Revolutionizing Baloxavir Intermediate Production via Cyclic Chiral Resolution Technology
Revolutionizing Baloxavir Intermediate Production via Cyclic Chiral Resolution Technology
The global demand for antiviral therapeutics has placed immense pressure on the supply chains of critical active pharmaceutical ingredients (APIs), particularly for next-generation influenza treatments like Baloxavir marboxil. A pivotal breakthrough in addressing the manufacturing bottlenecks of this drug is detailed in patent CN114014874A, which discloses a highly efficient preparation method for a key chiral intermediate. This technology fundamentally shifts the paradigm from linear, waste-generating resolution processes to a circular, near-quantitative conversion system. By integrating chiral resolution with a robust recycling mechanism for the mother liquor, this invention offers a compelling solution for pharmaceutical manufacturers seeking to optimize both purity and throughput. The strategic implementation of this cyclic resolution pathway not only enhances the economic viability of producing the Formula I intermediate but also aligns with modern green chemistry principles by drastically minimizing chemical waste.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral intermediates for complex heterocyclic drugs like Baloxavir has been plagued by the inherent inefficiencies of classical resolution techniques. In standard procedures, a racemic mixture is reacted with a chiral resolving agent to form diastereomers, which are then separated via crystallization. However, the fundamental flaw lies in the fate of the mother liquor; traditionally, the undesired enantiomer or diastereomer remaining in the solution is discarded or requires complex, low-yield racemization steps to be reused. Literature indicates that conventional resolution of the Formula II racemate typically yields only about 40% of the desired single-configuration compound, leaving approximately 60% of the valuable starting material trapped in the waste stream. This not only inflates the cost of goods sold (COGS) due to raw material loss but also creates significant environmental burdens associated with the disposal of chiral chemical waste, making the process unsustainable for large-scale commercial API manufacturing.
The Novel Approach
The methodology presented in patent CN114014874A introduces a transformative closed-loop system that effectively eliminates the concept of "waste" in the resolution step. Instead of discarding the mother liquor containing the mixture of compounds B and C (or pure C), the process subjects this filtrate to a chemical transformation that regenerates the racemic starting material, Formula II. This regenerated racemate is then fed back into the resolution cycle. Through this iterative looping, the theoretical conversion efficiency of the starting material to the desired Formula I intermediate approaches 100%. This approach decouples the yield of a single crystallization step from the overall process yield, allowing manufacturers to utilize nearly every gram of the initial racemic feedstock. The result is a streamlined workflow that maximizes asset utilization and significantly reduces the raw material intensity required per kilogram of final API produced.
Mechanistic Insights into Chiral Resolution and Recycling
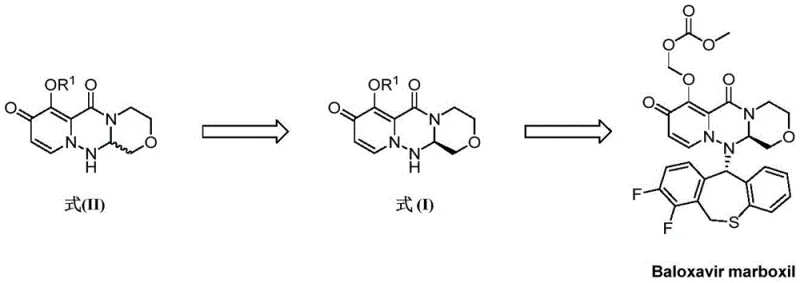
The core of this technology relies on the precise manipulation of diastereomeric interactions and selective hydrolysis. Initially, the racemic Formula II is condensed with a chiral acid, such as (R)-tetrahydrofuran-2-carboxylic acid, using coupling agents like T3P or HATU. This forms a pair of diastereomeric amides (Compound A), which exhibit different solubility profiles in solvents like ethyl acetate or ethanol. Upon recrystallization, the less soluble diastereomer (Compound B) precipitates as a high-purity filter cake, while the more soluble isomer (Compound C) remains in the supernatant. The critical innovation occurs in the subsequent treatment of these fractions. The filter cake B undergoes mild hydrolysis, often catalyzed by organic bases like DBU or inorganic bases like potassium carbonate, to cleave the chiral auxiliary and release the target Formula I intermediate with high enantiomeric excess (ee > 99%).
Simultaneously, the mother liquor, rich in the unwanted isomer C, is not discarded but treated under basic conditions to hydrolyze the amide bond, regenerating the free amine Formua II. This regeneration step is chemically elegant as it restores the racemic nature of the molecule, making it indistinguishable from fresh starting material and ready for re-entry into the condensation step. The patent details specific conditions, such as heating at 50 to 100°C in solvents like ethanol or DMF, to ensure complete conversion without degrading the sensitive triazine-oxazine scaffold. This mechanistic understanding allows process chemists to fine-tune the equilibrium between resolution and recycling, ensuring that impurities do not build up over multiple cycles and that the optical purity of the final product remains within stringent pharmacopeial limits.
How to Synthesize Baloxavir Key Intermediate Efficiently
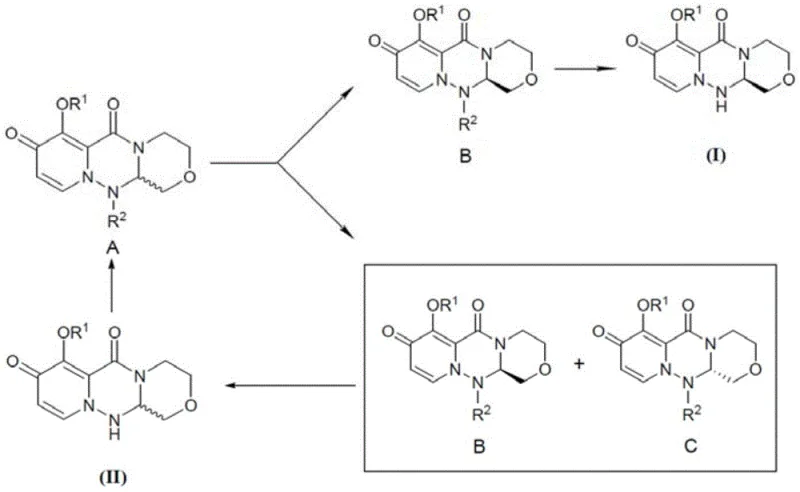
Implementing this cyclic resolution strategy requires careful control of reaction parameters to maintain high throughput and purity. The process begins with the activation of the chiral acid and its coupling to the racemic amine, followed by a controlled crystallization to maximize the recovery of the desired diastereomer. The true power of the method is realized in the recycling phase, where the mother liquor is processed to recover the starting material. This requires robust analytical monitoring to ensure that the recycled material meets the quality standards for the next resolution cycle. The following guide outlines the standardized operational framework derived from the patent examples, providing a roadmap for scaling this technology from laboratory benchtop to commercial production vessels.
- Condense racemic Formula II with a chiral acid (e.g., tetrahydrofuran-2-carboxylic acid) using a coupling agent to form diastereomeric mixture A.
- Recrystallize mixture A to separate the desired diastereomer B (filter cake) from the mother liquor containing B and C.
- Hydrolyze filter cake B to obtain pure Formula I, while chemically converting the mother liquor components back to racemic Formula II for re-processing.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this cyclic resolution technology translates directly into enhanced supply security and reduced operational risk. Traditional linear resolution processes are inherently vulnerable to fluctuations in raw material pricing because they consume nearly double the amount of starting material to produce the same amount of chiral product. By contrast, this novel approach drastically improves atom economy, meaning that the demand for expensive chiral precursors and racemic starting materials is significantly lowered. This reduction in material intensity acts as a buffer against market volatility, ensuring more stable pricing structures for long-term supply agreements. Furthermore, the simplification of the waste stream reduces the logistical complexity and cost associated with hazardous waste disposal, contributing to a leaner and more agile manufacturing operation.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the near-elimination of material loss during the chiral separation phase. In conventional methods, losing over half of the input material to the mother liquor represents a direct financial loss that must be absorbed by the final product price. By recycling the mother liquor components back to the racemic starting material, the process effectively converts what was once waste into valuable feedstock. This circular economy model within the reactor vessel leads to substantial cost savings in raw material procurement. Additionally, the use of common, non-proprietary reagents like potassium carbonate and DBU avoids the premium costs associated with specialized enzymatic or transition-metal catalysts, further driving down the variable cost of production.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the complexity of multi-step syntheses that rely on low-yield purification steps. This method enhances reliability by decoupling the single-pass yield from the overall process yield. Even if a single crystallization run yields only 40% of the target, the recycling loop ensures that the remaining 60% is not lost but recovered for future batches. This creates a more robust production schedule that is less susceptible to batch failures or yield dips. Moreover, the ability to stockpile and recycle intermediates provides a strategic buffer, allowing manufacturers to maintain consistent output levels even when facing temporary shortages of fresh starting materials, thereby securing the supply chain for downstream API formulation.
- Scalability and Environmental Compliance: From an operational perspective, the unit operations involved—condensation, crystallization, filtration, and hydrolysis—are well-understood and easily scalable from pilot plants to multi-ton reactors. The process avoids the use of heavy metals or cryogenic conditions, which often pose engineering challenges at scale. Environmentally, the drastic reduction in chemical waste generation aligns with increasingly strict global regulations on pharmaceutical effluent. By minimizing the volume of solvent and chemical waste requiring treatment, manufacturers can reduce their environmental footprint and compliance costs. This sustainability advantage is becoming a critical factor in vendor selection for major pharmaceutical companies committed to green chemistry initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this cyclic resolution technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on the feasibility and advantages of the method for potential partners and licensees.
Q: How does this patent improve the yield of Baloxavir intermediate compared to traditional methods?
A: Traditional resolution methods typically discard the mother liquor, resulting in a maximum theoretical yield of around 40-50%. This patent introduces a recycling loop where the mother liquor (containing unwanted isomers) is chemically converted back into the racemic starting material (Formula II), allowing for repeated resolution cycles that theoretically approach 100% conversion efficiency.
Q: What specific chiral resolving agents are compatible with this process?
A: The patent specifies several effective chiral acids, including (R)-tetrahydrofuran-2-carboxylic acid, (S)-tetrahydrofuran-2-carboxylic acid, L-proline, D-proline, and their derivatives like N-p-toluenesulfonyl-L-proline. The choice depends on the specific crystallization properties required for the target diastereomer.
Q: Is this process scalable for commercial API manufacturing?
A: Yes, the process relies on standard unit operations such as condensation, recrystallization, filtration, and hydrolysis. The use of common solvents like ethyl acetate, ethanol, and DMF, along with inexpensive bases like potassium carbonate or DBU, makes it highly suitable for large-scale commercial production without requiring exotic catalysts or extreme conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Baloxavir Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and robust infrastructure. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex chiral resolution processes like the one described in CN114014874A are executed with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of monitoring enantiomeric purity at every stage of the cycle, guaranteeing that the final intermediates meet stringent purity specifications required for global regulatory filings. We are committed to delivering high-quality pharmaceutical intermediates that empower our clients to bring life-saving medications to market faster and more efficiently.
We invite procurement leaders and R&D directors to engage with our technical team to explore how this advanced resolution technology can be integrated into your supply chain. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the specific economic benefits of implementing this recycling loop for your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our manufacturing capabilities can drive value and reliability for your Baloxavir marboxil projects.
