Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Scalable Drug Manufacturing
The pharmaceutical industry continuously seeks robust methodologies for constructing nitrogen-containing heterocycles, particularly those bearing fluorinated motifs which enhance metabolic stability and bioavailability. A significant breakthrough in this domain is detailed in patent CN113045503A, which discloses a highly efficient preparation method for 2-trifluoromethyl substituted quinazolinone compounds. These scaffolds are ubiquitous in medicinal chemistry, serving as core structures for agents with antifungal, antiviral, and anticancer properties. The disclosed technology leverages a transition metal palladium-catalyzed carbonylation tandem reaction, utilizing readily available trifluoroethylimidoyl chloride and various amines as starting materials. This approach not only circumvents the limitations of classical synthesis routes but also offers exceptional substrate compatibility, allowing for the modular design of diverse derivatives essential for modern drug discovery pipelines.
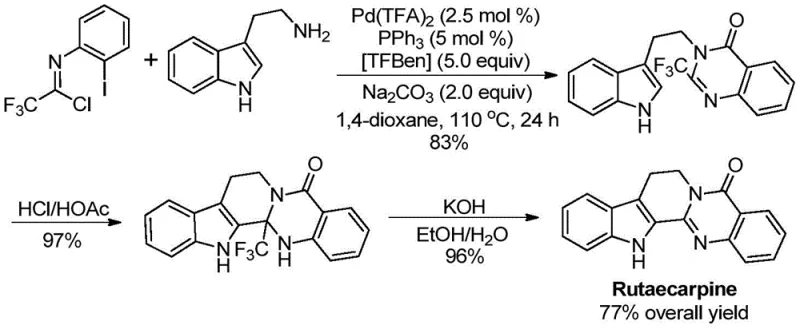
The strategic value of this invention lies in its direct applicability to the synthesis of complex drug molecules. As illustrated in the patent data, the methodology was successfully employed in the total synthesis of Rutaecarpine, a bioactive natural product with potent anti-inflammatory and vasorelaxant activities. The process achieved a remarkable 77% overall yield across three steps, highlighting the efficiency and reliability of the new catalytic system. For R&D directors and process chemists, this represents a viable pathway to access high-value intermediates that were previously difficult to synthesize on a large scale due to cost or safety constraints associated with traditional reagents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-trifluoromethyl substituted quinazolinones has been plagued by significant operational and economic hurdles. Conventional literature reports typically rely on the cyclization of anthranilamide with ethyl trifluoroacetate, trifluoroacetic anhydride, or trifluoroacetic acid under varying conditions. Alternatively, methods involving the cyclization of anthranilic acid esters with unstable trifluoroacetamides or the reaction of isatoic anhydride with trifluoroacetic anhydride have been employed. These traditional pathways suffer from inherent disadvantages, including the requirement for harsh reaction conditions that can degrade sensitive functional groups, the necessity for pre-activation of substrates which adds synthetic steps, and the use of expensive or hazardous reagents. Furthermore, these methods often exhibit low yields and narrow substrate scopes, limiting their utility in the rapid generation of diverse chemical libraries required for lead optimization in pharmaceutical development.
The Novel Approach
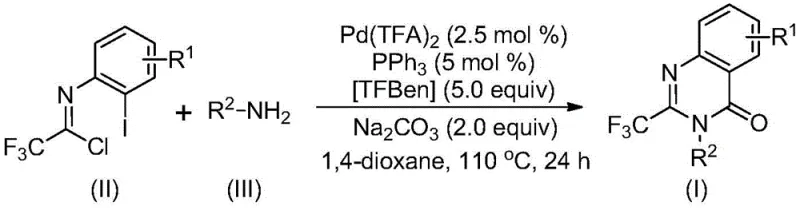
In stark contrast, the novel approach described in the patent utilizes a palladium-catalyzed carbonylation strategy that fundamentally reshapes the synthetic landscape for these heterocycles. By employing cheap and easily obtainable trifluoroethylimidoyl chloride and amines as the primary building blocks, the method eliminates the need for unstable or costly precursors. The reaction proceeds through a sophisticated tandem mechanism where a carbon monoxide substitute, specifically TFBen (1,3,5-tricarboxylic acid phenol ester), releases CO in situ under heating. This allows for the construction of the quinazolinone core under relatively mild conditions (110°C) in a common organic solvent like 1,4-dioxane. The result is a process characterized by high reaction efficiency, broad functional group tolerance, and operational simplicity, making it ideally suited for both laboratory-scale discovery and industrial-scale manufacturing.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cyclization
The mechanistic pathway of this transformation is a testament to the elegance of modern organometallic catalysis. The reaction likely initiates with an alkali-promoted intermolecular carbon-nitrogen bond coupling between the amine and the trifluoroethylimidoyl chloride, generating a trifluoroacetamidine derivative intermediate. Subsequently, the palladium catalyst, generated from palladium trifluoroacetate and triphenylphosphine, undergoes oxidative insertion into the carbon-iodine bond of the aromatic ring to form a divalent palladium intermediate. Concurrently, the thermal decomposition of TFBen releases carbon monoxide, which then inserts into the carbon-palladium bond to create an acyl palladium species. This acyl intermediate then undergoes intramolecular nucleophilic attack by the nitrogen base, promoted by the presence of alkali, to form a seven-membered ring palladium intermediate. The cycle concludes with a reductive elimination step that releases the final 2-trifluoromethyl-substituted quinazolinone product and regenerates the active palladium catalyst for the next turnover.
From an impurity control perspective, this mechanism offers distinct advantages over acid-mediated cyclizations. The use of a defined catalytic cycle minimizes the formation of polymeric byproducts often seen in harsh acidic conditions. The specificity of the palladium insertion and CO insertion steps ensures that the trifluoromethyl group is retained intact at the 2-position without defluorination, a common side reaction in other protocols. Furthermore, the use of sodium carbonate as a mild base helps to neutralize any acidic byproducts generated during the reaction, maintaining a stable pH environment that preserves the integrity of acid-sensitive substituents on the aromatic rings. This precise control over the reaction trajectory results in cleaner crude reaction profiles, significantly reducing the burden on downstream purification processes.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
The execution of this synthesis is designed for reproducibility and ease of handling in standard laboratory or pilot plant settings. The protocol involves charging a reaction vessel with the palladium catalyst system, the carbon monoxide source, the base, and the two primary organic substrates in an aprotic solvent. The mixture is then heated to facilitate the tandem reaction sequence. Detailed below is the standardized procedure derived from the patent examples, which serves as a robust starting point for process optimization.
- Combine palladium trifluoroacetate, triphenylphosphine, sodium carbonate, TFBen, trifluoroethylimidoyl chloride, and amine in an organic solvent like dioxane.
- Heat the reaction mixture to 110°C and stir for 16 to 30 hours to facilitate the carbonylation tandem reaction.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the final 2-trifluoromethyl-substituted quinazolinone compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this patented methodology offers tangible benefits that extend beyond mere chemical yield. The shift towards this catalytic process addresses several critical pain points in the sourcing and manufacturing of complex heterocyclic intermediates. By relying on commodity chemicals rather than specialized, custom-synthesized reagents, the supply chain becomes more resilient and less susceptible to market volatility. The simplification of the synthetic route also translates directly into reduced processing time and lower energy consumption per kilogram of product, aligning with modern sustainability goals and cost-reduction mandates.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the utilization of inexpensive and commercially abundant starting materials. Trifluoroethylimidoyl chloride and various amines are widely available in the global chemical market, eliminating the need for costly, multi-step precursor synthesis. Additionally, the catalytic nature of the reaction means that expensive palladium is used in minute quantities (as low as 2.5 mol %), and the ligand triphenylphosphine is equally affordable. The elimination of harsh reagents like trifluoroacetic anhydride reduces waste disposal costs and the need for specialized corrosion-resistant equipment. Consequently, the overall cost of goods sold (COGS) for these intermediates is significantly optimized compared to legacy methods.
- Enhanced Supply Chain Reliability: Supply continuity is paramount in pharmaceutical manufacturing, and this method enhances reliability by diversifying the raw material base. Since the key reagents are bulk chemicals rather than niche intermediates, the risk of supply disruption due to single-source dependency is minimized. The robustness of the reaction conditions (110°C in dioxane) allows for flexible manufacturing across different facilities without requiring highly specialized infrastructure. This flexibility ensures that production schedules can be maintained consistently, reducing lead times for high-purity pharmaceutical intermediates and enabling just-in-time delivery models for downstream API manufacturers.
- Scalability and Environmental Compliance: The patent data confirms that the reaction is scalable, having been successfully demonstrated at gram levels with high conversion rates. The use of 1,4-dioxane, a common industrial solvent, facilitates easy solvent recovery and recycling, further improving the environmental footprint. Moreover, the high atom economy of the carbonylation process, where the CO source is efficiently incorporated into the product, minimizes the generation of stoichiometric waste. This aligns with green chemistry principles and simplifies regulatory compliance regarding waste discharge, making the process attractive for large-scale commercial production in regions with stringent environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this synthesis technology. These answers are derived directly from the experimental data and specifications provided in the patent documentation, ensuring accuracy for technical decision-makers evaluating this route for their own production needs.
Q: What are the advantages of this Pd-catalyzed method over traditional cyclization routes?
A: Unlike traditional methods requiring harsh conditions or unstable substrates like trifluoroacetic anhydride, this method uses cheap, stable starting materials (trifluoroethylimidoyl chloride) and achieves high yields under moderate conditions with excellent functional group tolerance.
Q: Can this synthesis method be applied to complex drug molecules like Rutaecarpine?
A: Yes, the patent explicitly demonstrates the successful application of this methodology in the high-yield total synthesis of the bioactive alkaloid Rutaecarpine, achieving a 77% overall yield across three steps.
Q: What represents the optimal catalyst system for this transformation?
A: The preferred catalyst system consists of palladium trifluoroacetate (2.5 mol %) and triphenylphosphine (5 mol %) with TFBen as the carbon monoxide substitute and sodium carbonate as the base in 1,4-dioxane.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
The technological advancements presented in patent CN113045503A underscore the potential for high-efficiency manufacturing of critical pharmaceutical building blocks. At NINGBO INNO PHARMCHEM, we recognize the importance of translating such innovative academic and patent research into reliable commercial reality. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with rigorous QC labs and advanced analytical instrumentation to ensure that every batch of 2-trifluoromethyl quinazolinone meets stringent purity specifications required by global regulatory bodies. We are committed to delivering consistent quality and supply security for your most demanding projects.
We invite you to leverage our technical expertise to optimize your supply chain for these valuable intermediates. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and competitive quotations. Let us collaborate to bring your next-generation therapeutic candidates to market faster and more efficiently.
