Advanced Synthesis of Ruthenium Carbene Catalysts via Phosphorus Ylide Route for Industrial Scale-up
The landscape of organometallic catalysis has been revolutionized by the advent of ruthenium carbene complexes, particularly those known as Grubbs catalysts, which are indispensable for olefin metathesis reactions in the synthesis of complex pharmaceutical intermediates and advanced materials. However, the historical manufacturing processes for these high-value catalysts have been plagued by significant safety hazards and operational complexities, often relying on unstable and explosive precursors. Patent CN111468191A introduces a transformative synthetic methodology that addresses these critical bottlenecks by utilizing a phosphorus ylide route. This innovation shifts the paradigm from dangerous diazo-based chemistry to a safer, more controllable process involving benzyl halides and triphenylphosphine. For R&D directors and procurement specialists seeking a reliable ruthenium carbene catalyst supplier, this technology represents a pivotal advancement in ensuring supply chain continuity and process safety. The method not only simplifies the reaction conditions but also enhances atom economy, making it an ideal candidate for the commercial scale-up of complex catalytic systems required in modern fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of first-generation ruthenium carbene catalysts relied heavily on the use of phenyldiazomethane, a compound notorious for its extreme instability and explosive potential. As illustrated in early literature from 1996, the reaction required stringent cryogenic conditions, typically maintained at -78°C, to prevent the decomposition of the diazo species and to control the exothermic nature of the carbene transfer. 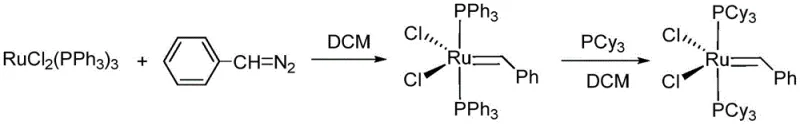 This reliance on hazardous reagents imposed severe limitations on batch sizes, effectively restricting production to small-scale laboratory preparations rather than industrial manufacturing. Furthermore, alternative methods developed later, such as those utilizing sulfur ylides, introduced new challenges regarding moisture sensitivity and difficult purification steps. These conventional pathways often resulted in lower overall yields due to side reactions and required complex post-processing to remove toxic by-products, thereby increasing the cost reduction in catalyst manufacturing efforts without delivering proportional efficiency gains. The operational rigidity of these older methods made them unsuitable for the dynamic demands of the global pharmaceutical supply chain.
This reliance on hazardous reagents imposed severe limitations on batch sizes, effectively restricting production to small-scale laboratory preparations rather than industrial manufacturing. Furthermore, alternative methods developed later, such as those utilizing sulfur ylides, introduced new challenges regarding moisture sensitivity and difficult purification steps. These conventional pathways often resulted in lower overall yields due to side reactions and required complex post-processing to remove toxic by-products, thereby increasing the cost reduction in catalyst manufacturing efforts without delivering proportional efficiency gains. The operational rigidity of these older methods made them unsuitable for the dynamic demands of the global pharmaceutical supply chain.
The Novel Approach
In stark contrast to the perilous diazo routes, the methodology disclosed in CN111468191A employs a benign phosphorus ylide strategy that fundamentally alters the risk profile of catalyst production. The process initiates with the quaternization of triphenylphosphine using readily available benzyl halides, such as benzyl bromide or benzyl chloride, to form a stable phosphonium salt. This intermediate is then deprotonated using strong bases like n-butyllithium or potassium tert-butoxide to generate the active phosphorus ylide in situ. 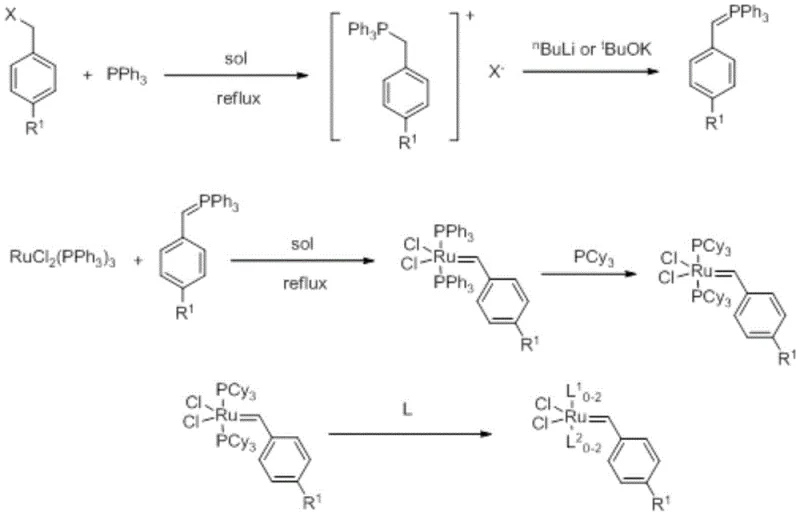 This ylide subsequently reacts with tris(triphenylphosphine)ruthenium(II) dichloride under mild conditions, typically ranging from -30°C to ambient temperature, to form the ruthenium carbene complex precursor. The subsequent ligand exchange with tricyclohexylphosphine (PCy3) is straightforward and high-yielding. This novel approach eliminates the need for cryogenic infrastructure and explosive reagents, thereby facilitating high-purity organometallic catalysts production with significantly reduced safety overheads. The simplicity of the workup, involving basic filtration and recrystallization, ensures that the final product meets the rigorous quality standards demanded by downstream applications in drug discovery and material science.
This ylide subsequently reacts with tris(triphenylphosphine)ruthenium(II) dichloride under mild conditions, typically ranging from -30°C to ambient temperature, to form the ruthenium carbene complex precursor. The subsequent ligand exchange with tricyclohexylphosphine (PCy3) is straightforward and high-yielding. This novel approach eliminates the need for cryogenic infrastructure and explosive reagents, thereby facilitating high-purity organometallic catalysts production with significantly reduced safety overheads. The simplicity of the workup, involving basic filtration and recrystallization, ensures that the final product meets the rigorous quality standards demanded by downstream applications in drug discovery and material science.
Mechanistic Insights into Phosphorus Ylide-Mediated Carbene Transfer
The core mechanistic advantage of this patented process lies in the controlled generation and immediate consumption of the phosphorus ylide species, which acts as a stable carbene equivalent. Unlike free carbenes which are transient and highly reactive, the phosphorus ylide offers a stabilized electronic environment that allows for a more predictable coordination to the ruthenium center. The reaction proceeds through a nucleophilic attack of the ylide carbon on the ruthenium atom, displacing a triphenylphosphine ligand and forming the initial metal-carbon double bond characteristic of the carbene complex. This step is critical as it determines the stereochemistry and electronic properties of the resulting catalyst. The patent highlights that the reaction kinetics can be efficiently modulated by adjusting the molar ratios of the ylide to the ruthenium precursor, typically between 1:1 and 1:10, allowing for precise control over the reaction rate and minimizing the formation of inactive ruthenium clusters. This level of control is essential for maintaining consistent batch-to-batch quality in a commercial setting.
Furthermore, the mechanism includes a crucial ligand exchange phase where the labile triphenylphosphine ligands are replaced by the more electron-donating and sterically bulky tricyclohexylphosphine (PCy3). This substitution enhances the thermal stability and catalytic activity of the final complex. The patent details that this exchange can be performed in common organic solvents such as toluene or tetrahydrofuran, avoiding the need for exotic or hazardous media. Impurity control is achieved through a targeted recrystallization process using specific solvent pairs, such as toluene and methanol or dichloromethane and petroleum ether. This purification step is vital for removing residual phosphine oxides and unreacted salts, which could otherwise poison the catalyst during downstream metathesis reactions. By understanding these mechanistic nuances, manufacturers can optimize reaction parameters to achieve reducing lead time for high-purity intermediates while ensuring the structural integrity of the active catalytic species.
How to Synthesize Ruthenium Carbene Catalyst Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing both first and second-generation Grubbs-type catalysts with high reproducibility. The process is divided into three distinct stages: the preparation of the phosphonium salt and subsequent ylide, the formation of the ruthenium carbene precursor, and the final functionalization with specific ligands. Each stage is designed to be operationally simple, utilizing standard laboratory equipment such as Schlenk flasks and standard filtration setups, which translates well to pilot and plant-scale reactors. The use of common solvents like dichloromethane, toluene, and ethers further simplifies the logistical requirements for solvent recovery and waste management. For technical teams looking to implement this route, the detailed experimental examples provided in the patent serve as a valuable reference for optimizing stoichiometry and temperature profiles to maximize yield and purity.
- React benzyl halide with triphenylphosphine to form a phosphonium salt, then treat with a strong base to generate the phosphorus ylide intermediate.
- React the phosphorus ylide with tris(triphenylphosphine)ruthenium(II) dichloride in a solvent to form the ruthenium carbene complex precursor.
- Perform ligand exchange using tricyclohexylphosphine and subsequently react with functional ligands to obtain the final target ruthenium carbene catalyst.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this phosphorus ylide-based synthesis route offers substantial strategic benefits for procurement managers and supply chain directors. The primary advantage stems from the drastic simplification of the raw material portfolio; replacing explosive diazo compounds with stable benzyl halides and phosphines significantly lowers the cost of goods sold (COGS) associated with hazardous material handling and storage. This shift eliminates the need for specialized cryogenic reactors and explosion-proof facilities, leading to significant capital expenditure savings and a streamlined manufacturing footprint. Moreover, the improved safety profile reduces insurance premiums and regulatory compliance burdens, contributing to overall cost reduction in catalyst manufacturing. The robustness of the process also means fewer batch failures and less downtime, ensuring a more reliable supply of critical catalytic materials for downstream customers.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents like phenyldiazomethane directly impacts the bottom line by reducing raw material costs and waste disposal fees. The process operates at near-ambient temperatures for significant portions of the reaction, lowering energy consumption compared to the energy-intensive cryogenic cooling required by legacy methods. Additionally, the high atom economy and efficient recrystallization steps minimize solvent usage and maximize product recovery, further driving down production costs without compromising on quality standards.
- Enhanced Supply Chain Reliability: By utilizing commodity chemicals such as benzyl bromide and triphenylphosphine, the supply chain becomes far less vulnerable to disruptions caused by the limited availability of specialized reagents. These starting materials are produced globally in large volumes, ensuring consistent access and price stability. The simplified synthesis also shortens the production cycle time, allowing manufacturers to respond more agilely to fluctuations in market demand and reducing the lead time for delivering high-purity organometallic catalysts to clients.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of toxic by-products make this method inherently scalable from gram to ton quantities. The waste stream is primarily composed of benign phosphine salts and common organic solvents, which are easier to treat and dispose of in compliance with environmental regulations. This eco-friendly profile aligns with the growing industry demand for sustainable chemical manufacturing practices, enhancing the corporate social responsibility standing of the production facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis technology. They are derived from the specific advantages and operational details described in the patent documentation, providing clarity for stakeholders evaluating this manufacturing route. Understanding these aspects is crucial for making informed decisions about integrating this technology into existing production lines or sourcing strategies.
Q: How does this synthesis method improve safety compared to traditional diazo routes?
A: Traditional methods often utilize explosive phenyldiazomethane at cryogenic temperatures (-78°C). This patented method replaces hazardous diazo compounds with stable benzyl halides and phosphorus ylides, allowing reactions to proceed at much milder temperatures ranging from -30°C to 100°C, significantly enhancing operational safety.
Q: What are the purity specifications achievable with this recrystallization process?
A: The patent describes a robust purification strategy involving filtration, washing, and recrystallization using specific solvent pairs like toluene/methanol or dichloromethane/methanol. This ensures the removal of phosphine oxide by-products and unreacted starting materials, yielding high-purity catalyst precursors suitable for sensitive pharmaceutical applications.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the method is designed for industrial scalability. It avoids the strict low-temperature constraints and explosive hazards of earlier generations, utilizes readily available raw materials like benzyl bromide, and features simple workup procedures such as filtration and recrystallization, making it highly amenable to multi-kilogram or ton-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ruthenium Carbene Catalyst Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance catalysts play in accelerating drug discovery and advanced material development. Leveraging the innovative synthetic pathways described in patents like CN111468191A, we have established ourselves as a leader in the production of complex organometallic compounds. Our facility is equipped with state-of-the-art reactor systems capable of handling sensitive organometallic chemistry under inert atmospheres, ensuring the highest levels of product integrity. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, adhering to stringent purity specifications and operating rigorous QC labs to verify every batch. Our commitment to quality ensures that every gram of catalyst delivered meets the exacting standards required for GMP-compliant pharmaceutical synthesis.
We invite potential partners to engage with our technical team to explore how our advanced manufacturing capabilities can optimize your supply chain. Whether you require custom synthesis of specific catalyst derivatives or large-volume supply of standard Grubbs catalysts, we are prepared to offer a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing you to validate our capabilities against your project needs. By partnering with us, you gain access to a secure, scalable, and cost-effective source of premium catalytic materials that will drive your innovation forward.
