-pot fashion with immediate borane protection, which stabilizes the sensitive phosphine moiety against oxidation and simplifies the isolation of the intermediate phosphinimine. The mechanistic pathway ensures that the phosphorus atom is introduced with high fidelity, setting the stage for the final stereochemical diversification.
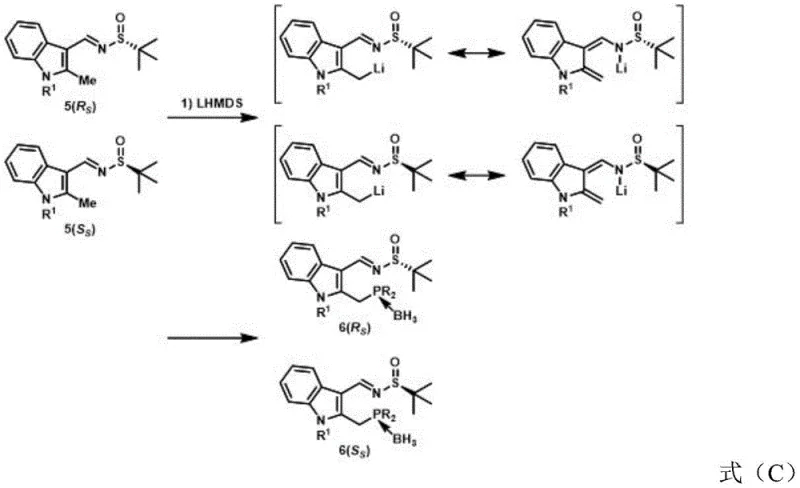
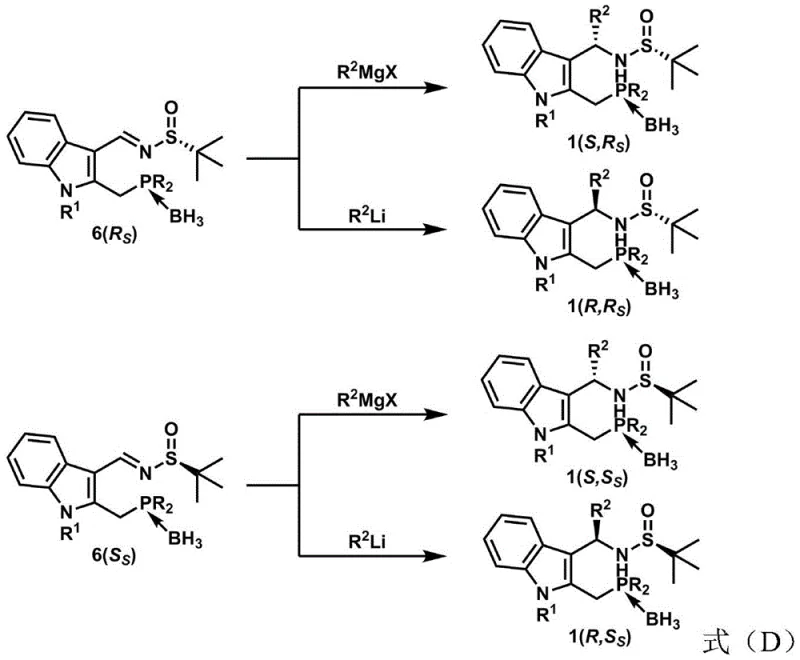
The final and perhaps most critical phase of the synthesis involves the nucleophilic addition to the imine bond, which establishes the second stereocenter and completes the chiral architecture of the ligand. By selecting specific metal reagents, such as Grignard reagents or organolithium compounds, chemists can dictate the facial selectivity of the addition, thereby accessing different diastereomers from the same phosphinimine precursor. This divergence is governed by the interplay between the existing chirality of the sulfinamide auxiliary and the steric properties of the incoming nucleophile, a phenomenon that allows for the comprehensive mapping of stereochemical space. The resulting compounds possess a rigid indole backbone that provides a stable platform for metal coordination in catalytic cycles, while the tunable R groups on the phosphorus and nitrogen atoms offer endless opportunities for fine-tuning electronic and steric properties. This level of control over impurity profiles and isomeric purity is essential for R&D teams focused on developing robust catalytic processes that meet regulatory standards for drug substance manufacturing.
How to Synthesize Na-Phos Efficiently
The practical implementation of this synthesis route requires careful attention to reaction conditions and reagent quality to ensure optimal yields and stereochemical integrity throughout the process. The standardized protocol begins with the N-protection of the indole aldehyde, followed by condensation with the chiral auxiliary, lithiation/phosphination, and finally nucleophilic addition. Each step has been optimized to balance reaction rate with selectivity, utilizing common laboratory equipment and solvents that are readily available in industrial settings. Detailed procedural guidelines regarding temperature control, stoichiometry, and workup procedures are essential for reproducing the high levels of performance reported in the patent literature. For technical teams looking to adopt this methodology, understanding the nuances of the lithiation step and the handling of air-sensitive phosphine intermediates is paramount to success.
- Protect the indole nitrogen of 2-methylindole-3-carboxaldehyde using a base and sulfonyl chloride to form the protected aldehyde intermediate.
- Condense the protected aldehyde with chiral tert-butylsulfinamide using a condensing agent to generate the chiral sulfinylimine.
- Perform lithiation at the 2-position methyl group followed by substitution with chlorophosphine and in-situ borane protection.
- React the borane-protected phosphinimine with Grignard or organolithium reagents to establish the final stereocenters and yield the target Na-Phos ligand.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of the Na-Phos synthesis technology offers compelling benefits that directly address the pain points of cost and reliability in the fine chemicals sector. The reliance on commodity chemicals such as indole derivatives and simple sulfinamides eliminates the dependency on volatile markets for exotic chiral building blocks, thereby stabilizing raw material costs and mitigating supply risks. This shift towards abundant feedstocks translates into substantial cost savings in manufacturing, as the elimination of expensive resolution steps and precious metal catalysts reduces the overall bill of materials. Furthermore, the shortened synthetic sequence decreases the consumption of solvents and energy per kilogram of product, aligning with sustainability goals and reducing waste disposal costs associated with complex chemical manufacturing. For supply chain heads, the robustness of the reaction conditions implies a lower risk of batch failures and a more predictable production schedule, which is critical for maintaining continuity in the supply of high-purity pharmaceutical intermediates to downstream clients.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the use of low-cost starting materials and the avoidance of chromatographic purifications in favor of crystallization or extraction methods where possible. By streamlining the synthesis to fewer steps, the cumulative yield loss typically seen in long linear sequences is minimized, resulting in higher overall throughput and better asset utilization. The removal of transition metal catalysts from the final ligand structure also means that costly metal scavenging processes are unnecessary, further driving down operational expenses. These factors combine to create a highly competitive cost structure that allows suppliers to offer premium chiral ligands at prices that support the economic feasibility of large-scale asymmetric synthesis projects.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the accessibility of the required reagents, which are produced by multiple global vendors, reducing the risk of single-source bottlenecks. The simplicity of the chemistry allows for flexible manufacturing across different facilities, enabling distributed production strategies that can respond quickly to regional demand fluctuations. Moreover, the stability of the borane-protected intermediates facilitates safer storage and transportation, reducing the logistical complexities associated with handling highly reactive or unstable chemical species. This reliability ensures that reducing lead time for high-purity chiral phosphines becomes a tangible reality, allowing pharmaceutical companies to accelerate their own development timelines without fear of material shortages.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with reaction conditions that can be safely translated from gram-scale laboratory experiments to multi-ton commercial production without significant re-engineering. The use of standard solvents like THF and DME simplifies solvent recovery and recycling systems, contributing to a smaller environmental footprint and easier compliance with increasingly stringent environmental regulations. The high atom economy of the condensation and addition steps minimizes waste generation, supporting green chemistry initiatives that are becoming a prerequisite for vendor qualification in the global pharmaceutical industry. This alignment with environmental standards not only reduces regulatory risk but also enhances the brand value of the supplier as a responsible partner in sustainable chemical manufacturing.
Frequently Asked Questions (FAQ)
To assist stakeholders in evaluating the potential of this technology for their specific applications, we have compiled answers to common technical and commercial inquiries based on the patent data. These questions address the versatility of the ligand, the scalability of the process, and the quality standards achievable through this synthetic route. Understanding these aspects is crucial for making informed decisions regarding the integration of Na-Phos ligands into existing catalytic workflows or new process development campaigns.
Q: What are the key advantages of the Na-Phos ligand synthesis described in CN112679540B?
A: The synthesis utilizes commercially available and low-cost raw materials, features a short reaction sequence, and allows for the efficient preparation of all four stereoisomers with high selectivity.
Q: How does this method improve supply chain reliability for chiral catalysts?
A: By avoiding expensive proprietary starting materials and complex resolution steps, the process ensures consistent availability of raw materials and reduces dependency on scarce chiral pools.
Q: Is the Na-Phos ligand suitable for large-scale industrial applications?
A: Yes, the robust reaction conditions, use of common solvents like THF and DME, and high yields in key steps make the process highly amenable to commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Na-Phos Supplier
The technical potential of the Na-Phos ligand system is immense, offering a versatile platform for advancing asymmetric catalysis in the production of complex drug molecules. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this innovative chemistry from the laboratory to the marketplace. Our facility is equipped with rigorous QC labs and advanced analytical instrumentation to ensure that every batch meets stringent purity specifications, providing our clients with the confidence they need to proceed with clinical and commercial manufacturing. We understand the critical nature of chiral intermediates in the pharmaceutical value chain and are committed to delivering consistent quality and performance.
We invite you to engage with our technical procurement team to discuss how this technology can optimize your specific synthesis requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this ligand system for your projects. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your target molecules, ensuring a seamless transition to a more efficient and cost-effective supply chain solution.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
