Advanced Palladium-Catalyzed Synthesis Of Z-Fluoroalkylated Alkenyl Boronates For Commercial Scale-Up
Introduction To Novel Fluoroalkylation Technology
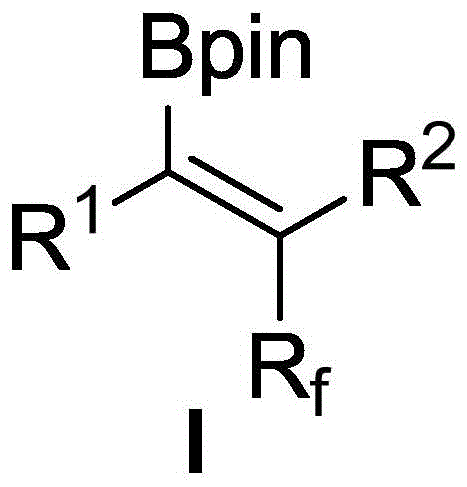
The landscape of modern organic synthesis is continually evolving to meet the rigorous demands of the pharmaceutical and agrochemical industries, particularly regarding the incorporation of fluorine atoms into complex molecular architectures. Patent CN108659028B represents a significant breakthrough in this domain by disclosing a highly efficient method for the preparation of (Z)-fluoroalkylated alkenyl boronates. These compounds serve as critical building blocks, offering enhanced metabolic stability and unique electronic properties that are indispensable in the design of next-generation active pharmaceutical ingredients (APIs). The invention addresses long-standing challenges in the field by enabling the direct, stereoselective construction of trisubstituted alkenes through a palladium-catalyzed three-component reaction. This approach not only streamlines the synthetic route but also ensures high atom economy, making it an attractive candidate for reliable pharmaceutical intermediate supplier networks seeking to optimize their manufacturing pipelines.
Traditionally, the synthesis of polysubstituted alkenyl boronates has been fraught with difficulties, especially when dealing with asymmetric internal alkynes. The steric hindrance and electronic complexity of these substrates often lead to poor regioselectivity and a mixture of stereoisomers, complicating downstream purification and reducing overall yield. The technology described in this patent overcomes these hurdles by utilizing a radical-induced mechanism that facilitates trans-carboboration. This results in the exclusive formation of the (Z)-isomer, providing chemists with a precise tool for molecular construction. By leveraging this innovation, manufacturers can achieve cost reduction in fluorochemical manufacturing by minimizing waste and reducing the number of synthetic steps required to reach complex fluorinated targets.
The Limitations Of Conventional Methods Vs. The Novel Approach
The Limitations Of Conventional Methods
Prior to this development, the carboboration of alkynes was largely limited to terminal alkynes or symmetric internal alkynes, where regiocontrol was less of an issue. For asymmetric internal alkynes, conventional borometallation strategies often failed to distinguish between the two carbon atoms of the triple bond, leading to inseparable mixtures of regioisomers. Furthermore, most metal-catalyzed additions to alkynes proceed via syn-addition mechanisms, yielding cis-alkenyl boronates. While useful, this limits the chemical space accessible to medicinal chemists who may require the trans-configuration to match the spatial requirements of a biological receptor. The inability to effectively functionalize internal alkynes with fluoroalkyl groups in a single step forced researchers to rely on multi-step sequences involving protection groups, harsh reaction conditions, and expensive stoichiometric reagents, all of which drive up production costs and extend lead times.
The Novel Approach
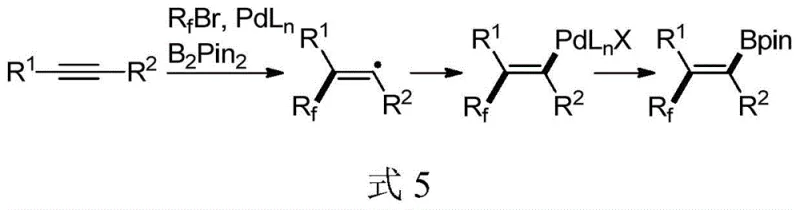
The methodology outlined in the patent introduces a paradigm shift by employing a palladium catalyst system capable of activating bromofluoroalkyl compounds to generate radical species in situ. This radical pathway bypasses the typical syn-addition constraint, allowing for the anti-addition of the fluoroalkyl group and the boron moiety across the triple bond. The result is a highly stereoselective transformation that delivers (Z)-fluoroalkylated alkenyl boronates with excellent fidelity. This novel approach simplifies the synthetic workflow significantly, as it combines three readily available components—asymmetric alkyne, bromofluoroalkyl halide, and bis(pinacolato)diboron—into a single reaction vessel. The mild reaction conditions, typically around 80°C in 1,2-dichloroethane, ensure compatibility with a wide range of sensitive functional groups, thereby expanding the scope of accessible chemical diversity for high-purity OLED material and pharmaceutical applications.
Mechanistic Insights Into Palladium-Catalyzed Trans-Carboboration
Understanding the mechanistic underpinnings of this transformation is crucial for R&D directors aiming to implement this chemistry on a commercial scale. The reaction is believed to initiate with the oxidative addition of the bromofluoroalkyl compound to the palladium(0) center, generating a fluoroalkyl-palladium(II) species. Subsequent homolytic cleavage or single-electron transfer processes release a fluoroalkyl radical, which adds selectively to the alkyne substrate. The regioselectivity of this radical addition is governed by the stability of the resulting vinyl radical intermediate, which is then trapped by the palladium-boron complex. This sequence ensures that the bulky fluoroalkyl group and the boronate ester end up in a trans-relationship, dictated by the stereoelectronic preferences of the intermediate species. The use of lithium iodide as an additive plays a pivotal role in facilitating the halogen exchange and stabilizing the catalytic cycle, while cesium carbonate acts as a base to promote the transmetallation step with the diboron reagent.
From an impurity control perspective, this mechanism offers distinct advantages. Because the reaction proceeds through a well-defined radical pathway rather than uncontrolled free-radical polymerization, the formation of side products such as homocoupled alkynes or defluorinated species is minimized. The high stereoselectivity means that the crude reaction mixture contains predominantly the desired (Z)-isomer, simplifying the purification process. For quality control teams, this translates to a cleaner impurity profile and higher assay values in the final product. The robustness of the catalytic system against moisture and oxygen, within reasonable limits, further enhances its practicality for large-scale operations where perfect inertness is difficult to maintain. This level of control is essential for producing commercial scale-up of complex polymer additives and fine chemicals where batch-to-batch consistency is paramount.
How To Synthesize Z-Fluoroalkylated Alkenyl Boronates Efficiently
The practical execution of this synthesis is designed to be straightforward, utilizing standard laboratory equipment and commercially sourced reagents. The process begins by charging a reaction vessel with the asymmetric alkyne, the specific bromofluoroalkyl halide, and bis(pinacolato)diboron in a molar ratio optimized for maximum conversion. The palladium catalyst, typically tetrakis(triphenylphosphine)palladium(0), is added along with lithium iodide and cesium carbonate. The reaction is conducted in 1,2-dichloroethane under a nitrogen atmosphere to prevent oxidation of the catalyst. Heating the mixture to 80°C for approximately 10 hours drives the reaction to completion. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during scale-up.
- Combine asymmetric alkyne, bromofluoroalkyl compound, B2pin2, Pd(PPh3)4 catalyst, LiI additive, and Cs2CO3 base in 1,2-dichloroethane solvent under nitrogen.
- Heat the reaction mixture to 80°C and stir for approximately 10 hours to ensure complete conversion to the (Z)-isomer.
- Quench with water, extract with ethyl acetate, wash with brine, dry over Na2SO4, and purify via column chromatography using neutral alumina.
Commercial Advantages For Procurement And Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers tangible strategic benefits beyond mere chemical novelty. The ability to synthesize complex fluorinated intermediates in a single step directly impacts the cost structure of the final API or material. By eliminating the need for multiple synthetic steps, protective group manipulations, and cryogenic conditions, the overall manufacturing footprint is significantly reduced. This consolidation of steps leads to substantial cost savings in terms of raw material consumption, energy usage, and labor hours. Furthermore, the use of stable, shelf-stable reagents like B2pin2 and common palladium catalysts ensures a reliable supply chain, mitigating the risks associated with sourcing exotic or unstable precursors that often plague fluorochemical production.
- Cost Reduction In Manufacturing: The streamlined one-pot nature of this reaction drastically reduces the operational expenses associated with intermediate isolation and purification. Traditional routes often require separate steps for installing the fluorine motif and the boron handle, each incurring yield losses and solvent costs. By merging these into a single catalytic event, the process intensity is lowered, and the throughput is increased. The high atom economy ensures that a greater proportion of the starting materials ends up in the final product, minimizing waste disposal costs and aligning with green chemistry principles. This efficiency allows for more competitive pricing models for high-purity pharmaceutical intermediates without compromising on quality standards.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes to a more resilient supply chain. Unlike processes that require strictly anhydrous conditions or temperatures below -78°C, this method operates at a moderate 80°C in a common chlorinated solvent. This tolerance makes the process easier to transfer between different manufacturing sites and reduces the likelihood of batch failures due to minor environmental fluctuations. The broad substrate scope means that a single platform technology can be used to produce a variety of derivatives, allowing suppliers to respond quickly to changing market demands for different fluorinated scaffolds. This flexibility is crucial for reducing lead time for high-purity pharmaceutical intermediates in a fast-paced development environment.
- Scalability And Environmental Compliance: Scaling this reaction from gram to kilogram scale is facilitated by the homogeneous nature of the catalytic system and the absence of hazardous gaseous reagents. The workup procedure involves standard aqueous quenching and extraction, which are easily adaptable to industrial separations equipment. Moreover, the high selectivity reduces the generation of hazardous fluorinated byproducts, simplifying waste treatment and ensuring compliance with increasingly stringent environmental regulations. The ability to produce these valuable intermediates with a lower environmental impact enhances the sustainability profile of the supply chain, a key metric for modern corporate procurement strategies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and scope defined in the patent documentation, providing a clear understanding of the technology's capabilities and limitations for potential partners.
Q: What is the primary advantage of this trans-carboboration method over traditional cis-addition?
A: This method uniquely achieves trans-fluoroalkylation borylation on asymmetric internal alkynes, which traditionally suffer from poor regioselectivity and predominantly yield cis-products. This allows access to distinct stereoisomers essential for specific biological activities.
Q: Can this process be scaled for industrial pharmaceutical intermediate production?
A: Yes, the reaction utilizes commercially available reagents like Pd(PPh3)4 and operates at moderate temperatures (80°C) in standard solvents like DCE, making it highly amenable to scale-up from gram to multi-kilogram quantities without requiring cryogenic conditions.
Q: What types of fluoroalkyl groups are compatible with this synthesis?
A: The protocol demonstrates broad substrate scope, successfully incorporating ethyl difluoroacetate, difluoroacetylaniline, and various perfluoroalkyl chains, providing versatility for designing diverse fluorinated drug candidates.
Partnering With NINGBO INNO PHARMCHEM: Your Reliable Z-Fluoroalkylated Alkenyl Boronate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced fluorination technologies in accelerating drug discovery and material science. Our team of expert chemists has thoroughly analyzed the methodology described in CN108659028B and is fully prepared to integrate this cutting-edge process into our production capabilities. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless and efficient. Our facilities are equipped with state-of-the-art rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Z-fluoroalkylated alkenyl boronate meets the highest industry standards for potency and impurity profiles.
We invite you to collaborate with us to leverage this innovative chemistry for your specific project needs. Whether you require custom synthesis of novel fluorinated building blocks or large-scale supply of established intermediates, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your current supply chain. We are eager to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our expertise can drive value and efficiency in your upcoming projects.
