Advanced Synthetic Route for Agrimonol B and Pseudoaspidinol Intermediates enabling Commercial Scale-up
Introduction to Patent CN113683496A
The pharmaceutical industry constantly seeks robust synthetic pathways for bioactive natural products that are otherwise difficult to source in quantity. Patent CN113683496A, published in late 2021, presents a significant breakthrough in the total synthesis of Agrimonol B, a potent antimalarial and antiparasitic agent derived from the fat-soluble phenol fraction of hairyvein agrimony. Historically, obtaining this compound relied heavily on extraction from natural sources, a process plagued by extremely low content levels and complex purification challenges that restricted both pharmacological research and commercial application. This patent introduces a comprehensive chemical synthesis strategy that not only bypasses the limitations of natural extraction but also optimizes the production of its critical intermediate, Pseudoaspidinol. By leveraging novel protection strategies and streamlined reaction sequences, this technology offers a viable path toward the commercial scale-up of complex phloroglucinol derivatives, ensuring a stable supply chain for downstream drug development.
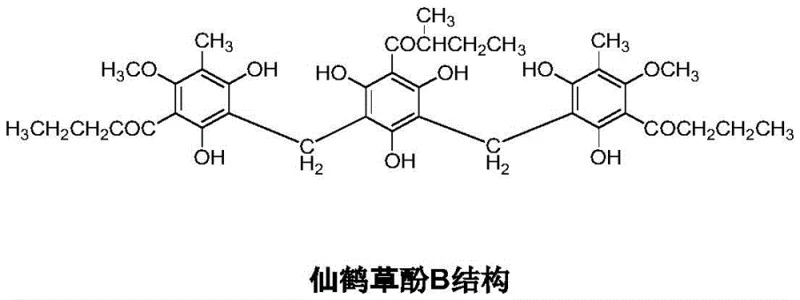
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of Agrimonol B and its intermediates faced substantial hurdles regarding safety, efficiency, and cost. Early literature, such as the work by Liliangquan et al., described routes where the final condensation step suffered from abysmal yields, reported as low as 6%, rendering the process economically unfeasible for industrial adoption. Furthermore, established methods for synthesizing the key intermediate, Pseudoaspidinol, often depended on the use of benzyl chloride to selectively protect phenolic hydroxyl groups. This reagent is not only highly toxic and lachrymatory but also necessitates a subsequent hydrogenolysis step using palladium on carbon, which introduces expensive heavy metal catalysts and complex removal procedures. Other approaches utilizing acyl chlorides for protection resulted in complicated operational workflows with excessive reagent consumption and mediocre overall yields, creating significant bottlenecks for any reliable pharmaceutical intermediate supplier attempting to manufacture these compounds at scale.
The Novel Approach
The methodology disclosed in CN113683496A fundamentally re-engineers the synthetic pathway to overcome these historical inefficiencies. The core innovation lies in the strategic use of sterically hindered protecting reagents, specifically di-tert-butyl dicarbonate (Boc2O), tert-butyldimethylchlorosilane (TBDMS), or triisopropylsilyltrifluoromethanesulfonate (TIPS-OTf). These reagents exhibit excellent reactivity with phenolic hydroxyl groups, allowing for the selective protection of the 4th and 6th positions on the benzene ring of the precursor Compound 3. This shift eliminates the need for toxic benzyl chloride and the associated palladium-catalyzed deprotection, replacing them with a rapid, high-yielding protection and mild acid deprotection sequence. The result is a streamlined process that drastically simplifies purification, reduces the variety of reagents required, and significantly enhances the overall throughput of the synthesis, directly addressing the need for cost reduction in API manufacturing.
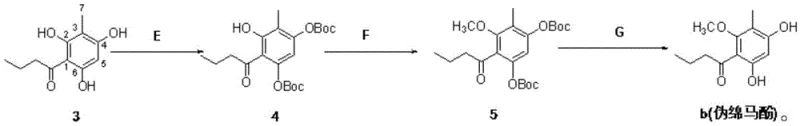
Mechanistic Insights into Selective Phenolic Protection and Acylation
The chemical elegance of this patent is best observed in the transformation of Compound 3 to Compound b (Pseudoaspidinol). In Step E, the reaction utilizes a base such as DMAP or triethylamine to catalyze the nucleophilic attack of the phenolic oxygen on the carbonyl carbon of Boc2O. The steric bulk of the tert-butyl group ensures high regioselectivity, protecting the specific hydroxyls required while leaving others available for subsequent functionalization. This selectivity is crucial for preventing side reactions that would otherwise generate difficult-to-remove impurities. Following protection, Step F employs a methylation agent like dimethyl sulfate under alkaline conditions to install the methoxy group. The final Step G utilizes mild acids, such as hydrochloric acid or trifluoroacetic acid, to cleave the acid-labile Boc group. This deprotection mechanism is clean and efficient, avoiding the harsh conditions often required for ether cleavage, thereby preserving the integrity of the sensitive phloroglucinol core and ensuring the production of high-purity agrimonol B precursors.
Furthermore, the upstream synthesis of the starting materials demonstrates a commitment to green chemistry principles. The conversion of Compound 1 to Compound 2 utilizes a zinc-hydrochloric acid reduction system to transform a carbonyl group into a methyl group. This is a far more economical and atom-efficient alternative to traditional hydride reductions using lithium aluminum hydride or sodium borohydride, which generate significant stoichiometric waste. The subsequent Friedel-Crafts acylation in Step D uses zinc chloride as a Lewis acid catalyst to introduce the butyryl chain. By optimizing the molar ratios and reaction temperatures (controlled between 30-50°C), the process minimizes poly-acylation side products. This rigorous control over reaction parameters ensures a clean impurity profile, which is essential for meeting the stringent quality standards required by global regulatory bodies for pharmaceutical intermediates.
How to Synthesize Pseudoaspidinol Efficiently
The synthesis of Pseudoaspidinol described in this patent represents a optimized balance between reaction speed, yield, and operational simplicity. The process begins with the preparation of the acylated phloroglucinol derivative, followed by the critical protection-methylation-deprotection sequence that defines the novelty of this approach. Operators should note that the choice of solvent in Step E, such as dichloromethane or acetone, plays a vital role in solubilizing the intermediates while maintaining the stability of the protecting groups. The detailed standardized synthetic steps, including specific molar ratios, temperature controls, and workup procedures like column chromatography purification, are outlined below to ensure reproducibility and safety in a laboratory or pilot plant setting.
- Selectively protect the phenolic hydroxyl groups at the 4th and 6th positions of the benzene ring in Compound 3 using a steric protective reagent like di-tert-butyl dicarbonate (Boc2O) in the presence of a base.
- Perform methylation on the protected intermediate (Compound 4) using a methylating agent such as dimethyl sulfate under alkaline conditions to obtain Compound 5.
- Execute acid-catalyzed deprotection of Compound 5 using hydrochloric acid or trifluoroacetic acid to remove the protecting groups and yield the final Pseudoaspidinol (Compound b).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from extraction-based sourcing to this advanced synthetic route offers profound strategic benefits. The reliance on agricultural extraction for Agrimonol B historically introduced volatility into the supply chain, subject to crop yields, seasonal variations, and geographical constraints. By adopting this chemical synthesis method, manufacturers can decouple production from biological variables, ensuring a consistent and predictable output of material. The use of commodity chemicals such as zinc powder, hydrochloric acid, and di-tert-butyl dicarbonate means that raw material sourcing is robust and less susceptible to market fluctuations compared to specialized botanical extracts. This stability is critical for maintaining continuous manufacturing schedules and meeting the delivery commitments of downstream pharmaceutical clients.
- Cost Reduction in Manufacturing: The elimination of toxic benzyl chloride and expensive palladium catalysts directly lowers the bill of materials and waste disposal costs. Traditional methods required costly hydrogenolysis equipment and rigorous heavy metal testing to ensure product safety; this new route bypasses those requirements entirely. Additionally, the high yields reported in the patent examples, such as the 97% yield in the final deprotection step, mean that less raw material is wasted per kilogram of finished product. The mild reaction temperatures (30-50°C) also translate to reduced energy consumption for heating and cooling, further driving down the operational expenditure associated with cost reduction in API manufacturing.
- Enhanced Supply Chain Reliability: By utilizing a synthetic route based on stable, shelf-stable chemical reagents rather than perishable plant matter, the supply chain becomes significantly more resilient. The process avoids the long lead times associated with harvesting and extracting natural products, allowing for reducing lead time for high-purity natural product intermediates. The simplicity of the workup procedures, often involving standard extraction and crystallization rather than complex chromatographic separations at every step, facilitates faster batch turnover. This agility enables suppliers to respond more rapidly to spikes in demand or urgent R&D requirements without compromising on quality or purity specifications.
- Scalability and Environmental Compliance: The synthetic pathway is designed with scalability in mind, utilizing solvents and reagents that are common in large-scale chemical processing. The avoidance of highly toxic reagents like benzyl chloride simplifies environmental, health, and safety (EHS) compliance, reducing the regulatory burden on manufacturing facilities. The waste streams generated are easier to treat, and the overall atom economy of the route is superior to previous iterations. This alignment with green chemistry principles not only mitigates environmental risk but also enhances the corporate sustainability profile of the manufacturer, a key consideration for modern pharmaceutical partnerships focused on responsible sourcing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of Agrimonol B and Pseudoaspidinol. These answers are derived directly from the technical disclosures and experimental data provided in Patent CN113683496A, offering clarity on the feasibility and advantages of this specific manufacturing route. Understanding these details is essential for technical teams evaluating the integration of this intermediate into their broader drug development pipelines.
Q: What are the primary advantages of this new synthesis method over traditional extraction?
A: Traditional extraction from hairyvein agrimony yields very low quantities, severely restricting research and production. This synthetic method provides a scalable, reliable source of high-purity Agrimonol B independent of seasonal plant availability.
Q: How does the new route improve safety compared to prior art?
A: Previous methods relied on toxic benzyl chloride for hydroxyl protection and required palladium carbon hydrogenolysis. This patent utilizes safer steric protecting groups like Boc2O and mild acid deprotection, significantly reducing hazardous waste and operational risk.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the reaction conditions are mild (30-50°C) and utilize economical, readily available reagents. The high yields demonstrated in examples (e.g., 97% in deprotection steps) indicate strong potential for efficient commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Agrimonol B Supplier
The technological advancements detailed in Patent CN113683496A underscore the immense potential of Agrimonol B as a therapeutic agent, yet realizing this potential requires a manufacturing partner with deep technical expertise. NINGBO INNO PHARMCHEM stands ready to support your development needs, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, ensuring that every batch of Agrimonol B or Pseudoaspidinol meets the exacting standards required for clinical and commercial applications. We understand the critical nature of intermediate quality in the final drug product and are committed to delivering consistency and reliability.
We invite you to engage with our technical procurement team to discuss how this optimized synthetic route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this safer, more efficient methodology. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate how our capabilities align with your goals for high-purity agrimonol B sourcing and supply chain optimization.
