Advanced Synthesis of Oxaspiro Diphosphine Ligands for High-Enantioselectivity Catalysis
The landscape of asymmetric catalysis continues to evolve with the introduction of novel chiral ligands that offer superior stereocontrol and operational simplicity. Patent CN110128471B discloses a groundbreaking class of oxaspiro diphosphine ligands that address critical limitations in existing bisphosphine systems. These compounds feature a rigid spiro-benzofuran backbone that imparts unique steric and electronic properties, making them highly effective for asymmetric transformations. As a leading manufacturer, we recognize the immense potential of this technology for producing high-value chiral intermediates required in modern drug discovery. The ability to synthesize these ligands from readily available racemic spiro diphenols represents a significant advancement in process chemistry, offering a streamlined pathway to optically pure catalysts.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for synthesizing chiral bisphosphine ligands often rely on complex resolution processes or expensive chiral pool starting materials that limit scalability. Many established ligands suffer from conformational flexibility that can lead to reduced enantioselectivity under demanding reaction conditions. Furthermore, the introduction of phosphorus atoms onto rigid scaffolds frequently requires harsh conditions or multiple protection-deprotection sequences that lower overall yield and increase waste generation. In industrial settings, the reliance on precious metal catalysts without efficient recovery protocols can drastically inflate the cost of goods sold. These factors collectively create bottlenecks for procurement teams seeking reliable sources of high-purity chiral catalysts for large-scale API manufacturing.
The Novel Approach
The patented methodology introduces a highly efficient stepwise strategy that overcomes these historical barriers through a clever combination of triflation, palladium-catalyzed coupling, and silane reduction. This approach allows for the modular installation of diverse aryl phosphine groups, enabling fine-tuning of the ligand's electronic environment without altering the core chiral scaffold. 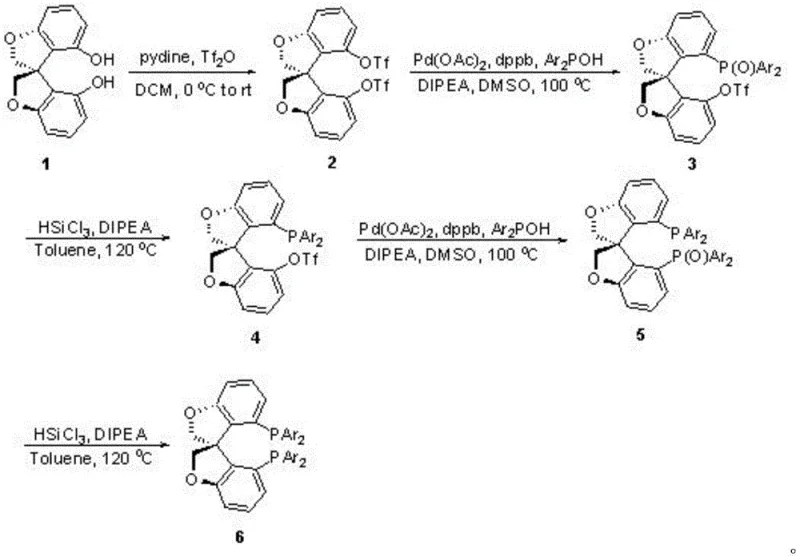 By utilizing a bis-triflate intermediate, the synthesis ensures high reactivity during the subsequent phosphorylation steps, leading to excellent conversion rates. The final reduction step using trichlorosilane is operationally simple and avoids the use of hazardous reducing agents often associated with phosphine oxide reduction. This novel route significantly simplifies the supply chain for complex chiral ligands by reducing the number of isolation steps and improving overall process robustness.
By utilizing a bis-triflate intermediate, the synthesis ensures high reactivity during the subsequent phosphorylation steps, leading to excellent conversion rates. The final reduction step using trichlorosilane is operationally simple and avoids the use of hazardous reducing agents often associated with phosphine oxide reduction. This novel route significantly simplifies the supply chain for complex chiral ligands by reducing the number of isolation steps and improving overall process robustness.
Mechanistic Insights into Ruthenium-Catalyzed Asymmetric Hydrogenation
The core value of these oxaspiro diphosphine ligands lies in their ability to form stable and highly active ruthenium complexes capable of inducing exceptional chirality. When coordinated with ruthenium precursors such as [Ru(p-cymene)Cl2]2 or bis-(2-methylallyl)cycloocta-1,5-dienylruthenium, the ligand creates a well-defined chiral pocket around the metal center. This precise spatial arrangement is critical for differentiating between the enantiotopic faces of the substrate during the hydrogenation cycle. The rigid spiro structure prevents unfavorable conformational changes that could otherwise lead to racemic background reactions. Mechanistic studies suggest that the oxygen atoms in the benzofuran rings may also participate in secondary interactions that stabilize the transition state, further enhancing stereoinduction.
In practical applications, this catalyst system has demonstrated remarkable performance in the asymmetric hydrogenation of unsaturated carboxylic acids. For instance, the hydrogenation of 2-methyl cinnamic acid proceeds with complete conversion and an enantiomeric excess exceeding 99 percent under mild conditions. 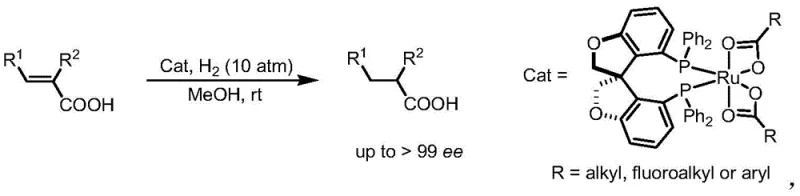 The reaction operates effectively at room temperature in methanol with a hydrogen pressure of 10 atm, showcasing the high activity of the catalyst. Such high levels of enantioselectivity are crucial for pharmaceutical applications where even trace amounts of the wrong enantiomer can be detrimental. The ability to achieve these results with low catalyst loading underscores the efficiency of the ligand design and its potential for cost-effective manufacturing processes.
The reaction operates effectively at room temperature in methanol with a hydrogen pressure of 10 atm, showcasing the high activity of the catalyst. Such high levels of enantioselectivity are crucial for pharmaceutical applications where even trace amounts of the wrong enantiomer can be detrimental. The ability to achieve these results with low catalyst loading underscores the efficiency of the ligand design and its potential for cost-effective manufacturing processes.
How to Synthesize Oxaspiro Diphosphine Ligand Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for producing these valuable ligands with high purity and yield. The process begins with the activation of the spiro diphenol core followed by sequential introduction of phosphorus functionalities. Each step has been optimized to minimize side reactions and facilitate easy purification, typically via column chromatography or crystallization. The use of common laboratory reagents and standard heating equipment makes this protocol accessible for both research and production environments. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during scale-up operations.
- Convert the spiro diphenol precursor into a bis-triflate intermediate using pyridine and Tf2O in DCM at 0 °C to room temperature.
- Perform a palladium-catalyzed coupling reaction with diarylphosphine oxide (Ar2POH) using Pd(OAc)2 and dppb in DMSO at 100 °C.
- Reduce the phosphine oxide groups to phosphines using trichlorosilane (HSiCl3) and DIPEA in toluene at 120 °C to obtain the final ligand.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel ligand synthesis offers tangible benefits regarding cost stability and supply security. The streamlined synthetic route reduces the dependency on exotic reagents, thereby mitigating risks associated with raw material shortages. By simplifying the manufacturing process, producers can achieve faster turnaround times and more consistent batch-to-batch quality. This reliability is essential for maintaining continuous production schedules in the fast-paced pharmaceutical industry. Furthermore, the high efficiency of the catalyst reduces the total amount of ligand required per batch of product, leading to substantial cost savings in the long run.
- Cost Reduction in Manufacturing: The elimination of complex resolution steps and the use of racemic starting materials significantly lowers the entry barrier for production. The high yields reported in the patent examples indicate a very atom-economical process that minimizes waste disposal costs. By avoiding expensive chiral resolving agents and reducing the number of synthetic steps, the overall cost of goods is drastically simplified. This economic efficiency allows for competitive pricing strategies when sourcing these critical chiral building blocks for API synthesis.
- Enhanced Supply Chain Reliability: The synthesis relies on robust chemical transformations that are less sensitive to minor variations in reaction conditions, ensuring consistent output. The starting materials, such as spiro diphenols and diarylphosphine oxides, are commercially available or easily synthesized, reducing supply chain fragility. This stability ensures that manufacturers can meet tight delivery deadlines without compromising on quality. A reliable supply of high-performance ligands is crucial for preventing production delays in downstream drug manufacturing processes.
- Scalability and Environmental Compliance: The reaction conditions utilize standard solvents like toluene and DMSO which are well-understood in terms of handling and recycling. The avoidance of highly toxic reagents aligns with modern green chemistry principles and simplifies regulatory compliance. The process is designed to be scalable from gram to kilogram quantities without loss of efficiency, supporting the transition from pilot plant to commercial production. This scalability ensures that the technology can meet the growing demand for chiral intermediates in the global market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxaspiro diphosphine ligand technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation. Understanding these details helps stakeholders make informed decisions about integrating this catalyst system into their existing workflows. We encourage technical teams to review these points when evaluating the feasibility of this approach for their specific projects.
Q: What is the enantioselectivity achieved with this oxaspiro ligand?
A: The ruthenium complex prepared with this ligand demonstrates exceptional enantioselectivity, achieving greater than 99% ee in the asymmetric hydrogenation of unsaturated carboxylic acids such as 2-methyl cinnamic acid.
Q: Can this synthesis be scaled for industrial production?
A: Yes, the synthesis utilizes robust reaction conditions including standard solvents like toluene and DMSO, and temperatures ranging from 100 °C to 120 °C, which are highly amenable to commercial scale-up in standard reactor vessels.
Q: What types of substrates are compatible with this catalyst system?
A: The catalyst system is particularly effective for the asymmetric hydrogenation of unsaturated carboxylic acids. The ligand structure allows for tunability via the aryl groups (Ar), enabling optimization for various substrate classes including alkyl, fluoroalkyl, or aryl substituted olefins.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Oxaspiro Diphosphine Ligand Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our rigorous QC labs and stringent purity specifications guarantee that every batch of oxaspiro diphosphine ligand meets the highest international standards required for pharmaceutical applications. We understand the critical nature of chiral catalysts in drug synthesis and are committed to providing uninterrupted supply continuity for our global partners. Our team of experts is ready to support your R&D efforts with custom synthesis solutions tailored to your specific process requirements.
We invite you to contact our technical procurement team to request specific COA data and route feasibility assessments for your upcoming projects. By collaborating with us, you can leverage our expertise to conduct a Customized Cost-Saving Analysis that identifies opportunities to optimize your manufacturing budget. Let us help you secure a stable source of high-quality chiral ligands that drive innovation and efficiency in your drug development pipeline. Reach out today to discuss how our advanced synthesis capabilities can support your long-term strategic goals.
