Scalable Visible Light Synthesis of Alpha,Beta-Unsaturated Amides for Global Pharma Supply Chains
Introduction: Revolutionizing Amide Synthesis with Green Photonics
The strategic synthesis of alpha,beta-unsaturated amide compounds represents a critical bottleneck in the development of modern pharmaceuticals and agrochemicals. These structural motifs are ubiquitous in high-value active ingredients, serving as key pharmacophores in molecules exhibiting anticonvulsant, antidepressant, and antitumor activities. As illustrated by the diverse bioactive structures in the industry, the demand for efficient access to these scaffolds is relentless. 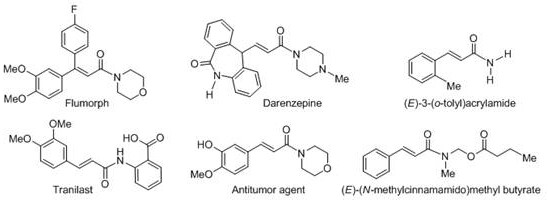 Recent intellectual property developments, specifically patent CN110818584B, have unveiled a transformative approach utilizing visible light catalysis to construct these bonds. This technology shifts the paradigm from harsh thermal conditions to mild, photon-driven chemistry, offering a sustainable pathway for producing reliable pharmaceutical intermediate supplier grades of materials. By leveraging the synergistic effect of organic photocatalysts and hypervalent iodine oxidants, this method bypasses traditional limitations, enabling the direct functionalization of inert C-H bonds with remarkable precision and efficiency.
Recent intellectual property developments, specifically patent CN110818584B, have unveiled a transformative approach utilizing visible light catalysis to construct these bonds. This technology shifts the paradigm from harsh thermal conditions to mild, photon-driven chemistry, offering a sustainable pathway for producing reliable pharmaceutical intermediate supplier grades of materials. By leveraging the synergistic effect of organic photocatalysts and hypervalent iodine oxidants, this method bypasses traditional limitations, enabling the direct functionalization of inert C-H bonds with remarkable precision and efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of alpha,beta-unsaturated amides has relied heavily on the amidation of unsaturated acyl chlorides with amines. This conventional route is fraught with significant operational and economic disadvantages that hinder scalable manufacturing. The prerequisite synthesis of highly reactive acyl chlorides often necessitates the use of corrosive reagents like thionyl chloride or oxalyl chloride, generating substantial hazardous waste streams that complicate environmental compliance. Furthermore, acyl chlorides are moisture-sensitive and thermally unstable, requiring strict anhydrous conditions and low-temperature storage, which escalates infrastructure costs. The multi-step nature of this traditional sequence inherently lowers the overall atom economy and cumulative yield, as each isolation and purification stage introduces material loss. For procurement managers, these inefficiencies translate into volatile pricing and extended lead times, while R&D teams struggle with the difficulty of removing trace acidic byproducts that can degrade sensitive drug candidates. The reliance on such aggressive chemistry also limits the scope of compatible functional groups, often forcing chemists to employ tedious protecting group strategies that further elongate the synthetic timeline.
The Novel Approach
In stark contrast, the visible light-mediated protocol described in the patent offers a streamlined, single-step alternative that directly couples phenylethylene derivatives with formamide compounds.  This innovative strategy operates under exceptionally mild conditions, typically at ambient temperatures ranging from 25°C to 38°C, thereby drastically reducing energy consumption compared to thermal reflux methods. The use of formamides, which serve as both the amide source and often the solvent, eliminates the need for pre-activating the carboxylic acid component, effectively collapsing a multi-step sequence into a single pot operation. The reaction is driven by visible light irradiation (380-780 nm), utilizing inexpensive LED sources that are safer and more energy-efficient than UV lamps. This methodology not only simplifies the operational workflow but also enhances safety profiles by avoiding the handling of hazardous acyl chlorides. For supply chain heads, this translates to a more robust process with fewer unit operations, reduced solvent usage, and a significantly smaller environmental footprint, aligning perfectly with modern green chemistry mandates.
This innovative strategy operates under exceptionally mild conditions, typically at ambient temperatures ranging from 25°C to 38°C, thereby drastically reducing energy consumption compared to thermal reflux methods. The use of formamides, which serve as both the amide source and often the solvent, eliminates the need for pre-activating the carboxylic acid component, effectively collapsing a multi-step sequence into a single pot operation. The reaction is driven by visible light irradiation (380-780 nm), utilizing inexpensive LED sources that are safer and more energy-efficient than UV lamps. This methodology not only simplifies the operational workflow but also enhances safety profiles by avoiding the handling of hazardous acyl chlorides. For supply chain heads, this translates to a more robust process with fewer unit operations, reduced solvent usage, and a significantly smaller environmental footprint, aligning perfectly with modern green chemistry mandates.
Mechanistic Insights into Visible Light Photocatalytic C-H Functionalization
The core of this technological breakthrough lies in the sophisticated interplay between the organic photocatalyst and the hypervalent iodine oxidant under visible light irradiation. The preferred photocatalyst, 1,2,3,4-tetra(carbazol-9-yl)-4,6-dicyanobenzene (often abbreviated as 4CzIPN), is a metal-free organic semiconductor that exhibits excellent redox properties and stability. 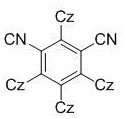 Upon absorption of photons from a blue or white LED source, the catalyst enters an excited state capable of engaging in single-electron transfer (SET) processes. This photo-excited species interacts with the hypervalent iodine oxidant, such as trifluoroacetic acid-(2,4,6-trimethoxyphenyl)iodo(III)benzene, to generate reactive radical intermediates.
Upon absorption of photons from a blue or white LED source, the catalyst enters an excited state capable of engaging in single-electron transfer (SET) processes. This photo-excited species interacts with the hypervalent iodine oxidant, such as trifluoroacetic acid-(2,4,6-trimethoxyphenyl)iodo(III)benzene, to generate reactive radical intermediates. 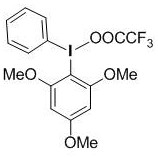 The oxidant plays a dual role: it regenerates the ground state of the photocatalyst to close the catalytic cycle and simultaneously activates the formamide substrate via hydrogen atom abstraction. This generates an alpha-amino alkyl radical which subsequently adds to the electron-deficient double bond of the phenylethylene substrate. The resulting benzylic radical undergoes further oxidation and deprotonation to restore aromaticity and establish the conjugated alpha,beta-unsaturated system. This mechanistic pathway avoids the use of toxic transition metals, ensuring that the final product is free from heavy metal contamination, a critical specification for API intermediates.
The oxidant plays a dual role: it regenerates the ground state of the photocatalyst to close the catalytic cycle and simultaneously activates the formamide substrate via hydrogen atom abstraction. This generates an alpha-amino alkyl radical which subsequently adds to the electron-deficient double bond of the phenylethylene substrate. The resulting benzylic radical undergoes further oxidation and deprotonation to restore aromaticity and establish the conjugated alpha,beta-unsaturated system. This mechanistic pathway avoids the use of toxic transition metals, ensuring that the final product is free from heavy metal contamination, a critical specification for API intermediates.
From an impurity control perspective, this radical mechanism offers distinct advantages over ionic pathways. The mildness of the reaction conditions suppresses common side reactions such as polymerization of the styrene substrate or hydrolysis of the amide bond, which are prevalent in acidic or basic thermal methods. The selectivity is governed by the redox potentials of the catalyst and oxidant, ensuring that only the specific C-H bond adjacent to the nitrogen is activated. This high chemoselectivity minimizes the formation of regioisomers and over-oxidized byproducts, simplifying the downstream purification process. For quality assurance teams, this means a cleaner crude profile and higher recovery rates during crystallization or chromatography. The ability to tune the reaction by selecting different organic dyes (like Eosin Y or Rose Bengal) or iodine reagents provides a versatile toolkit for optimizing the impurity profile for specific complex molecules, ensuring consistent batch-to-batch reproducibility essential for commercial validation.
How to Synthesize Alpha,Beta-Unsaturated Amides Efficiently
Implementing this visible light technology requires careful attention to reactor design and light penetration to ensure uniform irradiation of the reaction mixture. The process begins by charging a dry reactor with the phenylethylene substrate, the formamide reagent, the organic photocatalyst, and the hypervalent iodine oxidant under an inert nitrogen atmosphere to prevent oxygen quenching of the excited states. The detailed standardized synthesis steps are outlined below to guide process engineers in replicating the high yields reported in the patent literature. Proper scaling involves maintaining the surface-area-to-volume ratio or using flow chemistry reactors to ensure that all molecules receive adequate photon flux, which is the key driver of the reaction kinetics.
- Mix phenylethylene substrate, formamide reagent, organic photocatalyst (e.g., 4CzIPN), and hypervalent iodine oxidant in a reactor under nitrogen atmosphere.
- Irradiate the reaction mixture with visible light (blue or white LED, 10-50W) at mild temperatures (25-38°C) until starting material is consumed.
- Quench with water, extract with organic solvent (e.g., ethyl acetate), dry over sodium sulfate, and purify via column chromatography to isolate the target amide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this visible light catalytic method presents a compelling value proposition centered on cost optimization and risk mitigation. The elimination of transition metal catalysts removes a significant cost center associated with purchasing expensive palladium or rhodium complexes, as well as the downstream costs of scavenging residual metals to meet strict regulatory limits. Furthermore, the use of formamides as both reagent and solvent reduces the volume of auxiliary solvents required, leading to substantial savings in raw material procurement and waste disposal fees. The mild operating temperatures reduce the energy load on HVAC and cooling systems, contributing to lower utility costs per kilogram of product manufactured.
- Cost Reduction in Manufacturing: The transition from multi-step acyl chloride chemistry to a direct one-pot coupling significantly reduces labor hours and equipment occupancy time. By avoiding the generation of stoichiometric amounts of corrosive salt waste, the process lowers the burden on wastewater treatment facilities. The high atom economy of using formamide directly means less raw material is wasted as byproducts, directly improving the cost of goods sold (COGS). Additionally, the longevity and stability of the organic photocatalysts allow for potential recycling or lower loading factors, further driving down material costs without compromising reaction efficiency.
- Enhanced Supply Chain Reliability: The raw materials for this process, including substituted styrenes and dimethylformamide, are commodity chemicals available from multiple global suppliers, reducing the risk of single-source bottlenecks. The robustness of the reaction conditions means that production is less susceptible to disruptions caused by utility fluctuations, as it does not require extreme cryogenic cooling or high-pressure steam. This stability ensures consistent delivery schedules for downstream API manufacturers. The simplified workflow also reduces the likelihood of batch failures due to operator error, enhancing the overall reliability of the supply chain for critical pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling photochemical reactions has historically been challenging, but advancements in LED technology and flow reactor design have made kilogram-to-ton scale production feasible. This method aligns with increasingly stringent environmental regulations by minimizing the use of hazardous reagents like thionyl chloride. The absence of heavy metals simplifies the environmental impact assessment and permitting process for new manufacturing lines. The reduced waste generation and lower energy consumption contribute to a lower carbon footprint, supporting corporate sustainability goals and enhancing the marketability of the final drug product as 'green' or 'sustainably sourced'.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this visible light synthesis technology. These answers are derived directly from the experimental data and scope defined in the patent documentation to provide clarity for technical stakeholders. Understanding these nuances is essential for evaluating the feasibility of integrating this method into existing production portfolios.
Q: Does this visible light method require expensive transition metal catalysts?
A: No, the patented process utilizes organic photocatalysts such as 1,2,3,4-tetra(carbazol-9-yl)-4,6-dicyanobenzene or eosin dyes, completely eliminating the need for costly transition metals like palladium or ruthenium, which simplifies purification and reduces heavy metal residue risks.
Q: What are the typical reaction conditions for this synthesis?
A: The reaction proceeds under mild conditions, typically at temperatures between 25°C and 38°C under a nitrogen atmosphere, using standard blue or white LED light sources (10-50W), making it highly energy-efficient and safe for large-scale operations.
Q: Can this method tolerate diverse functional groups on the substrate?
A: Yes, the method demonstrates strong substrate applicability, successfully tolerating various substituents including halogens, alkoxy groups, nitriles, and heteroaryl rings, allowing for the synthesis of a wide library of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha,Beta-Unsaturated Amide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of visible light catalysis in modernizing the synthesis of complex organic molecules. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our facility is equipped with state-of-the-art photochemical reactors and stringent purity specifications are maintained through our rigorous QC labs, guaranteeing that every batch of alpha,beta-unsaturated amide meets the highest international standards. We are committed to helping our partners navigate the transition from traditional chemistry to greener, more efficient photoredox methodologies.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific molecule. We are ready to provide specific COA data and route feasibility assessments to demonstrate how this visible light method can optimize your supply chain and reduce your overall manufacturing costs while delivering high-purity pharmaceutical intermediates.
