Advanced Copper-Catalyzed Synthesis of 2-Phenylselenyl-3-Amino-1,4-Naphthoquinone Intermediates
The pharmaceutical and fine chemical industries are constantly seeking efficient pathways to construct complex heterocyclic scaffolds that serve as the backbone for next-generation therapeutics. A significant breakthrough in this domain is detailed in patent CN111606843A, which discloses a robust method for synthesizing 2-phenylselenyl-3-amino-1,4-naphthoquinone derivatives. These 2,3-difunctionalized naphthoquinones are not merely academic curiosities; they represent a critical class of structural motifs found in potent bioactive natural products such as Naphthomycin B, Jadomycin B, and various anticancer agents like Griffithazanone A. The ability to efficiently access these densely functionalized cores opens new avenues for medicinal chemistry campaigns aimed at developing novel antibiotics and antitumor drugs. This report analyzes the technical merits of this copper-catalyzed selenoamination strategy, highlighting its potential to redefine the supply chain for high-value pharmaceutical intermediates.
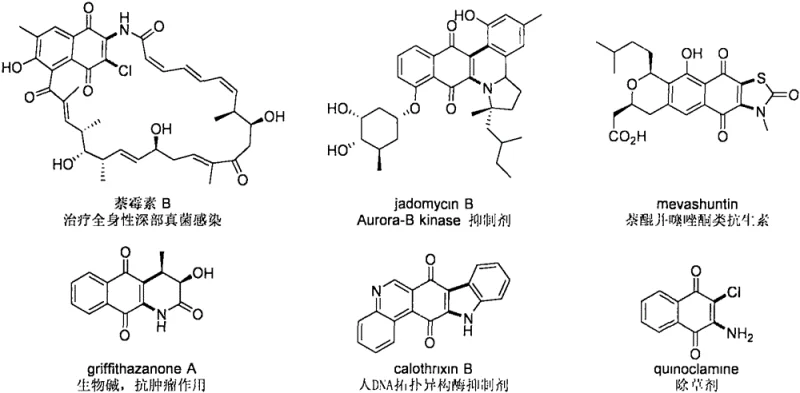
The strategic importance of this synthesis lies in its ability to introduce two distinct functional groups simultaneously onto the naphthoquinone core. Historically, modifying the naphthoquinone skeleton has been a challenge due to the reactivity of the quinone moiety and the difficulty in controlling regioselectivity. The patented method overcomes these hurdles by leveraging a transition metal copper catalyst under an oxygen atmosphere. This approach transforms simple, commercially available starting materials—diphenyl diselenide, secondary amines, and 1,4-naphthoquinone—into sophisticated molecular architectures. For R&D directors and procurement specialists, understanding the nuances of this transformation is key to evaluating its viability for large-scale production of reliable pharmaceutical intermediate supplies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the functionalization of naphthoquinones was often limited by the use of expensive precious metal catalysts and restrictive reaction scopes. For instance, research published by the John F. Bower group in 2016 demonstrated a rhodium-catalyzed C-H bond arylselenylation of naphthoquinones. While scientifically significant, this legacy technology suffers from substantial commercial drawbacks. The reliance on rhodium, a scarce and costly platinum-group metal, inherently drives up the raw material costs, making the process economically unfeasible for large-scale manufacturing. Furthermore, the rhodium-catalyzed pathway typically results in mono-functionalization, introducing only an arylselenyl group while leaving the adjacent position unmodified. This limitation necessitates additional synthetic steps to install the second functional group, thereby increasing waste generation, extending lead times, and reducing overall atom economy.
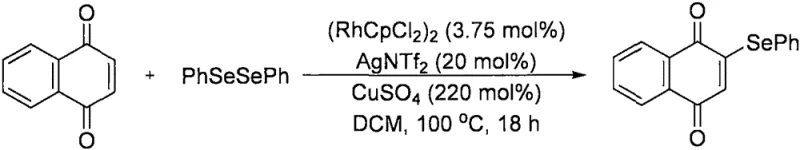
In addition to cost and step inefficiency, conventional methods often struggle with harsh reaction conditions that can degrade sensitive functional groups on the amine or selenium components. The requirement for silver additives in some rhodium systems further complicates the downstream purification process, as removing trace heavy metals to meet stringent pharmaceutical purity specifications can be technically demanding. These factors collectively create a bottleneck in the supply chain for complex naphthoquinone derivatives, limiting their availability for drug discovery programs and delaying the development of potential new medicines. The industry urgently requires a method that bypasses these economic and technical barriers.
The Novel Approach
The methodology described in CN111606843A represents a paradigm shift by utilizing an earth-abundant copper catalyst to achieve direct selenoamination. This novel approach enables the simultaneous introduction of both arylselenyl and amino functionalities onto the 1,4-naphthoquinone scaffold in a single operational step. The general reaction scheme involves the coupling of diphenyl diselenide and a secondary amine with the naphthoquinone substrate in the presence of copper acetate. This multi-component tandem reaction not only simplifies the synthetic route but also significantly enhances the structural diversity accessible to chemists. By varying the secondary amine component, a wide array of 2,3-disubstituted naphthoquinone derivatives can be generated rapidly, facilitating the exploration of structure-activity relationships (SAR) in drug design.
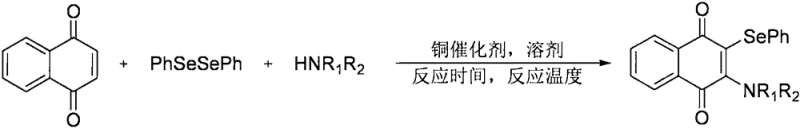
From a commercial perspective, this novel approach offers profound advantages in cost reduction in pharmaceutical intermediate manufacturing. The substitution of rhodium with copper acetate drastically reduces catalyst costs, while the elimination of silver additives simplifies the workup procedure. The reaction proceeds efficiently in polar aprotic solvents like N,N-dimethylformamide (DMF) under an oxygen atmosphere, conditions that are easily manageable in standard industrial reactors. The ability to achieve high yields and purity with such a streamlined process positions this technology as a superior alternative for the commercial scale-up of complex polymer additives and fine chemicals, ensuring a more reliable and cost-effective supply chain for downstream users.
Mechanistic Insights into Copper-Catalyzed Selenoamination
The success of this transformation hinges on the unique reactivity of the copper catalyst in activating the selenium-selenium bond of diphenyl diselenide. Under the oxidative conditions provided by the oxygen atmosphere, the copper species likely facilitates the homolytic cleavage of the diselenide, generating reactive phenylselenyl radicals or cationic selenium species. These active intermediates then attack the electron-deficient double bond of the 1,4-naphthoquinone. Subsequently, the secondary amine nucleophile adds to the activated intermediate, completing the difunctionalization. The specific choice of copper acetate is critical; experimental data within the patent indicates that other copper salts such as cuprous iodide, copper chloride, and copper bromide fail to promote the reaction, suggesting that the acetate ligand plays a crucial role in the catalytic cycle, possibly by modulating the Lewis acidity of the copper center or assisting in proton transfer steps.
Impurity control is another vital aspect of this mechanism that appeals to quality assurance teams. The reaction conditions are tuned to minimize side reactions such as over-oxidation or polymerization of the quinone core. The use of DMF as the preferred solvent is not arbitrary; screening revealed that solvents like toluene, acetonitrile, and tetrahydrofuran resulted in no reaction, while dimethyl sulfoxide (DMSO) gave lower yields compared to DMF. This specificity ensures a cleaner reaction profile, reducing the burden on purification teams. The high selectivity observed implies that the catalytic cycle is well-defined, preventing the formation of complex impurity profiles that often plague multi-component reactions. This mechanistic robustness is essential for maintaining high-purity OLED material or pharmaceutical intermediate standards required by regulatory bodies.
How to Synthesize 2-Phenylselenyl-3-Amino-1,4-Naphthoquinone Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific parameters to maximize yield and safety. The process is designed to be operationally simple, avoiding the need for inert gas gloveboxes or cryogenic conditions. The standard protocol involves charging the reactor with the naphthoquinone substrate, diphenyl diselenide, and the chosen secondary amine in a molar ratio of approximately 1:2:3. Copper acetate is added at a loading of 10 mol%, and the mixture is dissolved in DMF. The reaction vessel is then subjected to an oxygen atmosphere, typically by evacuating and refilling with oxygen gas three times to ensure sufficient oxidant is present. The mixture is heated to 120°C and stirred for 24 hours. Upon completion, the workup involves standard extraction techniques followed by column chromatography.
- Combine 1,4-naphthoquinone, diphenyl diselenide, and secondary amine in DMF solvent with copper acetate catalyst.
- Purge the reaction vessel with oxygen three times and maintain an oxygen atmosphere throughout the process.
- Heat the mixture to 120°C for 24 hours, then cool, extract with ethyl acetate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this copper-catalyzed methodology offers tangible benefits that extend beyond simple chemistry. The primary advantage lies in the drastic simplification of the sourcing strategy. Unlike precious metal catalysts which are subject to volatile market prices and geopolitical supply risks, copper acetate is a commodity chemical available from multiple global suppliers at stable prices. This shift mitigates supply chain disruptions and allows for more accurate long-term budgeting. Furthermore, the use of common secondary amines and diphenyl diselenide means that raw material inventory can be consolidated, reducing warehousing costs and administrative overhead associated with managing specialized reagents.
- Cost Reduction in Manufacturing: The economic impact of replacing rhodium with copper cannot be overstated. Precious metal catalysts often require complex recovery and recycling processes to be economically viable, adding capital expenditure for specialized equipment. By utilizing a base metal catalyst that does not require such intensive recovery protocols, manufacturers can significantly lower their operating expenses. Additionally, the high efficiency of the reaction reduces the amount of raw material wasted, improving the overall mass balance of the process. The simplified post-reaction processing, which avoids the removal of silver additives, further reduces solvent consumption and labor hours, contributing to substantial cost savings in the final product price.
- Enhanced Supply Chain Reliability: Reliability is the cornerstone of a resilient supply chain. The robustness of this reaction against variations in conditions ensures consistent batch-to-batch quality, reducing the risk of production delays caused by failed runs. Since the raw materials are widely produced industrial chemicals, the risk of shortage is minimal compared to specialized organometallic complexes. This stability allows suppliers to offer shorter lead times and more flexible delivery schedules, enabling pharmaceutical companies to maintain leaner inventories without compromising their production timelines. The ability to source reliable pharmaceutical intermediate supplier materials with confidence is a strategic asset in a competitive market.
- Scalability and Environmental Compliance: Scaling chemical processes from grams to tons often reveals hidden challenges, but this methodology is inherently scalable. The reaction uses standard solvents and operates at temperatures compatible with existing stainless steel reactors, eliminating the need for exotic metallurgy. From an environmental standpoint, the use of oxygen as the terminal oxidant produces water as the primary byproduct, aligning with green chemistry principles. The absence of toxic heavy metals like rhodium in the final waste stream simplifies effluent treatment and disposal, helping manufacturers meet increasingly stringent environmental regulations. This ease of scale-up and compliance facilitates the commercial scale-up of complex pharmaceutical intermediates with reduced regulatory friction.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The clarity provided here aims to bridge the gap between academic innovation and industrial application.
Q: Why is copper acetate preferred over other copper salts for this selenoamination?
A: According to patent CN111606843A, experimental data demonstrates that while cuprous iodide, copper chloride, and copper bromide fail to promote the reaction, copper acetate acts as the critical catalyst enabling high yields, likely due to its specific solubility and redox properties in DMF.
Q: What are the advantages of this method over previous rhodium-catalyzed routes?
A: Previous rhodium-catalyzed methods typically only introduce a single arylselenyl group and require expensive precious metal catalysts. This novel copper-catalyzed approach achieves dual functionalization (introducing both selenyl and amino groups simultaneously) using cost-effective base metals.
Q: Is this synthesis scalable for commercial pharmaceutical intermediate production?
A: Yes, the process utilizes readily available raw materials like diphenyl diselenide and common secondary amines, operates at moderate temperatures (120°C), and uses standard organic solvents, making it highly suitable for scale-up from laboratory to industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Phenylselenyl-3-Amino-1,4-Naphthoquinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the copper-catalyzed selenoamination described in CN111606843A. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate such innovative laboratory protocols into robust commercial processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can grow seamlessly from clinical trial material to full-scale market supply. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch.
We invite you to collaborate with us to leverage this cutting-edge technology for your drug development pipeline. Whether you require custom synthesis of specific naphthoquinone derivatives or optimization of the reaction parameters for your specific needs, our technical procurement team is ready to assist. Contact us today to request a Customized Cost-Saving Analysis tailored to your project volume. We encourage you to reach out for specific COA data and route feasibility assessments to see how we can accelerate your timeline and reduce your overall development costs.
