Advanced Synthesis and Resolution of Trans-2,3-Diamino-1,2,3,4-Tetrahydronaphthalene for Pharma
Advanced Synthesis and Resolution of Trans-2,3-Diamino-1,2,3,4-Tetrahydronaphthalene for Pharma
The pharmaceutical industry's relentless pursuit of chiral purity has driven significant innovation in the synthesis of key intermediates like trans-2,3-diamino-1,2,3,4-tetrahydronaphthalene. Patent CN100473640C introduces a groundbreaking methodology that addresses the longstanding challenges of low yields, toxic reagents, and complex purification associated with traditional routes. This technology represents a paradigm shift from cumbersome asymmetric synthesis to a highly efficient chiral resolution strategy, leveraging the differential solubility of diastereomeric salts formed with tartaric acid derivatives. For R&D directors and procurement specialists, this patent offers a viable pathway to secure high-purity intermediates essential for next-generation small molecule drugs and chiral ligands. The process not only enhances safety profiles by eliminating hazardous azide handling protocols but also optimizes the economic feasibility of large-scale production through simplified solvent systems.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trans-2,3-diamino-1,2,3,4-tetrahydronaphthalene has been plagued by inefficiencies that hinder commercial scalability and increase operational costs. Traditional literature, such as Tetrahedron: Asymmetry 2001, describes routes relying on dimethylformamide (DMF) mixed with water for the critical azide substitution step. This solvent system notoriously results in poor conversion rates, with yields stagnating between 20% and 40%, necessitating extensive recycling of starting materials and generating significant waste streams. Furthermore, alternative methods employing tetrabutylammonium azide introduce severe safety risks due to the explosive nature of the reagent and require extremely dilute conditions that are impractical for industrial reactors. The reliance on expensive starting materials and multi-step asymmetric syntheses further exacerbates the cost burden, making these conventional pathways unattractive for high-volume manufacturing of pharmaceutical intermediates.
The Novel Approach
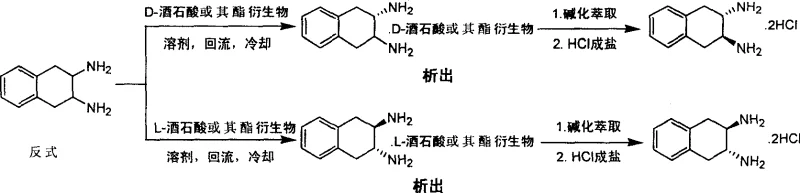
The innovative process detailed in the patent data revolutionizes this landscape by introducing a robust resolution technique coupled with an optimized synthetic route. By substituting the hazardous DMF/water system with a benign alcohol/water mixture, the nucleophilic substitution of the dimesylate with sodium azide achieves a remarkable yield improvement to approximately 60%. This modification not only enhances reaction kinetics but also drastically simplifies the downstream workup, allowing for easier product isolation. The core breakthrough lies in the chiral resolution stage, where readily available D- or L-tartaric acid and their ester derivatives serve as effective resolving agents. This approach enables the direct separation of racemic trans-2,3-diamino-1,2,3,4-tetrahydronaphthalene into its respective (S,S) and (R,R) enantiomers with exceptional optical purity, bypassing the need for complex chiral catalysts.
Mechanistic Insights into Chiral Resolution and Azide Substitution
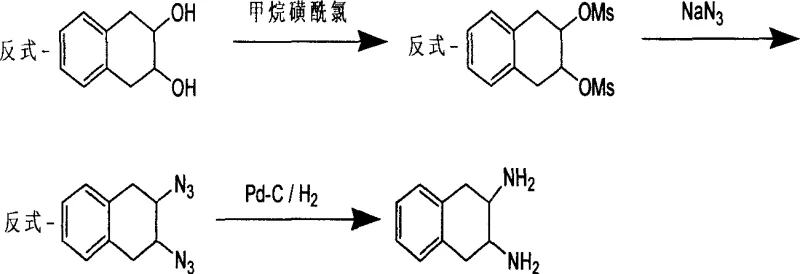
At the heart of this technological advancement is a precise understanding of stereoselective crystallization and nucleophilic substitution mechanisms. The synthesis begins with the activation of trans-1,2,3,4-tetrahydronaphthalene-2,3-diol via mesylation, creating a superior leaving group for the subsequent displacement. In the novel solvent system, the alcohol component likely stabilizes the transition state of the SN2 reaction, facilitating the inversion of configuration required to form the diazide intermediate without the side reactions common in polar aprotic solvents. Following hydrogenation to the racemic diamine, the resolution process exploits the subtle thermodynamic differences in lattice energy between the diastereomeric salts. When the racemate interacts with a single enantiomer of tartaric acid in a heated aqueous-alcoholic medium, one diastereomer preferentially crystallizes out of the solution due to lower solubility, while the other remains in the mother liquor.
This differential solubility is the key to achieving high enantiomeric excess (ee) values exceeding 97% in a single crystallization step. The choice of tartaric acid derivatives, such as diacetoxy or dibenzoyloxy variants, allows for fine-tuning the solubility profile to match specific process requirements, ensuring maximum recovery of the desired isomer. The mother liquor, enriched with the opposite enantiomer, can be recycled by treating it with the counter-enantiomer of the resolving agent, thereby maximizing overall material efficiency. This mechanistic elegance ensures that both (S,S) and (R,R) configurations can be accessed from a single racemic pool, providing a versatile supply chain solution for diverse drug development programs requiring specific stereochemistry.
How to Synthesize Trans-2,3-Diamino-1,2,3,4-Tetrahydronaphthalene Efficiently
Implementing this synthesis requires careful control of reaction parameters to maximize yield and optical purity. The process initiates with the preparation of the dimesylate precursor, followed by the critical azide substitution in an ethanol-water blend under reflux conditions for 40 to 60 hours. Once the diazide is reduced to the amine using palladium on carbon, the resulting racemate undergoes resolution. The detailed standardized synthesis steps, including specific molar ratios, temperature gradients, and crystallization times necessary for GMP compliance, are outlined below for technical reference.
- Convert trans-1,2,3,4-tetrahydronaphthalene-2,3-diol to dimesylate using methanesulfonyl chloride in pyridine.
- Perform nucleophilic substitution with sodium azide in an ethanol/water mixture to form the diazide intermediate.
- Reduce the diazide to racemic diamine via Pd-C catalyzed hydrogenation, followed by chiral resolution with tartaric acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates into tangible strategic benefits regarding cost stability and operational reliability. The elimination of exotic reagents like tetrabutylammonium azide and the shift away from difficult-to-remove solvents like DMF significantly reduces the raw material cost base. Moreover, the simplified workup procedures decrease the consumption of utilities and reduce the volume of hazardous waste requiring disposal, leading to substantial cost savings in manufacturing overheads. The ability to produce both enantiomers from a common racemic intermediate enhances supply chain flexibility, allowing manufacturers to respond dynamically to shifting market demands for specific chiral forms without maintaining separate inventory lines for distinct synthetic routes.
- Cost Reduction in Manufacturing: The transition to alcohol-water solvent systems eliminates the need for expensive polar aprotic solvents and complex distillation recovery units. By improving the yield of the azide substitution step from typical lows of 20-40% to a robust 60%, the process inherently lowers the cost of goods sold (COGS) by reducing the amount of starting material required per kilogram of final product. Additionally, the use of commodity chemicals like tartaric acid as resolving agents avoids the premium pricing associated with specialized chiral catalysts, ensuring long-term price stability for this critical pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: Reliance on widely available bulk chemicals such as sodium azide, methanesulfonyl chloride, and tartaric acid derivatives mitigates the risk of supply disruptions often associated with niche reagents. The robustness of the aqueous-alcoholic resolution process ensures consistent batch-to-batch quality, reducing the likelihood of production delays caused by failed crystallizations or purity issues. This reliability is crucial for maintaining continuous production schedules for downstream API manufacturing, ensuring that lead times for high-purity pharmaceutical intermediates remain predictable and short.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, avoiding the safety hazards of explosive reagents and the toxicity of heavy metal catalysts often found in asymmetric hydrogenation. The use of water and ethanol aligns with green chemistry principles, simplifying regulatory compliance and reducing the environmental footprint of the manufacturing facility. This ease of scale-up facilitates the commercial production of complex pharmaceutical intermediates from pilot plant quantities to multi-ton annual capacities without the need for significant process re-engineering or specialized containment infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this chiral resolution technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on yield expectations, solvent choices, and enantiomeric purity outcomes. Understanding these nuances is essential for process chemists evaluating the feasibility of integrating this route into existing manufacturing workflows.
Q: What is the primary advantage of the new resolution method described in CN100473640C?
A: The primary advantage is the replacement of toxic and expensive reagents with safe, commercially available chiral tartaric acid derivatives, achieving high optical purity (ee > 97%) and improved yields compared to traditional asymmetric synthesis.
Q: How does the solvent system impact the azide substitution step?
A: Switching from DMF/water to alcohol/water mixtures significantly improves the reaction yield from 20-40% to approximately 60%, while simplifying the workup procedure and reducing environmental hazards associated with polar aprotic solvents.
Q: Can both (R,R) and (S,S) enantiomers be produced using this method?
A: Yes, by utilizing either D-tartaric acid or L-tartaric acid (and their derivatives) as resolving agents, the process allows for the selective crystallization and isolation of both (S,S) and (R,R) configurations with high enantiomeric excess.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trans-2,3-Diamino-1,2,3,4-Tetrahydronaphthalene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral intermediates play in the development of life-saving medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated resolution techniques described in CN100473640C are executed with precision and consistency. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced chiral HPLC capabilities to guarantee that every batch of trans-2,3-diamino-1,2,3,4-tetrahydronaphthalene meets the exacting standards required by global regulatory bodies.
We invite you to collaborate with us to leverage this advanced technology for your specific drug development needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your project volume. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your supply chain efficiency and reduce overall development timelines.
