Scalable Chemical Synthesis of Cochinchinenin B for Advanced Analgesic Development
Introduction to Sustainable Analgesic Intermediate Manufacturing
The pharmaceutical industry is currently witnessing a paradigm shift towards sustainable sourcing of bioactive natural product derivatives, particularly for potent analgesics like Cochinchinenin B. Patent CN102408401B presents a groundbreaking chemical total synthesis method that effectively bypasses the traditional reliance on the extraction of Dracaena cochinchinensis, commonly known as dragon's blood. This innovation is critical because the natural resource is rapidly depleting due to over-harvesting, creating severe supply chain bottlenecks for manufacturers of non-addictive painkillers. By establishing a robust synthetic pathway starting from readily available commodity chemicals like resorcinol, this technology lays a solid foundation for the research, development, and mass production of next-generation analgesics. The route described in the patent not only secures the supply of this high-value homoisoflavan but also ensures a level of purity and consistency that is unattainable through botanical extraction methods.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the procurement of Cochinchinenin B has been entirely dependent on the isolation from the chloroform extract of Dracaena cochinchinensis bark, a process fraught with significant logistical and ecological challenges. The primary limitation lies in the biological variability of the plant source, where the content of the active ingredient fluctuates wildly based on harvest season, geographical location, and plant age, leading to inconsistent batch quality. Furthermore, the extraction process is inherently inefficient, requiring massive quantities of plant biomass to yield minute amounts of the target compound, which drives up costs and exacerbates the endangerment of the species. From a regulatory perspective, botanical extracts often contain complex impurity profiles that are difficult to characterize and remove, posing risks for clinical trial approval and final drug safety. Consequently, relying on this conventional extraction method creates a fragile supply chain that cannot support the demands of large-scale pharmaceutical manufacturing or the development of new non-addicted anodynes.
The Novel Approach
The novel approach detailed in the patent revolutionizes the production landscape by utilizing a fully synthetic organic chemistry route that begins with resorcinol, a cheap and abundant industrial feedstock. This method employs a sequence of well-understood reactions, including Friedel-Crafts acylation, selective methylation, and the sophisticated ELBS oxidation, to construct the complex homoisoflavan skeleton with high precision. Unlike extraction, this chemical synthesis allows for the exact control of stereochemistry and substitution patterns, ensuring that every molecule of the final product is identical, thereby eliminating batch-to-batch variability. The route is designed to be modular, meaning that individual steps can be optimized independently to maximize yield and minimize waste, offering a clear path to cost reduction in pharmaceutical intermediate manufacturing. By decoupling production from agriculture, this approach guarantees a continuous, reliable supply of high-purity Cochinchinenin B regardless of environmental conditions or resource scarcity.
Mechanistic Insights into the Multi-Step Synthetic Pathway
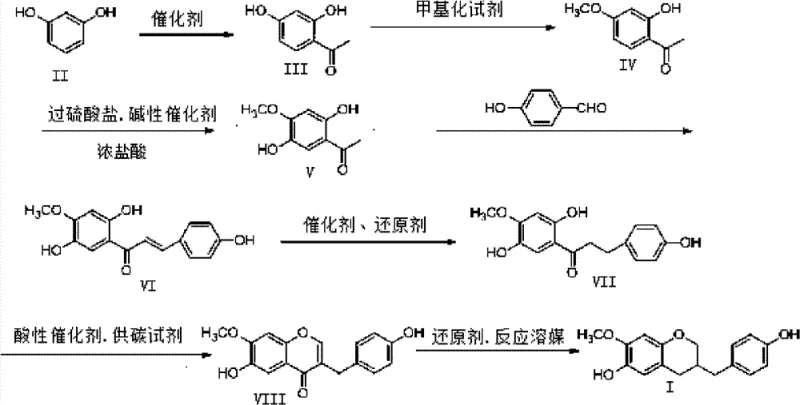
The core of this synthesis lies in the strategic construction of the polyphenolic framework, beginning with a Friedel-Crafts acylation where resorcinol reacts with acetic anhydride or acetyl chloride in the presence of zinc chloride to form 2,4-dihydroxyacetophenone. This initial step is crucial as it establishes the acetophenone core, which serves as the anchor for subsequent functionalizations. Following this, a selective methylation reaction is performed using dimethyl sulfate under basic conditions to protect specific hydroxyl groups, directing the regioselectivity of the subsequent oxidation. The most chemically demanding step is the ELBS (Elbs Persulfate Oxidation) reaction, where potassium persulfate acts as the oxidant to introduce a hydroxyl group at the para-position relative to the existing phenol, generating the critical 2,5-dihydroxy-4-methoxyacetophenone intermediate. This transformation is vital for mimicking the natural substitution pattern found in the dragon's blood extract, and it requires careful temperature control between 20°C and 80°C to prevent over-oxidation or decomposition of the sensitive phenolic structure.
Following the construction of the acetophenone fragment, the synthesis proceeds through an aldol condensation with p-hydroxybenzaldehyde to form a chalcone intermediate, which is subsequently reduced via catalytic hydrogenation using palladium on carbon and ammonium formiate. The final ring closure to form the homoisoflavan skeleton is achieved through a Lewis acid-catalyzed cyclization using boron trifluoride etherate and phosphorus pentachloride, followed by a Clemmensen reduction to remove the carbonyl oxygen. Each of these steps is optimized to minimize the formation of regioisomers and by-products, which is essential for maintaining a clean impurity profile suitable for pharmaceutical applications. The use of common reagents like zinc amalgam for the final reduction step further underscores the practicality of this route, as it avoids the need for expensive or hazardous specialized catalysts that could complicate scale-up and purification processes.
How to Synthesize Cochinchinenin B Efficiently
The synthesis of Cochinchinenin B requires a disciplined approach to reaction monitoring and purification to ensure high yields across the seven distinct chemical transformations. Operators must pay close attention to the stoichiometry of the oxidizing agents in the ELBS step and the acidity levels during the cyclization phase to prevent side reactions. The patent provides specific embodiments detailing solvent choices, such as using glacial acetic acid for acylation and dimethyl sulfoxide for condensation, which are critical for solubility and reaction kinetics. For a comprehensive understanding of the precise operational parameters, including exact temperatures, reaction times, and workup procedures for each intermediate, please refer to the standardized guide below.
- Perform Friedel-Crafts acylation on resorcinol using zinc chloride and acetic acid to form 2,4-dihydroxyacetophenone.
- Execute selective methylation followed by ELBS oxidation using potassium persulfate to install the critical hydroxyl pattern.
- Conduct aldol condensation with p-hydroxybenzaldehyde, followed by hydrogenation, Lewis acid cyclization, and Clemmensen reduction.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from botanical extraction to chemical synthesis represents a fundamental improvement in supply security and cost predictability. The traditional model is vulnerable to agricultural failures, trade restrictions on endangered species, and price volatility driven by scarcity, whereas the synthetic route relies on stable petrochemical feedstocks that are available globally year-round. This shift eliminates the risk of supply interruption due to ecological factors, ensuring that production schedules for downstream analgesic drugs can be maintained without unexpected delays. Furthermore, the ability to produce the intermediate on demand allows for better inventory management and reduces the need for holding large safety stocks of expensive natural extracts, thereby freeing up working capital.
- Cost Reduction in Manufacturing: The synthetic route significantly lowers the cost of goods sold by replacing the expensive and labor-intensive process of plant harvesting and extraction with efficient chemical transformations. By utilizing commodity chemicals like resorcinol and acetic acid, the raw material costs are drastically reduced compared to the procurement of rare dragon's blood resin. Additionally, the elimination of complex chromatographic separations often required to purify natural extracts simplifies the downstream processing, leading to substantial savings in solvent usage and energy consumption. The overall process efficiency is enhanced by the high selectivity of the reactions, which minimizes waste generation and reduces the burden on waste treatment facilities.
- Enhanced Supply Chain Reliability: Adopting this synthetic method insulates the supply chain from the geopolitical and environmental risks associated with sourcing raw materials from specific geographic regions where Dracaena cochinchinensis grows. Manufacturers can source the necessary starting materials from multiple global suppliers, creating a resilient network that is not dependent on a single point of failure. This diversification of the supply base ensures continuity of supply even in the face of regional disruptions, allowing pharmaceutical companies to meet their production commitments reliably. The consistent quality of the synthetic intermediate also reduces the need for extensive incoming quality control testing, speeding up the release of materials for further processing.
- Scalability and Environmental Compliance: The chemistry described in the patent is inherently scalable, utilizing standard reactor types and conditions that are easily transferable from pilot plant to commercial manufacturing scales. The process avoids the use of heavy metals or persistent organic pollutants that would trigger stringent environmental regulations, making it easier to obtain the necessary permits for large-scale production. By moving away from the depletion of natural resources, companies can also align their operations with corporate sustainability goals and ESG (Environmental, Social, and Governance) criteria, enhancing their brand reputation among stakeholders who prioritize eco-friendly manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of Cochinchinenin B synthesized via this patented route. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation to ensure accuracy and relevance for industry professionals. Understanding these details is essential for evaluating the feasibility of integrating this intermediate into your specific drug development pipeline.
Q: Why is chemical synthesis preferred over plant extraction for Cochinchinenin B?
A: Plant extraction relies on Dracaena cochinchinensis, an endangered resource with seasonal variability and low yields. Chemical synthesis ensures consistent supply, higher purity, and independence from ecological constraints.
Q: What are the key challenges in the ELBS oxidation step?
A: The ELBS oxidation requires precise temperature control (20-80°C) and stoichiometry of potassium persulfate to selectively oxidize the phenol to the para-diphenol without over-oxidation or side reactions.
Q: Is this synthesis route scalable for commercial production?
A: Yes, the route utilizes common industrial reagents like zinc chloride, dimethyl sulfate, and palladium on carbon, avoiding exotic catalysts, which facilitates scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cochinchinenin B Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable supply of high-value intermediates like Cochinchinenin B for the development of advanced analgesic therapies. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and speed. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of Cochinchinenin B delivered meets the highest standards required for pharmaceutical applications. We are committed to supporting our partners through every stage of the product lifecycle, from early-stage process optimization to full-scale commercial manufacturing.
We invite you to contact our technical procurement team to discuss how our synthetic capabilities can enhance your supply chain resilience and reduce your overall manufacturing costs. By requesting a Customized Cost-Saving Analysis, you can gain valuable insights into the economic benefits of switching to our synthetic grade Cochinchinenin B. We encourage you to reach out today to obtain specific COA data and route feasibility assessments tailored to your unique project requirements, ensuring a seamless integration of this critical intermediate into your production workflow.
