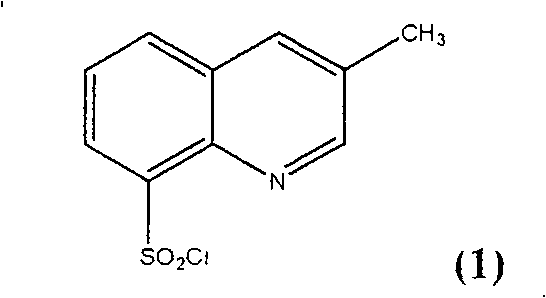
Advanced Synthesis of 3-Methylquinoline-8-Sulfonyl Chloride for High-Purity Argatroban Manufacturing
The pharmaceutical industry's relentless pursuit of high-purity intermediates for anticoagulant therapies has found a significant breakthrough in the methodology detailed in Chinese Patent CN100422154C. This patent discloses a robust and efficient preparation method for 3-methylquinoline-8-sulfonyl chloride, a critical building block in the synthesis of Argatroban, a potent thrombin inhibitor used for treating acute ischemic stroke. Traditional synthetic routes have long struggled with regioselectivity issues, often yielding products contaminated with structural isomers that complicate downstream purification and compromise final drug safety. The innovation presented in this patent addresses these challenges head-on by introducing a specialized sulfonation auxiliary system that dramatically enhances the selectivity for the 8-position over the 5-position on the quinoline ring. By optimizing the molar ratios of chlorosulfonic acid and employing specific inorganic salts, the process achieves conversion rates approaching 100% while maintaining exceptional product quality. This technical advancement not only resolves long-standing purity bottlenecks but also establishes a new benchmark for the reliable pharmaceutical intermediate supplier seeking to secure consistent supply chains for high-value cardiovascular medications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the prevailing technology, exemplified by Japanese Patent JP59-184161, suffered from inherent chemical limitations that rendered it suboptimal for modern GMP manufacturing standards. The conventional direct chlorosulfonation of 3-methylquinoline typically results in a heterogeneous mixture where the desired 8-sulfonyl chloride is accompanied by substantial quantities of the 5-sulfonyl chloride isomer. This lack of regiocontrol manifests physically in the product's appearance; historical batches were frequently described as yellow solids, indicative of oxidative degradation or persistent organic impurities that are difficult to remove. Furthermore, the isolation of the pure 8-isomer from this mixture often requires extensive and costly chromatographic separations or multiple recrystallization cycles, which drastically reduce the overall mass balance and increase solvent waste. For a procurement manager, these inefficiencies translate into higher raw material costs and unpredictable lead times, as the yield of usable API intermediate can fluctuate wildly depending on the batch's isomeric profile. The presence of these impurities also poses a regulatory risk, as strict limits on related substances must be met for any substance intended for human therapeutic use.
The Novel Approach
The methodology outlined in CN100422154C represents a paradigm shift by integrating a sulfonation auxiliary directly into the reaction matrix to steer the electrophilic substitution. By reacting 3-methylquinoline with chlorosulfonic acid in the presence of agents like sodium sulfate or ammonium chloride, the reaction environment is modified to favor attack at the 8-position. This strategic modification effectively suppresses the formation of the 5-isomer, leading to a crude product that is already significantly purer than what was previously achievable. The subsequent treatment with chlorinating agents such as thionyl chloride or phosphorus pentachloride ensures complete conversion of any intermediate sulfonic acids into the desired sulfonyl chloride functionality. The result is an off-white crystalline solid with a sharp melting point of 161-162°C and a purity consistently exceeding 98.5%, as verified by HPLC analysis. This approach simplifies the workflow for the commercial scale-up of complex pharmaceutical intermediates by reducing the reliance on aggressive purification techniques and ensuring a more predictable and stable output quality that aligns with rigorous international pharmacopeia standards.
Mechanistic Insights into Regioselective Chlorosulfonation
The core of this technological advancement lies in the nuanced interaction between the quinoline substrate and the added inorganic auxiliaries during the electrophilic aromatic substitution. In the absence of these auxiliaries, the electron density distribution of the 3-methylquinoline ring allows for competitive attack at both the 5 and 8 positions by the chlorosulfonyl cation. However, the introduction of salts like sodium sulfate likely influences the solvation shell of the reactive species or interacts with the nitrogen atom of the quinoline ring, subtly altering the electronic environment to disfavor the 5-position. This mechanistic control is critical because the 5-isomer is structurally very similar to the target molecule, making separation via standard physical methods energetically expensive and chemically inefficient. The patent data indicates that by fine-tuning the molar ratio of the auxiliary to the substrate (between 0.01 and 0.15), the reaction kinetics are optimized to maximize the formation of the 8-sulfonic acid/sulfonyl chloride precursor. This level of control demonstrates a sophisticated understanding of physical organic chemistry applied to process development, ensuring that the high-purity pharmaceutical intermediate is generated at the molecular level rather than just purified at the end of the line.
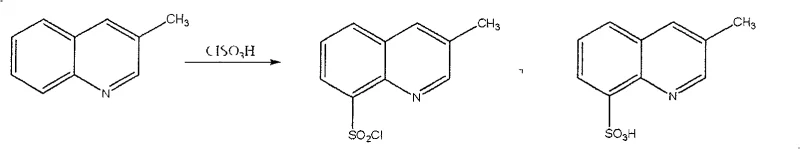
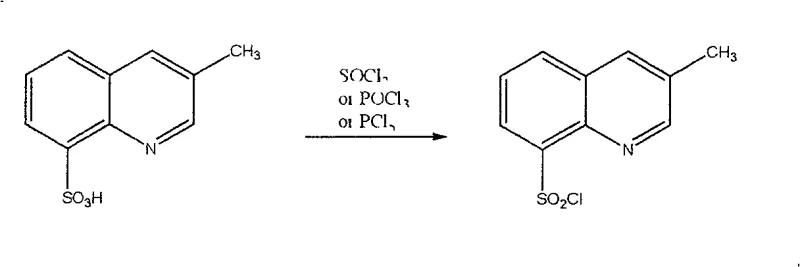
Following the initial substitution, the second stage of the mechanism involves the activation of the sulfonic acid moiety. The patent describes a sequential addition where the sulfonation mixture is treated with a chlorinating agent like thionyl chloride (SOCl2) or phosphorus pentachloride (PCl5). This step is vital because the initial reaction with chlorosulfonic acid often produces a mixture of the free sulfonic acid and the sulfonyl chloride. The chlorinating agent serves to convert the less reactive sulfonic acid groups into the highly reactive sulfonyl chloride functionality, which is essential for the subsequent coupling reactions in Argatroban synthesis. The choice of chlorinating agent offers flexibility; for instance, using PCl5 might be preferred in certain solvent systems, while SOCl2 generates gaseous byproducts (SO2 and HCl) that can be easily vented, driving the equilibrium forward. This two-stage mechanism ensures that virtually all sulfur-containing species on the ring are converted to the desired functional group, minimizing waste and maximizing the atom economy of the process. Such mechanistic clarity provides R&D teams with the confidence to troubleshoot and optimize the process further if specific scale-up challenges arise.
How to Synthesize 3-Methylquinoline-8-Sulfonyl Chloride Efficiently
The synthesis protocol defined in the patent offers a streamlined pathway that balances reaction severity with product integrity. The process begins with the careful addition of 3-methylquinoline to chlorosulfonic acid under cooling, followed by the introduction of the selected auxiliary salt. The mixture is then heated to promote the substitution reaction before being cooled for the chlorination step. This sequence is designed to manage the exothermic nature of sulfonation while ensuring complete conversion. The detailed operational parameters, including specific temperature ramps from 25°C to 120°C and precise molar equivalents, are critical for reproducing the high yields reported in the examples. For laboratory and pilot plant chemists, adhering to these thermal profiles is essential to prevent thermal degradation or the formation of tars. The following guide summarizes the standardized steps derived from the patent's preferred embodiments to assist in the rapid implementation of this superior synthetic route.
- React 3-methylquinoline with chlorosulfonic acid and a sulfonation auxiliary (e.g., sodium sulfate) at controlled temperatures to form a sulfonated mixture.
- Treat the resulting mixture with a chlorinating agent such as thionyl chloride or phosphorus pentachloride to convert sulfonic acids to sulfonyl chlorides.
- Quench the reaction in ice water, filter the precipitate, and recrystallize from toluene to obtain off-white crystals with purity exceeding 98.5%.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented methodology offers profound advantages that extend well beyond simple chemical yield improvements. For procurement professionals managing the budget for API production, the ability to source an intermediate with inherently higher purity reduces the burden on quality control laboratories and minimizes the risk of batch rejection. The elimination of the yellow discoloration and the significant reduction in isomeric impurities mean that downstream processing becomes more predictable and less resource-intensive. This stability in raw material quality is a key factor in cost reduction in pharmaceutical intermediate manufacturing, as it allows for tighter inventory control and reduces the need for safety stock to account for variable quality. Furthermore, the use of commodity chemicals like sodium sulfate and ammonium chloride as auxiliaries ensures that the supply chain remains resilient against fluctuations in the availability of exotic reagents. This reliance on ubiquitous industrial chemicals mitigates supply risk and stabilizes pricing structures over the long term.
- Cost Reduction in Manufacturing: The primary driver for cost efficiency in this process is the dramatic improvement in yield and the simplification of purification. By achieving yields of up to 94% after recrystallization compared to roughly 42% in older comparative methods, the amount of starting material required per kilogram of final product is effectively halved. This massive gain in material efficiency directly lowers the cost of goods sold (COGS). Additionally, because the crude product is of higher quality (off-white vs. yellow), the solvent consumption for recrystallization is optimized, and the loss of product during mother liquor disposal is minimized. The removal of complex purification steps also reduces energy consumption and labor hours, contributing to a leaner and more profitable manufacturing operation without compromising on the stringent quality requirements of the pharmaceutical sector.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by processes that rely on sensitive catalysts or hard-to-source reagents. This method circumvents those risks by utilizing robust, shelf-stable reagents like chlorosulfonic acid and common inorganic salts. The process tolerance is also improved; the patent demonstrates that the reaction works effectively with a range of chlorinating agents (SOCl2, POCl3, PCl5), giving manufacturers the flexibility to switch reagents based on market availability or price without revalidating the entire process. This adaptability is crucial for maintaining reducing lead time for high-purity pharmaceutical intermediates, as production schedules are less likely to be disrupted by single-source reagent shortages. The straightforward workup procedure involving ice-water quenching and filtration is easily scalable and does not require specialized equipment, further enhancing the reliability of supply from contract manufacturing organizations.
- Scalability and Environmental Compliance: As regulatory pressure mounts regarding waste disposal and environmental impact, the cleaner profile of this synthesis offers a distinct advantage. The higher selectivity means fewer byproducts are generated, reducing the load on wastewater treatment facilities. The ability to produce off-white crystals directly suggests fewer oxidative side reactions, which often generate difficult-to-treat organic sludge. Moreover, the process is amenable to large-scale batch reactors, as the thermal profile (heating to 120°C) is well within the capabilities of standard glass-lined steel reactors used in the fine chemical industry. This ease of commercial scale-up ensures that as demand for Argatroban grows, the supply of its key intermediate can be ramped up quickly to meet market needs without requiring capital-intensive new infrastructure or facing significant regulatory hurdles related to process safety or emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 3-methylquinoline-8-sulfonyl chloride. These answers are derived directly from the experimental data and claims within the patent documentation, providing a transparent view of the technology's capabilities. Understanding these details is essential for technical buyers evaluating the feasibility of integrating this intermediate into their existing supply chains. The focus is on practical implementation, quality metrics, and the specific advantages this route holds over legacy methods.
Q: How does this method improve upon the traditional Japanese patent JP59-184161?
A: Unlike the prior art which yields yellow products contaminated with significant amounts of the 3-methylquinoline-5-sulfonyl chloride isomer, this novel method utilizes sulfonation auxiliaries to enhance regioselectivity, resulting in off-white crystals with purity greater than 98.5% and significantly reduced isomeric impurities.
Q: What specific auxiliaries are used to control the reaction selectivity?
A: The process employs inorganic salts such as sodium sulfate, ammonium sulfate, or ammonium chloride as sulfonation auxiliaries. These additives play a critical role in directing the sulfonyl group to the 8-position of the quinoline ring, thereby suppressing the formation of the unwanted 5-isomer.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the method is designed for scalability. It uses readily available raw materials like chlorosulfonic acid and common inorganic salts, operates under manageable temperature conditions (25°C to 120°C), and avoids complex catalytic systems, making it highly conducive to industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Methylquinoline-8-Sulfonyl Chloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the quality of the final API is inextricably linked to the purity of its precursors. Leveraging the advanced synthetic strategies outlined in patents like CN100422154C, we have established ourselves as a leader in the production of high-value heterocyclic intermediates. Our facility is equipped to handle the specific thermal and corrosive requirements of chlorosulfonation chemistry safely and efficiently. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of material regardless of market volume fluctuations. Our commitment to quality is backed by stringent purity specifications and rigorous QC labs that utilize state-of-the-art HPLC and NMR instrumentation to verify every batch against the highest international standards.
We invite pharmaceutical manufacturers and procurement specialists to collaborate with us to optimize their Argatroban supply chains. By partnering with our technical team, you can access a Customized Cost-Saving Analysis that quantifies the potential efficiencies of switching to our high-purity grade material. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments tailored to your project's unique requirements. Let us help you secure a competitive advantage through superior chemistry and reliable supply.
