Advanced Green Synthesis of Fenthiolamide: A Strategic Breakthrough for Herbicide Manufacturing
The global agrochemical industry is currently undergoing a significant paradigm shift towards sustainable manufacturing practices, driven by stringent environmental regulations and the urgent need for carbon neutrality. In this context, the Chinese patent CN111303073A, published on June 19, 2020, presents a groundbreaking methodology for the synthesis of Fenthiolamide, a highly effective herbicide known for its selectivity against grasses in rice paddies. This intellectual property introduces a novel synthetic pathway that leverages carbon dioxide (CO2) as a direct carbonylation reagent to construct the benzothiazolone core, subsequently coupling it with 2-halo-N-methyl-N-phenylacetamide. For R&D directors and procurement strategists seeking a reliable agrochemical intermediate supplier, this technology represents a critical evolution from legacy processes, offering a route that is not only chemically elegant but also industrially robust. By integrating CO2 fixation directly into the synthetic sequence, the method addresses both raw material efficiency and waste reduction, positioning it as a superior alternative for modern herbicide manufacturing supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Fenthiolamide has relied on synthetic routes that are fraught with significant environmental and operational challenges. The traditional 2-chlorobenzothiazole method, for instance, necessitates the condensation of N-methyl-N-phenyl-2-hydroxyacetamide with 2-chlorobenzothiazole. A major drawback of this approach is the synthesis of the 2-chlorobenzothiazole intermediate itself, which typically generates sulfur dioxide (SO2) as a toxic and corrosive byproduct, requiring expensive scrubbing systems and creating substantial disposal liabilities. Alternatively, the benzothiazole ethyl sulfonate method involves complex substitution reactions followed by alkaline desulfonylation. These multi-step sequences involve excessive intermediate handling, leading to cumulative yield losses and increased solvent consumption. Furthermore, the reliance on sulfonate leaving groups introduces additional purification burdens to remove sulfur-containing impurities, which can be detrimental to the quality of the final high-purity agrochemical intermediate. These legacy constraints result in higher operational expenditures and a larger environmental footprint, making them increasingly untenable in a regulatory landscape focused on green chemistry.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN111303073A streamlines the synthesis into a concise, high-efficiency sequence that bypasses the problematic chlorination and sulfonation steps entirely. The core innovation lies in the direct alkylation of a benzothiazolone salt with a halogenated acetamide derivative. This approach utilizes benzothiazolone, which can be sustainably sourced via CO2 fixation from o-aminothiophenol, reacting it with 2-halo-N-methyl-N-phenylacetamide under mild basic conditions. 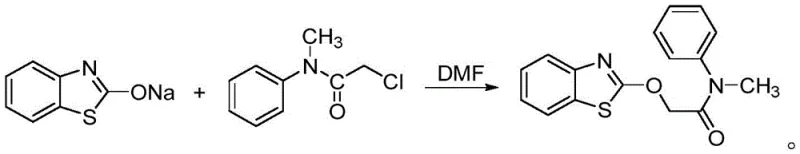 As illustrated in the reaction scheme above, the final coupling step is a straightforward nucleophilic substitution that proceeds with exceptional cleanliness. By eliminating the need for sulfur dioxide-generating steps and reducing the total number of synthetic operations, this novel route drastically simplifies the process flow. This simplification translates directly into cost reduction in herbicide manufacturing, as it lowers energy consumption, reduces solvent waste, and minimizes the requirement for complex downstream purification infrastructure.
As illustrated in the reaction scheme above, the final coupling step is a straightforward nucleophilic substitution that proceeds with exceptional cleanliness. By eliminating the need for sulfur dioxide-generating steps and reducing the total number of synthetic operations, this novel route drastically simplifies the process flow. This simplification translates directly into cost reduction in herbicide manufacturing, as it lowers energy consumption, reduces solvent waste, and minimizes the requirement for complex downstream purification infrastructure.
Mechanistic Insights into Benzothiazolone Alkylation
To fully appreciate the technical superiority of this process, one must examine the mechanistic underpinnings that drive its high efficiency and selectivity. The synthesis begins with the activation of benzothiazolone, a weakly acidic heterocycle, into its corresponding nucleophilic salt. As shown in the reaction below, treating benzothiazolone with a strong base such as sodium hydroxide generates the benzothiazolone sodium salt in situ. 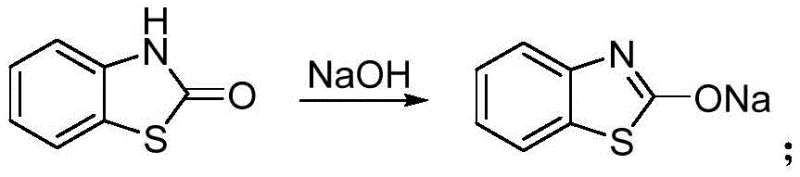 This deprotonation step is critical, as it converts the relatively inert nitrogen atom of the benzothiazolone ring into a potent nucleophile capable of attacking the electrophilic carbon of the acetamide side chain. The subsequent reaction with 2-halo-N-methyl-N-phenylacetamide (where the halogen R is preferably Cl, Br, or I) proceeds via an SN2-type mechanism. The choice of solvent plays a pivotal role here; polar aprotic solvents like DMF (N,N-Dimethylformamide) are identified as optimal because they effectively solvate the cationic sodium species while leaving the anionic benzothiazolone nucleophile "naked" and highly reactive. This solvation effect accelerates the reaction kinetics, allowing the coupling to proceed rapidly at moderate temperatures.
This deprotonation step is critical, as it converts the relatively inert nitrogen atom of the benzothiazolone ring into a potent nucleophile capable of attacking the electrophilic carbon of the acetamide side chain. The subsequent reaction with 2-halo-N-methyl-N-phenylacetamide (where the halogen R is preferably Cl, Br, or I) proceeds via an SN2-type mechanism. The choice of solvent plays a pivotal role here; polar aprotic solvents like DMF (N,N-Dimethylformamide) are identified as optimal because they effectively solvate the cationic sodium species while leaving the anionic benzothiazolone nucleophile "naked" and highly reactive. This solvation effect accelerates the reaction kinetics, allowing the coupling to proceed rapidly at moderate temperatures.
Furthermore, the patent data provides profound insights into impurity control, a key concern for any R&D Director overseeing process validation. Experimental results indicate that temperature control is paramount; while the reaction can technically occur between 40°C and 100°C, operating at approximately 50°C yields the highest purity and conversion rates. Elevated temperatures (e.g., 80-100°C) were observed to promote side reactions, likely due to the thermal instability of the halo-acetamide or competing hydrolysis pathways, leading to a drop in yield from nearly 90% down to roughly 54%. Additionally, the stoichiometry of the reagents is finely tuned, with a molar ratio of benzothiazolone to base to halo-acetamide of approximately 1:1.2:1.1 proving optimal. This slight excess of base ensures complete deprotonation of the benzothiazolone without introducing excessive alkalinity that could degrade the sensitive amide bond in the electrophile. Such precise control over reaction parameters ensures a clean impurity profile, facilitating easier crystallization and reducing the burden on analytical quality control labs.
How to Synthesize Fenthiolamide Efficiently
The practical implementation of this synthesis route is designed for seamless integration into existing fine chemical manufacturing facilities. The process is divided into three logical stages: the preparation of the benzothiazolone core via CO2 fixation, the formation of the reactive salt, and the final coupling and isolation. The use of common industrial solvents like NMP and DMF, combined with standard unit operations such as extraction and recrystallization, ensures that the barrier to adoption is low. For technical teams looking to replicate or scale this chemistry, the following guide outlines the standardized operational protocol derived from the patent examples. Detailed standardized synthesis steps are provided in the guide below.
- Synthesize benzothiazolone by reacting o-aminothiophenol with Na2S·9H2O in NMP solvent under CO2 pressure (5MPa) at 60°C.
- Dissolve the resulting benzothiazolone in an aqueous alkali solution (e.g., NaOH) to form the reactive sodium salt.
- React the benzothiazolone salt with 2-halo-N-methyl-N-phenylacetamide in an organic solvent (preferably DMF) at 50°C, followed by extraction and recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers compelling strategic advantages that extend beyond mere chemical elegance. The primary value proposition lies in the substantial optimization of the cost structure associated with producing high-purity Fenthiolamide. By shortening the synthetic route and eliminating the generation of hazardous sulfur dioxide gas, manufacturers can achieve significant savings in waste treatment and environmental compliance costs. The removal of complex sulfonate intermediates also reduces the inventory of specialized raw materials required, simplifying the procurement portfolio and mitigating supply risks associated with niche reagents. Furthermore, the high yields reported (exceeding 98% under optimal conditions) mean that less raw material is wasted per kilogram of finished product, directly improving the gross margin profile of the manufacturing operation.
- Cost Reduction in Manufacturing: The elimination of toxic byproduct management and the reduction in synthetic steps lead to a leaner production process. Without the need for specialized equipment to handle corrosive SO2 gas or complex desulfonylation reactors, capital expenditure (CapEx) for new production lines is minimized. Additionally, the high atom economy of the direct alkylation step ensures that a greater proportion of input mass is converted into saleable product, driving down the variable cost per unit.
- Enhanced Supply Chain Reliability: The starting materials for this route, specifically o-aminothiophenol and simple halo-acetamides, are commodity chemicals with robust global supply chains. Unlike specialized sulfonate esters which may have limited suppliers, these precursors are readily available from multiple sources, reducing the risk of supply disruption. The mild reaction conditions also allow for flexible scheduling and faster batch turnover times, enabling manufacturers to respond more agilely to market demand fluctuations for this critical herbicide intermediate.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing standard heating and stirring protocols that translate easily from laboratory benchtop to multi-ton reactor vessels. The alignment with green chemistry principles—specifically the utilization of CO2 as a feedstock—positions manufacturers favorably regarding future carbon taxes and environmental regulations. This forward-looking compliance strategy ensures long-term operational continuity and protects the brand reputation of downstream agrochemical companies committed to sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These answers are derived directly from the experimental data and beneficial effects described in patent CN111303073A, providing clarity for stakeholders evaluating the feasibility of this synthesis route for their specific applications.
Q: How does this new synthesis route improve environmental compliance compared to traditional methods?
A: Traditional methods often involve 2-chlorobenzothiazole intermediates which generate toxic sulfur dioxide (SO2) byproducts. This patented route utilizes CO2 as a carbonylation reagent and avoids sulfonate intermediates, significantly reducing hazardous waste generation and aligning with green chemistry principles.
Q: What are the optimal reaction conditions for maximizing yield in this process?
A: According to the experimental data, the optimal reaction temperature is around 50°C. Higher temperatures (e.g., 80-100°C) lead to increased byproduct formation and lower yields. Additionally, using DMF as the solvent and maintaining a specific molar ratio of reactants (approx. 1:1.2:1.1) ensures yields exceeding 98%.
Q: Is this process scalable for industrial production of agrochemical intermediates?
A: Yes, the process features mild reaction conditions (40-100°C range), uses commercially available solvents like DMF and NMP, and eliminates complex purification steps associated with sulfonate removal. These factors make it highly suitable for commercial scale-up of complex agrochemical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fenthiolamide Supplier
As the global demand for efficient and environmentally friendly agrochemicals continues to rise, the ability to manufacture key intermediates like Fenthiolamide via sustainable pathways becomes a distinct competitive advantage. NINGBO INNO PHARMCHEM stands at the forefront of this transition, leveraging advanced process chemistry to deliver superior value to our partners. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this green synthesis are fully realized in large-scale operations. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs, guaranteeing that every batch of Fenthiolamide meets the exacting standards required for modern herbicide formulations.
We invite procurement leaders and technical directors to engage with us to explore how this optimized synthesis route can enhance your supply chain resilience. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in commercial scale-up of complex agrochemical intermediates can drive your business forward.
