Advanced Manufacturing of 2-Amino-6-(Piperidine-4-Acyl) Pyridine Derivatives for Migraine Therapeutics
Introduction to Next-Generation Migraine Drug Intermediates
The pharmaceutical industry is constantly seeking robust and safe pathways for synthesizing critical intermediates, particularly for neurological therapeutics targeting acute migraine. Patent CN113637003A introduces a groundbreaking methodology for the preparation of 2-amino-6-(piperidine-4-acyl) pyridine derivatives, which serve as pivotal building blocks for Serotonin 5-HT1F agonist drugs. Unlike legacy processes that rely on hazardous gaseous ammonia or costly transition metal couplings, this innovation utilizes stable ammonium salts under mild thermal conditions. This shift not only enhances operator safety but also streamlines the purification workflow, delivering exceptional chemical purity essential for regulatory compliance. As a reliable pharmaceutical intermediate supplier, understanding these mechanistic advancements is crucial for securing a stable supply chain for next-generation CNS medications.
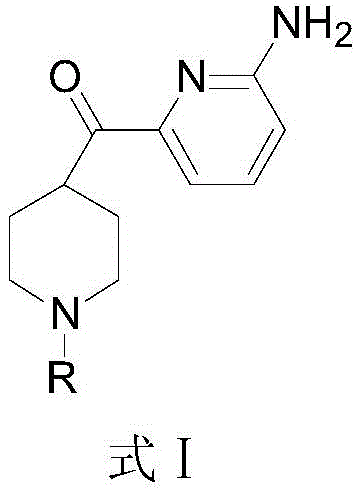
The Limitations of Conventional Methods vs. The Novel Approach
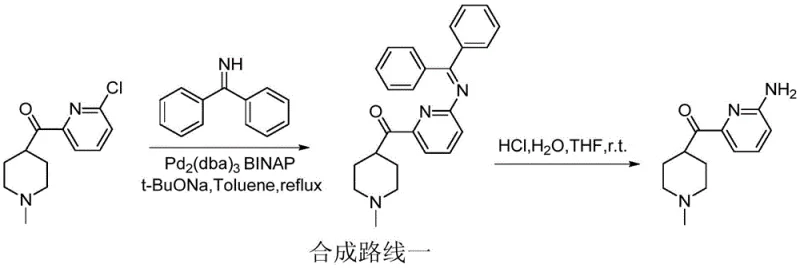
The Limitations of Conventional Methods
Historically, the synthesis of this pyridine scaffold has been plagued by significant operational hurdles and economic inefficiencies. Traditional Route I, for instance, employs palladium-catalyzed cross-coupling reactions requiring ligands like BINAP and bases such as sodium tert-butoxide, followed by complex deprotection steps that often result in yield losses around 20%. Furthermore, Route II necessitates the use of high-pressure ammonia gas at elevated temperatures, posing severe safety risks regarding containment and requiring specialized autoclave infrastructure that limits scalability. Other multi-step sequences involving protection-deprotection strategies with sulfonyl chlorides or acetyl groups introduce unnecessary atom economy waste and extend lead times, making cost reduction in API manufacturing difficult to achieve with legacy technologies.
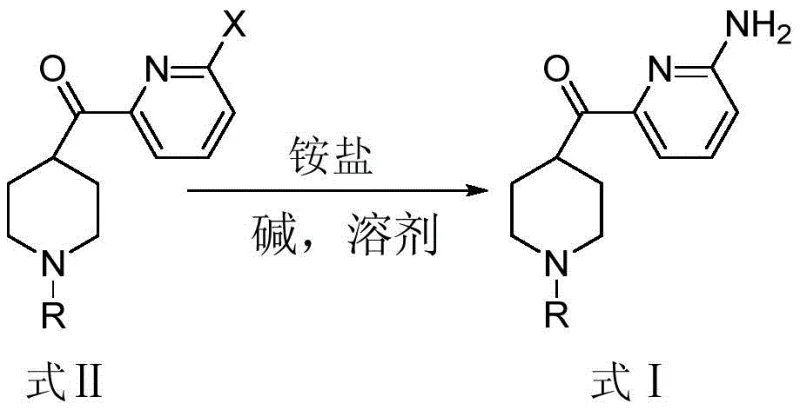
The Novel Approach
The disclosed invention fundamentally reimagines this transformation by replacing volatile ammonia gas with solid ammonium salts, such as ammonium chloride or ammonium carbonate, in the presence of a copper catalyst. This approach, illustrated in the reaction scheme below, operates at atmospheric pressure and moderate temperatures between 60-90°C, drastically simplifying the reactor requirements. By utilizing ethylene glycol as a high-boiling polar solvent, the system facilitates effective mass transfer without the need for cryogenic conditions or inert gas manifolds typically associated with sensitive organometallic chemistry. This streamlined protocol not only mitigates the risk of explosive hazards but also ensures a cleaner reaction profile, directly addressing the pain points of procurement managers seeking cost-effective and safe production methodologies.
Mechanistic Insights into Copper-Catalyzed Amination
The core of this technological breakthrough lies in the efficient copper-catalyzed nucleophilic aromatic substitution mechanism. In this system, the cuprous oxide (Cu2O) catalyst activates the halogenated pyridine substrate, likely forming a transient copper-amine complex that facilitates the displacement of the halide leaving group by the nitrogen nucleophile generated in situ from the ammonium salt. The presence of a strong base, such as sodium hydroxide, is critical for deprotonating the ammonium species to generate the reactive ammonia equivalent necessary for the attack on the electron-deficient pyridine ring. This catalytic cycle avoids the formation of stable off-cycle complexes that often plague palladium systems, thereby maintaining high turnover numbers and ensuring consistent reaction kinetics throughout the batch process.
From an impurity control perspective, the mild reaction conditions play a pivotal role in maintaining product integrity. High-temperature ammonia reactions often lead to over-alkylation or ring degradation, generating difficult-to-remove byproducts that compromise the purity profile. In contrast, the controlled thermal environment of 60-90°C combined with the specific stoichiometry of ammonium salt to base (optimized at 1:1.1) minimizes side reactions. The subsequent workup involving acid-base extraction effectively partitions the basic amine product from neutral organic impurities and inorganic salts, resulting in a crude material with purity levels exceeding 99%, which significantly reduces the burden on downstream crystallization or chromatography units.
How to Synthesize 2-Amino-6-(Piperidine-4-Acyl) Pyridine Efficiently
The operational simplicity of this method makes it highly attractive for process chemists aiming to transfer technology from the lab to the pilot plant. The procedure involves a straightforward charge of reagents into a standard reactor, followed by a heating phase and a conventional aqueous workup. Detailed standardized synthesis steps see the guide below.
- Combine 2-halo-6-(piperidine-4-acyl)pyridine, ammonium salt (e.g., NH4Cl), alkali (e.g., NaOH), and cuprous oxide catalyst in ethylene glycol solvent.
- Heat the reaction mixture to 60-90°C and stir for 0.5-10 hours to facilitate the nucleophilic substitution.
- Cool to room temperature, extract with organic solvent, wash with acid/base to isolate the high-purity target amine product.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement specialists, the adoption of this ammonium salt-based route offers transformative benefits regarding cost structure and logistical reliability. By eliminating the dependency on specialized high-pressure equipment and expensive noble metal catalysts, manufacturers can significantly lower capital expenditure and raw material costs. The use of commodity chemicals like ammonium chloride and ethylene glycol ensures a resilient supply chain that is less susceptible to the market volatility often seen with specialized organometallic reagents. Furthermore, the simplified isolation procedure reduces solvent consumption and waste generation, aligning with modern environmental sustainability goals while enhancing overall process economics.
- Cost Reduction in Manufacturing: The elimination of palladium catalysts and high-pressure ammonia infrastructure leads to substantial cost savings in both raw materials and equipment maintenance. The process utilizes inexpensive ammonium salts and copper catalysts, which are orders of magnitude cheaper than phosphine ligands and noble metals. Additionally, the high yield and purity reduce the need for extensive purification steps, lowering solvent usage and energy consumption per kilogram of product, thereby optimizing the total cost of ownership for the intermediate.
- Enhanced Supply Chain Reliability: Sourcing stability is greatly improved as the key reagents, including ammonium chloride, sodium hydroxide, and cuprous oxide, are globally available commodity chemicals with robust supply networks. This contrasts sharply with routes relying on custom-synthesized ligands or hazardous gases that require specialized logistics and storage facilities. The ability to manufacture under atmospheric pressure further reduces regulatory hurdles and transportation risks, ensuring consistent delivery schedules and reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The process is inherently scalable due to its operation under mild thermal conditions and atmospheric pressure, removing the engineering bottlenecks associated with high-pressure hydrogenation or ammonolysis reactors. The reduced generation of heavy metal waste, specifically avoiding palladium residues, simplifies wastewater treatment and disposal protocols. This environmental compatibility facilitates easier permitting for commercial scale-up of complex pharmaceutical intermediates, allowing manufacturers to rapidly increase capacity from pilot batches to multi-ton production without significant infrastructure upgrades.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the experimental data and process parameters detailed in the patent literature, providing a clear understanding of the method's capabilities and limitations for potential partners and stakeholders evaluating this technology for their supply chains.
Q: What are the safety advantages of this new synthesis route compared to traditional methods?
A: This method eliminates the need for high-pressure ammonia gas and expensive palladium catalysts, significantly reducing operational risks and equipment costs.
Q: What yields and purity levels can be achieved with this ammonium salt protocol?
A: Experimental data demonstrates yields exceeding 95% with product purity reaching 99.9%, suitable for direct use in downstream API synthesis.
Q: Is this process scalable for industrial production of migraine drug intermediates?
A: Yes, the use of commodity chemicals like ammonium chloride and simple workup procedures makes this route highly adaptable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino-6-(Piperidine-4-Acyl) Pyridine Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in the development of life-saving migraine therapies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to market availability is seamless and efficient. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify every batch against the highest industry standards, guaranteeing the quality required for global regulatory submissions.
We invite you to collaborate with us to leverage this advanced manufacturing technology for your specific project needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized processes can enhance your supply chain efficiency and drive down overall production costs.
