Advanced Palladium-Catalyzed Carbonylation for Scalable 2,3-Dihydroquinolone Production
Advanced Palladium-Catalyzed Carbonylation for Scalable 2,3-Dihydroquinolone Production
The development of efficient synthetic routes for nitrogen-containing heterocycles remains a cornerstone of modern medicinal chemistry, particularly for scaffolds exhibiting potent biological activities. The 2,3-dihydroquinolone core is a privileged structure found in numerous bioactive molecules, including significant antitumor agents and analgesics, as illustrated by the structural diversity shown in early pharmacological studies. 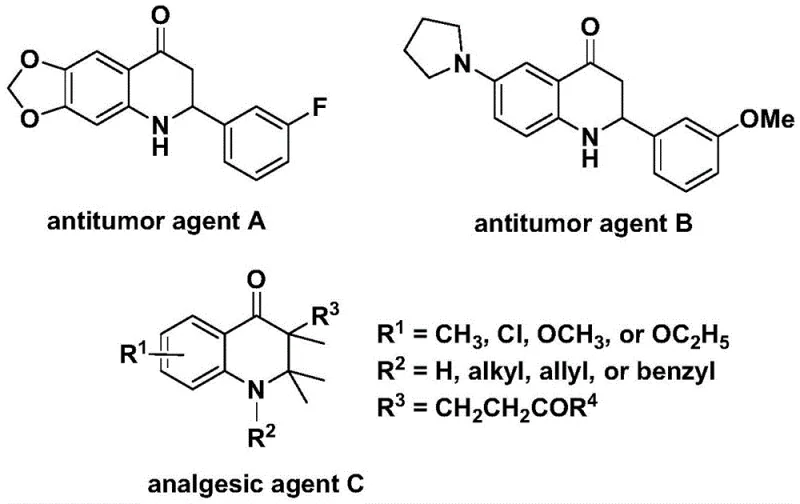 . Addressing the critical need for robust manufacturing processes, Patent CN112239456B discloses a groundbreaking preparation method for substituted 2,3-dihydroquinolone compounds. This technology leverages a transition metal palladium-catalyzed carbonylation reaction, utilizing N-pyridine sulfonyl-o-iodoaniline and olefins as primary starting materials. By replacing toxic carbon monoxide gas with safer solid substitutes and employing mild reaction conditions, this innovation offers a transformative approach for reliable pharmaceutical intermediate supplier networks seeking to optimize their production pipelines for high-value heterocyclic building blocks.
. Addressing the critical need for robust manufacturing processes, Patent CN112239456B discloses a groundbreaking preparation method for substituted 2,3-dihydroquinolone compounds. This technology leverages a transition metal palladium-catalyzed carbonylation reaction, utilizing N-pyridine sulfonyl-o-iodoaniline and olefins as primary starting materials. By replacing toxic carbon monoxide gas with safer solid substitutes and employing mild reaction conditions, this innovation offers a transformative approach for reliable pharmaceutical intermediate supplier networks seeking to optimize their production pipelines for high-value heterocyclic building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 2,3-dihydroquinolone skeleton has relied on multi-step sequences that often suffer from poor atom economy and limited functional group tolerance. Traditional Friedel-Crafts acylations or condensation reactions frequently require harsh acidic conditions, elevated temperatures, and stoichiometric amounts of Lewis acids, which generate substantial waste streams and complicate downstream purification. Furthermore, many existing carbonylation strategies depend on the direct use of gaseous carbon monoxide, posing severe safety hazards and requiring specialized high-pressure equipment that is not universally available in standard pilot plants. These operational complexities often lead to inconsistent batch-to-batch quality and restrict the ability to introduce diverse substituents at the C2 and C3 positions, thereby limiting the chemical space accessible for drug discovery campaigns targeting this specific scaffold.
The Novel Approach
In stark contrast, the methodology outlined in CN112239456B introduces a streamlined, one-pot carbonylative cyclization that dramatically simplifies the synthetic landscape. By employing 1,3,5-trimesic acid phenol ester as a safe and solid carbon monoxide surrogate, the process eliminates the need for handling hazardous gases while maintaining high reaction efficiency. The use of a palladium catalyst system, specifically bis(acetylacetone)palladium paired with a diphosphine ligand like dppp, enables the activation of the carbon-iodine bond under relatively mild thermal conditions ranging from 100 °C to 120 °C. This novel approach not only enhances safety profiles but also significantly broadens the substrate scope, allowing for the seamless integration of various aryl, alkyl, and silyl-substituted olefins. 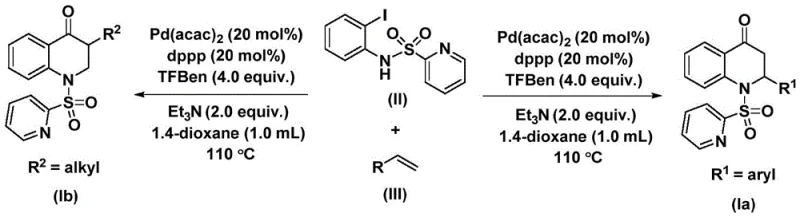 . The result is a versatile platform capable of cost reduction in API manufacturing by reducing step counts and minimizing waste generation associated with traditional heterocycle synthesis.
. The result is a versatile platform capable of cost reduction in API manufacturing by reducing step counts and minimizing waste generation associated with traditional heterocycle synthesis.
Mechanistic Insights into Palladium-Catalyzed Carbonylative Cyclization
The catalytic cycle driving this transformation is a sophisticated interplay of organometallic steps that ensure high regioselectivity and yield. Initially, the active palladium(0) species undergoes oxidative addition into the carbon-iodine bond of the N-pyridine sulfonyl-o-iodoaniline substrate, generating a key aryl-palladium(II) intermediate. This step is crucial as it activates the aromatic ring for subsequent functionalization. Following this activation, the carbon monoxide molecule, released in situ from the thermal decomposition of the mesityl ester surrogate, inserts into the palladium-carbon bond to form an acyl-palladium species. This acyl intermediate is highly reactive and serves as the electrophilic center for the next stage of the cycle. The coordination and subsequent migratory insertion of the olefin into the acyl-palladium bond create a new carbon-carbon bond, establishing the core framework of the dihydroquinolone ring system with precise stereochemical control dictated by the ligand environment.
Following the olefin insertion, the resulting alkyl-palladium intermediate undergoes an intramolecular nucleophilic attack or reductive elimination sequence to close the six-membered ring, releasing the final substituted 2,3-dihydroquinolone product and regenerating the palladium catalyst. This mechanism is particularly robust because the pyridine sulfonyl group acts not only as a protecting group for the nitrogen but also potentially assists in stabilizing intermediates through coordination. The tolerance for diverse R groups, as evidenced by the successful synthesis of compounds I-1 through I-5 with yields ranging from 59% to 88%, underscores the resilience of this catalytic system against steric and electronic variations. 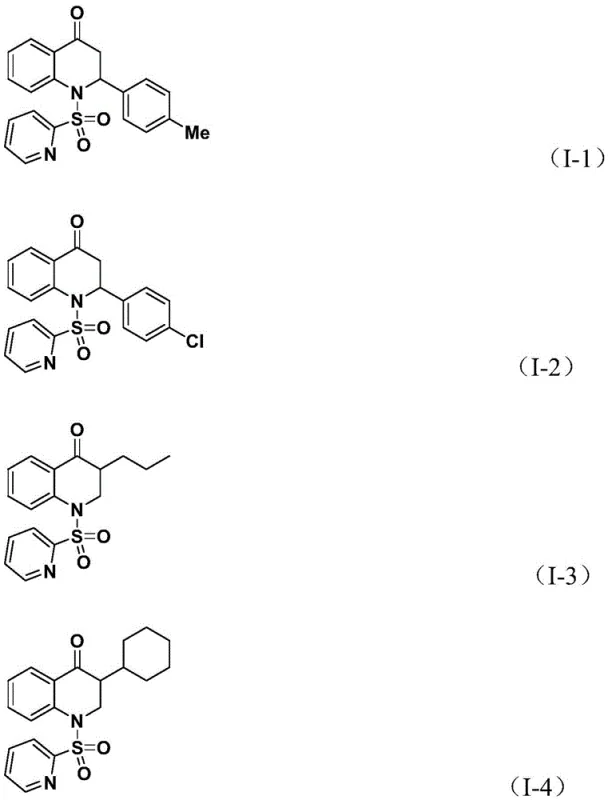 . Such mechanistic clarity allows R&D teams to predict impurity profiles accurately and design analogues with confidence, knowing that the catalytic cycle can accommodate functionalities like halogens, ethers, and even silyl groups without deactivation.
. Such mechanistic clarity allows R&D teams to predict impurity profiles accurately and design analogues with confidence, knowing that the catalytic cycle can accommodate functionalities like halogens, ethers, and even silyl groups without deactivation.
How to Synthesize Substituted 2,3-Dihydroquinolone Efficiently
Implementing this synthesis in a laboratory or pilot setting requires careful attention to reagent ratios and reaction parameters to maximize conversion. The protocol dictates the use of a polar aprotic solvent such as 1,4-dioxane, which effectively dissolves both the organic substrates and the inorganic additives while promoting the catalytic turnover. Triethylamine is added as a base to neutralize acidic byproducts and facilitate the reaction progress. The detailed standardized synthesis steps below outline the precise molar equivalents and thermal profiles required to achieve the high yields reported in the patent data, ensuring reproducibility for process chemists aiming to scale this technology.
- Combine N-pyridine sulfonyl-o-iodoaniline, olefin, palladium catalyst, ligand, and carbon monoxide substitute in an organic solvent.
- Heat the reaction mixture to 100-120 °C and stir for 24-48 hours to facilitate the carbonylation cyclization.
- Filter the mixture, adsorb onto silica gel, and purify via column chromatography to isolate the final 2,3-dihydroquinolone product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this technology offers substantial strategic benefits by leveraging commodity chemicals as feedstocks. The primary starting materials, including various substituted olefins and o-iodoanilines, are widely available from global chemical suppliers, reducing the risk of supply chain bottlenecks often associated with exotic or custom-synthesized reagents. The substitution of gaseous carbon monoxide with a solid ester derivative simplifies logistics and storage requirements, eliminating the need for specialized gas cylinders and monitoring systems. This shift translates directly into lower operational overheads and enhanced safety compliance, making the process attractive for facilities with strict environmental and safety regulations. Furthermore, the high substrate compatibility means that a single manufacturing line can be adapted to produce a wide library of derivatives simply by swapping the olefin input, providing immense flexibility for contract manufacturing organizations.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal scavengers typically required for removing residual palladium is a significant cost driver here, as the catalyst loading is optimized to 20 mol% but can likely be reduced further upon process optimization. Additionally, the use of a solid CO surrogate avoids the capital expenditure associated with high-pressure autoclaves needed for gas-phase carbonylations. The simplified workup procedure, involving basic filtration and standard column chromatography, reduces solvent consumption and labor hours compared to multi-step traditional syntheses. These factors collectively contribute to a more economical production cost per kilogram, enhancing the margin potential for high-purity pharmaceutical intermediates destined for clinical or commercial use.
- Enhanced Supply Chain Reliability: By relying on robust, commercially available catalysts like Pd(acac)2 and ligands like dppp, the process mitigates the risk of delays caused by custom reagent synthesis. The reaction conditions are moderate (110 °C), which reduces energy consumption and wear on reactor vessels compared to high-temperature pyrolysis methods. The ability to synthesize both 2-aryl and 3-alkyl substituted variants from a common intermediate streamlines inventory management, allowing manufacturers to respond rapidly to changing market demands for specific analogues. This agility is crucial for maintaining continuity in the supply of critical API precursors, ensuring that downstream drug production schedules are not disrupted by raw material shortages.
- Scalability and Environmental Compliance: The protocol has been demonstrated to be effective on a gram scale with clear pathways for expansion to multi-kilogram batches, supported by the use of common solvents like dioxane that are easily recovered and recycled. The absence of toxic gas emissions and the generation of benign byproducts align with green chemistry principles, facilitating easier regulatory approval for new manufacturing sites. The high conversion rates observed across diverse substrates minimize the formation of difficult-to-separate side products, thereby reducing the volume of hazardous waste requiring disposal. This environmental efficiency not only lowers disposal costs but also strengthens the sustainability profile of the supply chain, a key metric for modern pharmaceutical procurement strategies.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented synthesis method. These answers are derived directly from the experimental data and mechanistic descriptions provided in the patent documentation, offering clarity for technical teams evaluating this route for adoption. Understanding these nuances is essential for assessing the feasibility of integrating this chemistry into existing production workflows.
Q: What are the key advantages of this carbonylation method over traditional synthesis?
A: This method utilizes readily available olefins and avoids harsh conditions, offering broader substrate compatibility and higher reaction efficiency compared to conventional routes.
Q: Can this process be scaled for industrial production of API intermediates?
A: Yes, the patent explicitly demonstrates gram-level scalability and uses common solvents like dioxane, making it highly suitable for commercial scale-up of complex pharmaceutical intermediates.
Q: What types of substituents are tolerated in this reaction?
A: The reaction shows excellent tolerance for various functional groups including alkyl, aryl, silyl, halogen, and alkoxy substituents on both the aniline and olefin components.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3-Dihydroquinolone Supplier
The technological advancements presented in CN112239456B represent a significant leap forward in the efficient construction of complex heterocyclic scaffolds essential for modern drug discovery. NINGBO INNO PHARMCHEM stands at the forefront of translating such innovative academic and patent literature into practical, industrial-scale reality. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to full-scale manufacturing is seamless and compliant. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 2,3-dihydroquinolone intermediate meets the exacting standards required by global regulatory bodies.
We invite procurement leaders and R&D directors to collaborate with us to leverage this advanced carbonylation technology for your specific project needs. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your volume requirements and timeline constraints. We encourage you to reach out today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary targets, ensuring a secure and efficient supply chain for your critical pharmaceutical intermediates.
