Advanced Metal-Free Synthesis of Trans-3,4-Diaryl Dihydrocoumarins for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust synthetic methodologies for constructing complex heterocyclic scaffolds that serve as critical cores for bioactive molecules. Patent CN113105422B discloses a groundbreaking metal-free strategy for the preparation of trans-3,4-diaryl dihydrocoumarin compounds, a structural motif prevalent in potent anti-breast cancer and anti-osteogenic agents. This innovation addresses a significant gap in organic synthesis by utilizing a Brønsted acid-catalyzed [4+2] cyclization between alkynylamines and o-hydroxybenzyl alcohols. Unlike traditional methods that often struggle with stereocontrol or rely on toxic transition metals, this novel approach leverages the unique localization effect of sulfonamide groups to achieve exceptional regioselectivity and diastereoselectivity. For R&D directors and procurement specialists, this represents a pivotal shift towards greener, more efficient manufacturing pathways for high-value pharmaceutical intermediates.
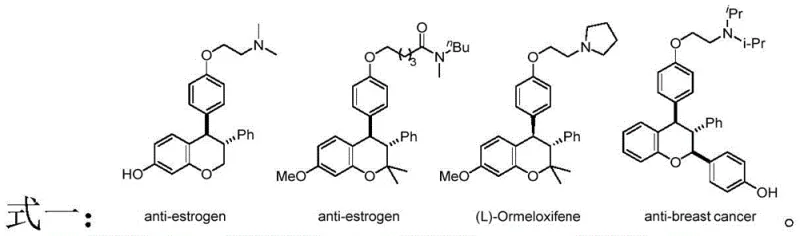
The development of efficient routes to oxygen-containing heterocycles has long been a priority in medicinal chemistry, particularly for coumarin derivatives which exhibit diverse pharmacological properties. The significance of this patent lies not only in the chemical transformation itself but in its potential to streamline the supply chain for critical drug candidates. By establishing a reliable method to access the trans-isomer specifically, manufacturers can reduce the burden of separating difficult diastereomeric mixtures, thereby enhancing overall process efficiency. This technology positions suppliers who adopt it as leaders in providing high-purity pharmaceutical intermediates with superior quality profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3,4-diaryl dihydrocoumarins has relied heavily on transition metal catalysis or harsh Lewis acid conditions, which present substantial drawbacks for modern GMP manufacturing. Conventional protocols often utilize aluminum trichloride or various triflate salts, which necessitate rigorous downstream processing to remove trace metal contaminants that are strictly regulated in final drug substances. Furthermore, existing literature predominantly reports the formation of cis-3,4-diaryl dihydrocoumarin skeletons, leaving the synthesis of the thermodynamically stable trans-isomers largely underexplored and difficult to access directly. The reliance on stoichiometric amounts of activating agents and the generation of significant chemical waste further exacerbate the environmental and economic costs associated with these legacy processes. Additionally, the lack of stereocontrol often results in complex mixtures requiring energy-intensive chromatographic separations, which drastically reduces overall yield and increases production lead times.
The Novel Approach
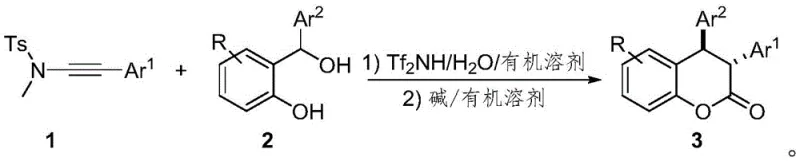
The methodology described in CN113105422B introduces a paradigm shift by employing a metal-free Brønsted acid catalytic system that operates under mild conditions with high atom economy. This novel approach utilizes alkynylamines as bifunctional 2-π partners reacting with in situ generated o-quinone methides from o-hydroxybenzyl alcohols. The key innovation is the use of a sulfonamide moiety as a traceless directing group, which guides the cyclization to occur with high regioselectivity, distinct from previous aluminum-catalyzed reactions. Crucially, the process incorporates a subsequent base-mediated isomerization step that efficiently converts the initial cyclization products into the desired trans-configuration with excellent diastereoselectivity ratios. This two-step one-pot style strategy eliminates the need for expensive transition metal catalysts and simplifies the purification workflow, offering a clear pathway for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Brønsted Acid-Catalyzed Cyclization
The reaction mechanism proceeds through a sophisticated cascade initiated by the activation of the alkynylamine by the strong Brønsted acid catalyst, HNTf2. Upon hydration, the alkynylamine transforms into an amide intermediate which tautomerizes to an enol form, acting as a nucleophilic species. Simultaneously, the o-hydroxybenzyl alcohol undergoes dehydration to generate a reactive o-quinone methide (o-QM) intermediate. The sulfonamide group plays a critical role here, stabilizing the transition state and ensuring that the [4+2] cycloaddition occurs with precise orientation. This mechanistic pathway avoids the random polymerization or side reactions often seen with highly reactive o-QMs, ensuring that the majority of the starting material is channeled towards the desired heterocyclic product. The initial cyclization yields a diastereomeric mixture which serves as the precursor for the final stereochemical adjustment.
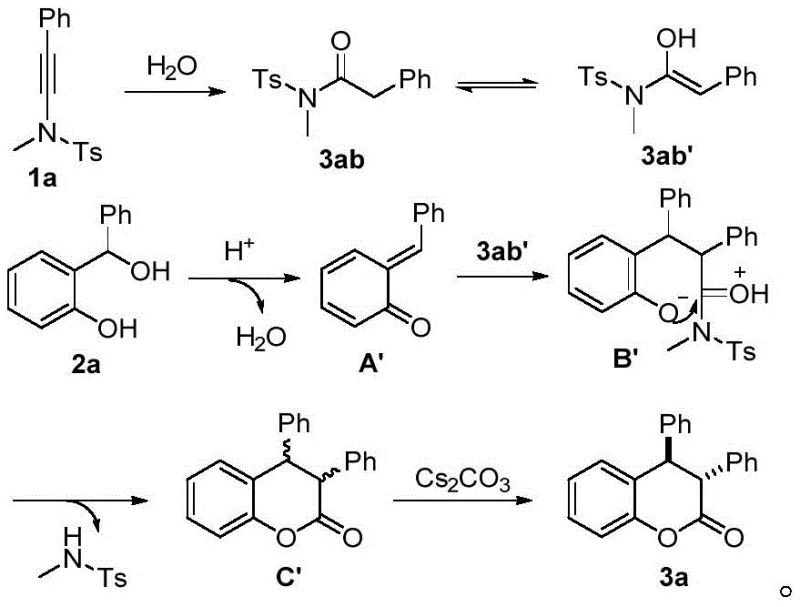
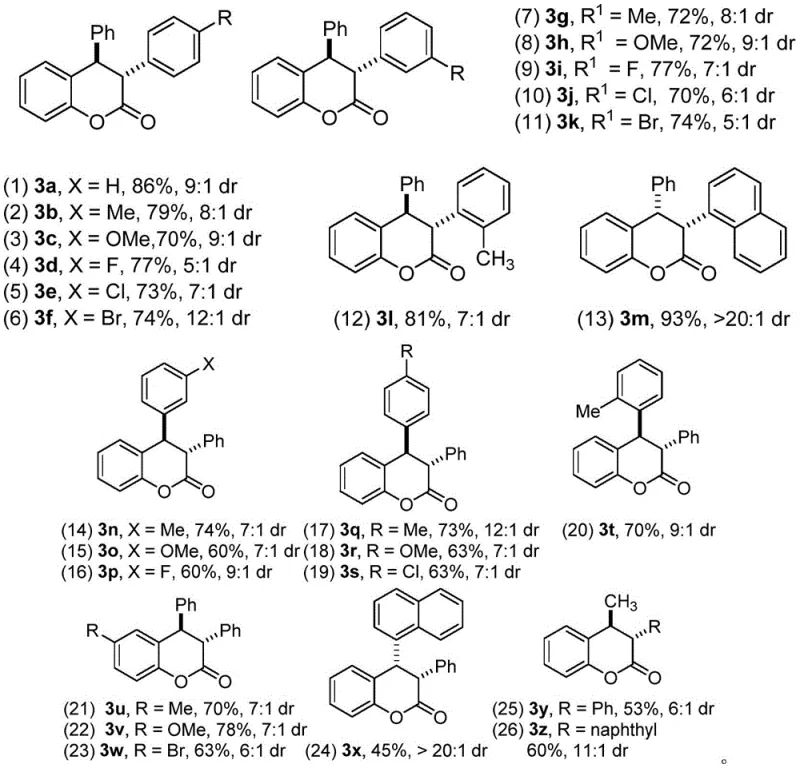
Following the initial cyclization, the addition of a base such as cesium carbonate triggers a crucial isomerization event that drives the equilibrium towards the trans-diastereomer. This step is vital for impurity control, as it effectively suppresses the formation of the cis-isomer which is often the kinetic product in similar systems. The base facilitates the epimerization at the stereocenter adjacent to the carbonyl group, leveraging the thermodynamic stability of the trans-configuration where the bulky aryl groups are positioned anti to each other to minimize steric hindrance. This high level of stereocontrol means that the final crude product contains minimal diastereomeric impurities, significantly reducing the load on purification columns. The broad substrate scope demonstrated in the patent indicates that this mechanism is robust against various electronic and steric variations on the aromatic rings, making it a versatile tool for synthesizing diverse libraries of coumarin analogs.
How to Synthesize Trans-3,4-Diaryl Dihydrocoumarins Efficiently
The synthesis protocol outlined in the patent offers a straightforward operational procedure suitable for both laboratory optimization and pilot plant scale-up. The process begins with the mixing of the alkynylamine and o-hydroxybenzyl alcohol in a chlorinated solvent, followed by the careful addition of the catalyst and water to initiate the cyclization. After monitoring the reaction to completion, the intermediate is isolated and subjected to the isomerization conditions without the need for extensive intermediate purification. Detailed standardized synthesis steps are provided below to ensure reproducibility and safety during implementation.
- Cyclization: React alkyne amine with o-hydroxybenzyl alcohol using HNTf2 catalyst in DCE at 80°C.
- Workup: Concentrate the mixture and purify the intermediate via silica gel column chromatography.
- Isomerization: Treat the intermediate with Cs2CO3 in DCE at room temperature to obtain the trans-product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free synthesis route offers tangible strategic benefits that extend beyond simple yield improvements. The elimination of transition metal catalysts removes a major bottleneck in the supply chain related to the sourcing of expensive and sometimes scarce metal salts. Furthermore, it obviates the need for specialized metal scavenging resins and the associated validation testing required to prove residual metal levels are within ICH guidelines. This simplification of the downstream processing directly translates to reduced manufacturing cycle times and lower operational expenditures. The use of readily available commodity chemicals as starting materials ensures a stable supply base, mitigating risks associated with raw material shortages that often plague complex synthetic routes relying on bespoke reagents.
- Cost Reduction in Manufacturing: The most significant economic driver of this technology is the complete removal of heavy metal catalysts from the process flow. By replacing expensive transition metals with a catalytic amount of Brønsted acid, manufacturers can achieve substantial cost savings on raw material procurement. Additionally, the simplified workup procedure reduces the consumption of silica gel and solvents during purification, as the high selectivity minimizes the presence of hard-to-remove impurities. The avoidance of metal removal steps also lowers utility costs associated with additional filtration and washing stages, contributing to a leaner and more cost-effective production model.
- Enhanced Supply Chain Reliability: The robustness of this synthetic method enhances supply chain resilience by reducing dependency on single-source suppliers for specialized catalysts. The starting materials, alkynylamines and o-hydroxybenzyl alcohols, are structurally simple and can be sourced from multiple global vendors, ensuring continuity of supply even during market fluctuations. The mild reaction conditions also reduce the risk of batch failures due to thermal runaway or sensitivity to moisture, leading to more predictable production schedules. This reliability allows supply chain planners to maintain lower safety stock levels while still meeting delivery commitments to downstream pharmaceutical clients.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this metal-free process aligns perfectly with green chemistry principles and increasingly stringent environmental regulations. The absence of toxic metal waste simplifies effluent treatment and disposal, lowering the environmental compliance burden on manufacturing sites. The process has been demonstrated to work efficiently on gram scales with high yields, indicating strong potential for seamless translation to kilogram and ton-scale commercial production. This scalability ensures that the technology can support the growing demand for coumarin-based therapeutics without requiring significant capital investment in new reactor infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation to assist decision-makers in evaluating feasibility.
Q: What is the primary advantage of this metal-free method over traditional transition metal catalysis?
A: The primary advantage is the elimination of toxic heavy metal residues, which simplifies purification and ensures compliance with strict pharmaceutical impurity guidelines (ICH Q3D).
Q: How does this method achieve high trans-diastereoselectivity?
A: High trans-selectivity is achieved through a specific base-mediated isomerization step using cesium carbonate or DBU after the initial cyclization.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes commercially available reagents, mild reaction conditions, and avoids expensive ligands, making it highly scalable for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trans-3,4-Diaryl Dihydrocoumarin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists has thoroughly analyzed the technology disclosed in CN113105422B and is fully prepared to implement this metal-free route for the commercial production of trans-3,4-diaryl dihydrocoumarins. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our facility is equipped with stringent purity specifications and rigorous QC labs capable of verifying the high diastereoselectivity and low impurity profiles guaranteed by this novel process.
We invite you to collaborate with us to leverage this cutting-edge technology for your next drug development program. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that demonstrate how our optimized manufacturing capabilities can accelerate your timeline to market while reducing overall production costs.
