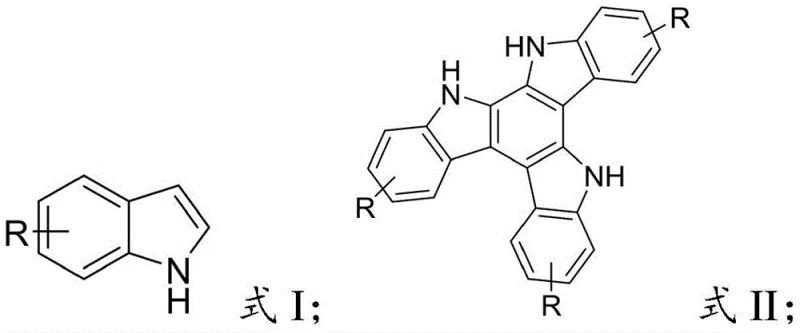
Advanced Copper-Catalyzed Synthesis of Asymmetric Trimeric Indoles for High-Performance OLED Applications
Advanced Copper-Catalyzed Synthesis of Asymmetric Trimeric Indoles for High-Performance OLED Applications
The rapid evolution of organic optoelectronics has placed immense pressure on supply chains to deliver high-purity intermediates with consistent quality and scalable production capabilities. In this context, patent CN114014865A introduces a groundbreaking methodology for the synthesis of asymmetric trimeric indole compounds, which serve as critical electron-rich donor units in organic electroluminescent devices. This technology addresses the longstanding challenges associated with constructing complex indole oligomers by utilizing a mild, copper-catalyzed oxidative coupling strategy. Unlike traditional methods that often require harsh conditions or expensive palladium catalysts, this novel approach leverages the unique reactivity of copper(II) species to facilitate C-C bond formation at the C-2 position of the indole ring. For R&D directors and procurement managers in the electronic chemicals sector, this represents a significant opportunity to optimize the manufacturing of OLED materials, ensuring both cost efficiency and supply chain reliability for next-generation display technologies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trimeric indole derivatives has relied on methodologies that are fraught with operational complexity and economic inefficiency. Prior art, such as the work by Andrikaityte et al., utilized phosphorus oxychloride to induce dehydration condensation, a process that generates corrosive waste and requires stringent safety controls. Alternatively, symmetric trimeric indoles have been accessed via Suzuki-Miyaura coupling reactions mediated by palladium, as described by Reghu et al. While effective for symmetric structures, these palladium-catalyzed routes are often prohibitively expensive for large-scale commercial production due to the high cost of the metal and the necessity for rigorous removal of residual heavy metals to meet electronic grade specifications. Furthermore, achieving asymmetry in these trimeric structures using conventional cross-coupling techniques typically involves multi-step protection and deprotection sequences, drastically reducing overall throughput and increasing the environmental footprint of the manufacturing process.
The Novel Approach
The methodology disclosed in patent CN114014865A offers a transformative alternative by employing a direct oxidative coupling mechanism catalyzed by inexpensive copper(II) salts. This process operates under remarkably mild conditions, typically between 0°C and 20°C, and utilizes bis(trifluoroacetoxy)iodobenzene (PIFA) as a stoichiometric oxidant in a toluene medium. The reaction proceeds rapidly, often completing within 5 to 10 minutes, which stands in stark contrast to the prolonged reaction times associated with traditional cross-coupling protocols. By eliminating the need for precious metal catalysts and simplifying the workup procedure to standard extraction and column chromatography, this novel approach significantly streamlines the production workflow. The ability to directly couple three indole units in a single pot reaction not only enhances atom economy but also minimizes the generation of hazardous waste, aligning perfectly with modern green chemistry principles required by leading multinational corporations.
Mechanistic Insights into Cu(II)-Catalyzed Oxidative Coupling
The mechanistic pathway elucidated in the patent provides a fascinating glimpse into the radical-mediated nature of this transformation, which is crucial for R&D teams aiming to replicate or modify the process. The cycle initiates when the copper(II) catalyst interacts with the oxidant, generating a highly reactive copper species that inserts into the C-2 position of the first indole molecule to form a transient C-Cu bond. This bond subsequently undergoes homolytic cleavage to produce an indole radical, a key reactive intermediate that drives the oligomerization forward. This radical species then attacks the C-2 position of a second indole molecule, forming a dimeric intermediate, which further reacts with a third indole unit via radical addition. The final step involves a single electron transfer (SET) oxidation catalyzed by Cu(II), generating a carbocation intermediate that undergoes oxidative dehydrogenation to yield the fully aromatic asymmetric trimeric indole structure. Understanding this radical cascade is essential for controlling selectivity and minimizing the formation of higher-order oligomers or polymeric byproducts.
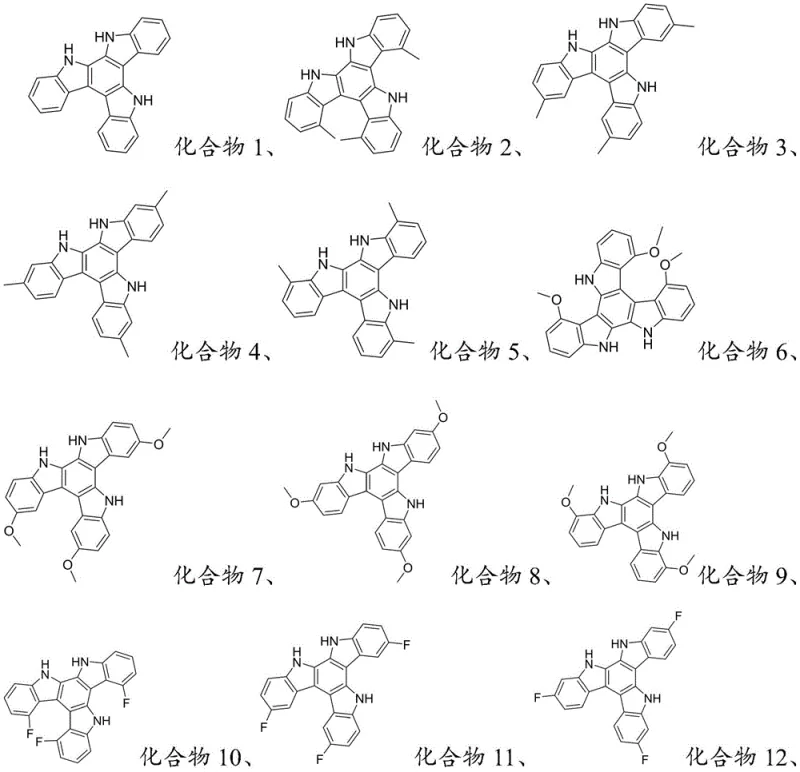
From an impurity control perspective, the mild reaction temperature and the specific choice of toluene as a solvent play pivotal roles in ensuring high product purity. Comparative examples within the patent data highlight that switching solvents to dichloroethane or ethyl acetate results in a dramatic drop in yield, suggesting that solvent polarity and coordination ability significantly influence the stability of the radical intermediates. Furthermore, the use of PIFA as the oxidant ensures a clean oxidation profile without introducing halogenated impurities that could compromise the electronic properties of the final OLED material. The robustness of this mechanism is evidenced by its tolerance to a wide variety of substituents, including alkyl, alkoxy, halogen, and acyl groups, allowing for the fine-tuning of electronic properties such as HOMO-LUMO levels without sacrificing reaction efficiency. This level of control is indispensable for producing high-purity electronic chemicals where even trace impurities can degrade device performance.
How to Synthesize Asymmetric Trimeric Indole Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to the order of addition and temperature control to maximize the yield and reproducibility of the reaction. The protocol generally involves preparing two separate solutions: one containing the indole substrate and the copper catalyst dissolved in toluene, and another containing the PIFA oxidant also dissolved in toluene. The oxidant solution is then added dropwise to the indole-catalyst mixture over a short period, typically 1 to 3 minutes, while maintaining the reaction temperature between 0°C and 20°C. This controlled addition prevents the accumulation of excessive radical species that could lead to uncontrolled polymerization. Following the brief reaction period, the mixture is quenched and subjected to a standard aqueous workup involving saturated sodium bicarbonate and ethyl acetate extraction. The detailed standardized synthesis steps for scaling this process are outlined below.
- Dissolve the indole compound (Formula I) and a copper(II) catalyst such as Cu(OAc)2 or CuBr2 in toluene to form a catalyst solution.
- Dissolve the oxidant bis(trifluoroacetoxy)iodobenzene (PIFA) in toluene separately to prepare the oxidant solution.
- Dropwise add the oxidant solution to the indole-catalyst mixture at 0-20°C, stir for 5-10 minutes, then extract, dry, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this copper-catalyzed methodology offers tangible benefits that extend beyond mere technical feasibility. The primary advantage lies in the substantial cost reduction achieved by replacing expensive palladium catalysts with abundant and inexpensive copper salts. This shift not only lowers the direct material cost but also eliminates the need for specialized scavenging resins or complex purification steps required to remove trace palladium to ppb levels, which is a mandatory requirement for electronic grade materials. Additionally, the simplified one-pot nature of the reaction reduces labor costs and equipment occupancy time, allowing for higher throughput in existing manufacturing facilities. The use of common solvents like toluene further simplifies logistics, as there is no need for specialized solvent handling infrastructure, thereby enhancing the overall agility of the supply chain.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts represents a major financial saving, as copper salts are orders of magnitude cheaper than palladium complexes. Furthermore, the high atom economy of the oxidative coupling means less raw material is wasted in the form of byproducts, leading to a more efficient utilization of starting indole derivatives. The simplified workup procedure, which avoids complex chromatographic separations often needed for cross-coupling byproducts, reduces the consumption of silica gel and eluents, contributing to lower operational expenditures. These cumulative savings allow for a more competitive pricing structure for the final asymmetric trimeric indole products, making them accessible for broader applications in the organic electronics industry.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable reagents such as Cu(OAc)2, CuBr2, and PIFA ensures a robust supply chain that is less susceptible to geopolitical disruptions often associated with rare earth or precious metal sourcing. Since the starting indole compounds are commodity chemicals available from multiple global suppliers, there is minimal risk of single-source bottlenecks. The short reaction time of merely 5 to 10 minutes also implies that production capacity can be rapidly scaled up by increasing batch frequency rather than requiring massive capital investment in new reactors. This flexibility allows suppliers to respond quickly to fluctuating market demands from OLED panel manufacturers, ensuring continuity of supply even during peak production cycles.
- Scalability and Environmental Compliance: The mild reaction conditions (0-20°C) significantly reduce the energy consumption associated with heating or cryogenic cooling, aligning with corporate sustainability goals and reducing the carbon footprint of the manufacturing process. The absence of toxic heavy metals like palladium simplifies waste disposal and regulatory compliance, as the effluent streams are easier to treat and do not require specialized hazardous waste handling protocols. The high yields reported (62-80%) across a diverse range of substrates indicate that the process is robust enough for multi-kilogram or ton-scale production without significant loss of efficiency. This scalability is critical for meeting the growing volume requirements of the display industry while maintaining strict environmental, health, and safety (EHS) standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric trimeric indole synthesis technology. These answers are derived directly from the experimental data and comparative examples provided in the patent documentation to ensure accuracy and relevance for potential partners. Understanding these nuances is vital for assessing the feasibility of integrating this chemistry into your existing production lines or R&D pipelines. We encourage technical teams to review these points carefully to appreciate the full scope of advantages offered by this innovative copper-catalyzed approach.
Q: What is the typical yield range for this copper-catalyzed trimerization?
A: According to the experimental data in patent CN114014865A, the preparation method achieves yields ranging from 62% to 80% depending on the specific substituents on the indole ring.
Q: Why is toluene preferred over other solvents like dichloroethane?
A: Comparative examples in the patent demonstrate that using toluene results in significantly higher yields (e.g., 80%) compared to dichloroethane (40%) or ethyl acetate (30%), indicating superior solubility and reaction kinetics in toluene.
Q: Can this method tolerate halogen substituents on the indole ring?
A: Yes, the method exhibits excellent functional group tolerance. Examples 12 through 21 successfully synthesized trifluoro, trichloro, and tribromo derivatives with yields often exceeding 70-80%, proving robustness for halogenated substrates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Asymmetric Trimeric Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance intermediates play in the advancement of organic optoelectronic devices. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering asymmetric trimeric indoles with stringent purity specifications, utilizing our rigorous QC labs to verify that every batch meets the exacting standards required for OLED applications. Our expertise in copper-catalyzed oxidative couplings allows us to optimize this specific patent technology for maximum yield and cost-effectiveness, providing our clients with a distinct competitive edge in the marketplace.
We invite you to engage with our technical procurement team to discuss how this novel synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this copper-catalyzed method for your supply chain. We are ready to provide specific COA data and route feasibility assessments to demonstrate our capability to support your long-term material requirements. Let us partner with you to drive innovation and efficiency in the next generation of electronic materials.
