Advanced Thermal Cyclization for Polysubstituted Dibenzoylbenzene Derivatives: Scalable Manufacturing Insights
Advanced Thermal Cyclization for Polysubstituted Dibenzoylbenzene Derivatives: Scalable Manufacturing Insights
The landscape of fine chemical synthesis is constantly evolving, driven by the need for more complex molecular architectures that can serve as potent scaffolds in medicinal chemistry. A significant breakthrough in this domain is documented in Chinese Patent CN110746305A, which discloses a novel and efficient method for synthesizing polysubstituted dibenzoylbenzene derivatives. This technology leverages a sophisticated cascade reaction sequence involving a thermal Diels-Alder cycloaddition followed by spontaneous air oxidation. Unlike traditional methods that often rely on harsh Lewis acids or multi-step functional group manipulations, this approach utilizes a unique tetrayne precursor that undergoes intramolecular rearrangement to form a reactive benzyne intermediate. This intermediate then traps 2,4-diphenyloxazole to construct the highly substituted benzene core with remarkable precision. The ability to access such structurally diverse and complex frameworks through a relatively straightforward thermal process represents a substantial value proposition for R&D teams seeking new chemical space for drug discovery programs.
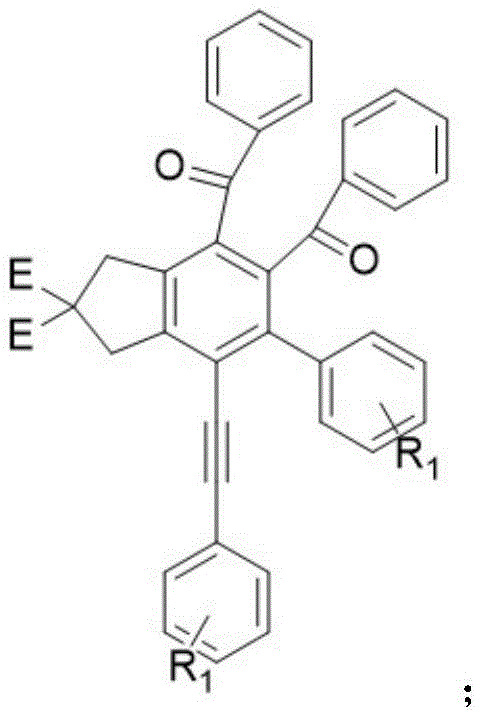
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of polysubstituted benzene rings, particularly those bearing multiple carbonyl functionalities like dibenzoyl motifs, has been a formidable challenge in organic synthesis. Conventional strategies typically rely on Friedel-Crafts acylation, which, while fundamental, suffers from significant drawbacks when applied to complex substrates. These reactions often lack regioselectivity, leading to difficult-to-separate isomeric mixtures that drastically reduce overall process efficiency and increase purification costs. Furthermore, traditional acylation requires stoichiometric amounts of corrosive Lewis acids such as aluminum chloride, generating large volumes of acidic waste that pose environmental and disposal challenges. Additionally, introducing multiple substituents in specific patterns often necessitates protecting group strategies, adding several synthetic steps that lower the overall atom economy. For procurement and supply chain managers, these inefficiencies translate into higher raw material consumption, longer lead times, and increased regulatory burden regarding waste management, making conventional routes less attractive for large-scale commercial production of high-value intermediates.
The Novel Approach
In stark contrast to these legacy methods, the technology outlined in CN110746305A introduces a paradigm shift by utilizing a pericyclic reaction strategy that builds molecular complexity in a single operational step. The core innovation lies in the use of a specifically designed tetrayne precursor which, upon heating, generates a transient benzyne species capable of undergoing a [4+2] Diels-Alder cycloaddition with 2,4-diphenyloxazole. This approach bypasses the regioselectivity issues inherent in electrophilic aromatic substitution because the connectivity is dictated by the orbital symmetry and geometry of the diene and dienophile. Moreover, the subsequent oxidation step, which installs the ketone functionalities, occurs spontaneously upon exposure to ambient air during the cooling phase, eliminating the need for external oxidants or catalysts. This streamlined workflow not only simplifies the reaction setup but also enhances the safety profile by avoiding hazardous oxidizing agents. For manufacturing teams, this translates to a process that is inherently safer, cleaner, and more predictable, facilitating easier technology transfer from the laboratory to pilot and commercial scales.
Mechanistic Insights into Thermal Benzyne Generation and DA Cycloaddition
To fully appreciate the elegance of this synthesis, one must delve into the mechanistic details of the tetrayne transformation. The process begins with the thermal activation of the tetrayne precursor in a high-boiling solvent like toluene at temperatures between 100°C and 110°C. Under these conditions, the conjugated alkyne system undergoes a Bergman-like cyclization or similar electrocyclic rearrangement to generate a highly reactive benzyne intermediate. This fleeting species acts as a powerful dienophile, immediately engaging with the 2,4-diphenyloxazole, which serves as the diene component in this inverse electron-demand Diels-Alder reaction. The choice of 2,4-diphenyloxazole is critical; unlike dihydrooxazoles which might undergo nucleophilic attacks, this aromatic heterocycle provides the necessary electronic and steric environment to facilitate the cycloaddition efficiently. The resulting adduct is an unstable bicyclic intermediate that rapidly rearomatizes. This mechanistic pathway ensures that the carbon skeleton is assembled with high fidelity, minimizing the formation of side products that typically plague radical-based or ionic substitution reactions.
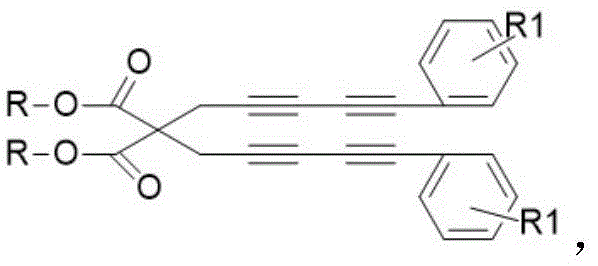
Following the cycloaddition, the reaction mixture undergoes a crucial oxidative aromatization step to yield the final dibenzoylbenzene derivative. Interestingly, the patent highlights that this oxidation does not require the addition of exogenous oxidants like DDQ or manganese dioxide. Instead, the intermediate formed after the DA reaction is susceptible to auto-oxidation by molecular oxygen present in the reaction headspace. As the reaction vessel cools naturally to room temperature, atmospheric oxygen diffuses into the solution and abstracts hydrogen atoms or facilitates electron transfer to establish the final aromatic ketone system. This reliance on ambient air is a masterstroke of green chemistry design, as it removes the cost and hazard associated with handling strong oxidizers. From an impurity control perspective, this mild oxidation profile helps prevent over-oxidation or degradation of sensitive functional groups on the phenyl rings, ensuring a cleaner crude product profile. For quality control laboratories, this means simpler analytical profiles and less time spent identifying and quantifying oxidative byproducts, thereby accelerating the release of batches for downstream processing.
How to Synthesize Polysubstituted Dibenzoylbenzene Derivatives Efficiently
Implementing this synthesis in a production environment requires careful attention to the preparation of the tetrayne precursor, as its purity directly impacts the efficiency of the final cyclization. The precursor itself is accessed via a robust two-step sequence starting from readily available malonates and propargyl bromide, followed by a palladium-catalyzed Sonogashira coupling. Once the tetrayne is secured, the main transformation is operationally simple: mixing the precursor with 2,4-diphenyloxazole in toluene and applying heat. The detailed standardized operating procedures, including precise molar ratios, solvent volumes, and workup protocols required to achieve the reported 77-80% yields, are critical for reproducibility. Manufacturers should note that while the reaction is tolerant to air during the oxidation phase, the initial setup benefits from standard inert atmosphere techniques to ensure the stability of the sensitive alkyne starting materials before thermal activation. The following guide outlines the critical process parameters derived from the patent examples to assist technical teams in replicating this high-value transformation.
- Preparation of Tetrayne Precursor: Synthesize the key tetrayne intermediate via Sonogashira coupling of a malonate-derived alkyne with substituted phenylethynyl bromides using a Pd/Cu catalytic system.
- Thermal Diels-Alder Cyclization: Heat the tetrayne with 2,4-diphenyloxazole in toluene at 100-110°C for approximately 8 hours to generate the benzyne intermediate and effect cycloaddition.
- Oxidative Workup and Purification: Allow the reaction to cool naturally where ambient oxygen facilitates oxidation, followed by aqueous workup and column chromatography to isolate the white solid product.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and supply continuity, the adoption of this synthetic route offers compelling economic and logistical benefits. The primary driver for cost reduction lies in the simplification of the reaction workflow. By consolidating ring construction and functionalization into a single thermal step that utilizes air as the oxidant, the process eliminates the need for expensive stoichiometric reagents and the associated waste disposal costs. Furthermore, the starting materials—malonates, propargyl bromide, and substituted phenylethynyl bromides—are commodity chemicals available from multiple global suppliers, mitigating the risk of single-source dependency. This abundance ensures stable pricing and reliable availability, which is crucial for long-term project planning. The elimination of cryogenic conditions or high-pressure reactors also reduces capital expenditure requirements for manufacturing facilities, allowing existing infrastructure to be utilized with minimal modification. These factors combine to create a manufacturing process that is not only chemically elegant but also commercially resilient against market fluctuations.
- Cost Reduction in Manufacturing: The economic advantages of this process are multifaceted, stemming primarily from the high atom economy and the avoidance of costly reagents. Traditional methods for installing dibenzoyl motifs often require multiple protection-deprotection cycles and expensive Lewis acid catalysts that must be quenched and disposed of safely. In contrast, this thermal method operates with catalytic efficiency in the precursor step and reagent-free efficiency in the final cyclization. The use of toluene, a common and recyclable industrial solvent, further lowers operational expenses compared to specialized fluorinated or chlorinated solvents. Additionally, the spontaneous air oxidation removes the line item for oxidizing agents entirely. When scaled to metric ton quantities, these savings per kilogram compound significantly, offering a distinct competitive advantage in pricing for the final active pharmaceutical ingredient or intermediate. The streamlined purification process, which typically involves standard extraction and chromatography, also reduces labor hours and solvent consumption during downstream processing.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of generic, non-proprietary starting materials that are produced by a wide network of chemical manufacturers globally. Unlike complex chiral building blocks or bespoke reagents that may have long lead times and limited suppliers, the malonates and alkynes required for this synthesis are part of the standard inventory of most fine chemical distributors. This diversity of supply sources protects against disruptions caused by geopolitical issues or production outages at a single vendor. Moreover, the robustness of the thermal reaction conditions means that the process is less sensitive to minor variations in utility supplies, such as cooling water temperature fluctuations, which can sometimes halt more sensitive cryogenic reactions. The ability to run the reaction under ambient pressure and with simple heating requirements ensures that production can be maintained consistently across different manufacturing sites, facilitating a decentralized supply strategy that minimizes logistics risks.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges regarding heat transfer and mixing, but this thermal cyclization is well-suited for scale-up due to its homogeneous nature and moderate temperature requirements. The reaction operates at 100-110°C, a range easily achievable with standard steam or oil heating systems found in most multipurpose plants, avoiding the need for specialized high-temperature equipment. From an environmental perspective, the process aligns well with modern green chemistry principles. The absence of heavy metal catalysts in the final step and the use of air as an oxidant significantly reduce the toxic load of the effluent. While the precursor synthesis uses palladium, established scavenging technologies can recover this valuable metal, minimizing environmental impact and recovering cost. The reduction in hazardous waste generation simplifies regulatory compliance and lowers the fees associated with waste treatment, making the process more sustainable and socially responsible for corporate citizenship goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this polysubstituted dibenzoylbenzene synthesis. These insights are derived directly from the experimental data and mechanistic explanations provided in the patent literature. Understanding these nuances is essential for process chemists and project managers evaluating this technology for integration into their development pipelines. The answers reflect the balance between theoretical potential and practical execution, highlighting both the strengths of the methodology and the operational considerations necessary for success. We encourage technical teams to review these points carefully to ensure alignment with their specific project requirements and quality standards.
Q: What is the primary advantage of the thermal DA route over traditional Friedel-Crafts acylation for these derivatives?
A: The thermal Diels-Alder route described in CN110746305A offers superior regioselectivity and structural complexity control compared to traditional Friedel-Crafts acylation, which often suffers from polyacylation issues and requires stoichiometric Lewis acids that generate significant waste.
Q: Does the oxidation step require specialized oxygen equipment?
A: No, the process is designed for operational simplicity. The patent specifies that the oxidation occurs naturally upon cooling in air, eliminating the need for pressurized oxygen tanks or complex gas handling systems, which significantly reduces equipment costs and safety risks.
Q: What are the typical yields reported for this synthesis?
A: The patent documentation reports column chromatography yields in the range of 77% to 80% for the final cyclization and oxidation step, indicating a robust and efficient transformation suitable for fine chemical production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Dibenzoylbenzene Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic methodologies like the one described in CN110746305A for accelerating drug discovery and development. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries can be seamlessly transitioned into reliable supply chains. We understand that accessing complex intermediates with stringent purity specifications is critical for maintaining the integrity of your clinical trials and commercial launches. Our state-of-the-art rigorous QC labs are equipped to handle the analytical challenges posed by these polysubstituted frameworks, guaranteeing that every batch meets the highest international standards. Whether you require custom synthesis of specific analogs or bulk manufacturing of the core scaffold, our infrastructure is designed to support your growth with agility and precision.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. Our technical procurement team is ready to provide specific COA data and comprehensive route feasibility assessments to help you make informed decisions. Contact us today to discuss how we can optimize your supply chain and reduce your time-to-market with our high-quality polysubstituted dibenzoylbenzene derivatives.
