Advanced Synthesis of 5-Trifluoromethyl Imidazoles for Pharmaceutical Intermediates Manufacturing
Advanced Synthesis of 5-Trifluoromethyl Imidazoles for Pharmaceutical Intermediates Manufacturing
The landscape of modern pharmaceutical synthesis is constantly evolving, driven by the need for more efficient, cost-effective, and scalable routes to complex heterocyclic scaffolds. A significant breakthrough in this domain is detailed in patent CN113735778A, which discloses a novel preparation method for 5-trifluoromethyl substituted imidazole compounds. These structures are not merely academic curiosities; they serve as critical building blocks for a wide array of bioactive molecules, including renowned drugs like Metronidazole and Losartan. The introduction of a trifluoromethyl group is particularly strategic, as it profoundly enhances the metabolic stability, lipophilicity, and bioavailability of the parent molecule, properties that are highly coveted by medicinal chemists during lead optimization phases. This patent presents a robust methodology that leverages a transition metal silver oxide promoted [3+2] cycloaddition reaction, offering a streamlined alternative to traditional synthetic pathways that often suffer from harsh conditions or expensive reagents.
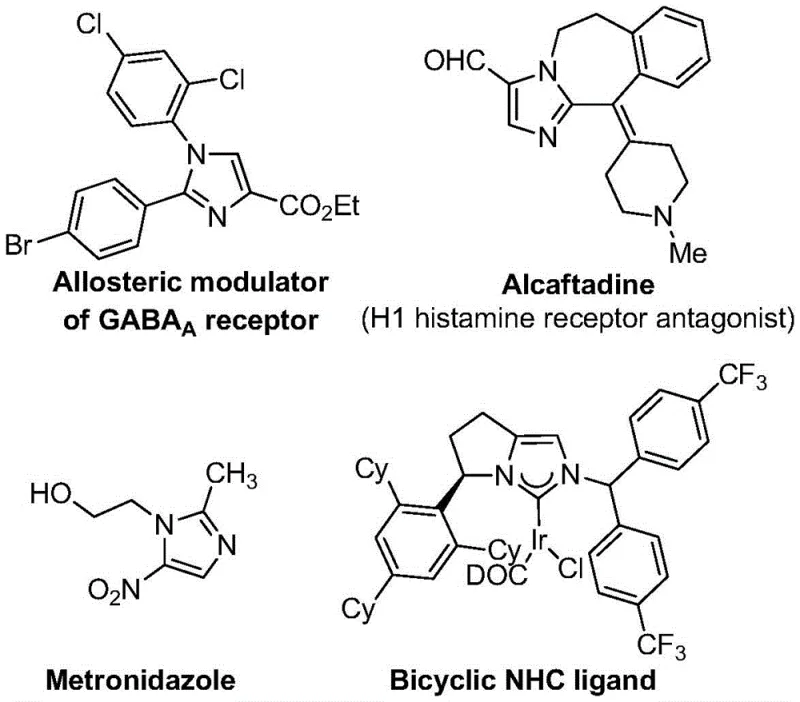
The strategic importance of this technology cannot be overstated for stakeholders in the fine chemical industry. For R&D directors, the ability to access diverse, fully substituted imidazole libraries with high purity is essential for accelerating drug discovery pipelines. For procurement managers, the shift towards using cheap and easily obtainable starting materials represents a tangible opportunity for cost reduction in API manufacturing. Furthermore, supply chain heads will find value in the method's operational simplicity and its potential for seamless scale-up, ensuring a reliable pharmaceutical intermediate supplier can maintain continuity of supply even during periods of high demand. By addressing the limitations of prior art, this invention paves the way for the commercial scale-up of complex polymer additives and electronic chemicals that rely on similar heterocyclic cores, although its primary impact remains in the life sciences sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted imidazoles has been fraught with challenges that hindered their widespread adoption in industrial settings. Traditional literature methods primarily relied on [3+2] cycloaddition reactions between methylene amine ylides and trifluoromethyl-substituted imines. While chemically sound, these approaches often necessitated the use of expensive and unstable synthons, such as trifluoro-acetaldehyde ethyl hemiacetal compounds. The reliance on such specialized reagents not only inflated the raw material costs but also introduced significant logistical complexities regarding storage and handling, thereby limiting the scale application of these processes. Additionally, many conventional routes required harsh reaction conditions or multiple protection-deprotection steps, which inevitably led to lower overall yields and generated substantial amounts of chemical waste, conflicting with modern green chemistry principles.
The Novel Approach
In stark contrast, the methodology described in CN113735778A revolutionizes this synthetic challenge by utilizing trifluoroethylimidoyl chloride and imidoesters as the foundational building blocks. This novel approach capitalizes on the high reactivity of these precursors under the promotion of silver oxide, facilitating a direct and efficient construction of the imidazole ring. The reaction proceeds under remarkably mild conditions, typically requiring temperatures between 40°C and 80°C, which significantly reduces energy consumption compared to high-temperature alternatives. Moreover, the use of inexpensive additives like sodium carbonate and common organic solvents such as acetonitrile further enhances the economic viability of the process. This shift allows for the synthesis of a diverse range of 1,2,4-substituted fully-substituted imidazole compounds with exceptional flexibility, enabling chemists to tailor the molecular architecture to specific biological targets without being constrained by synthetic feasibility.
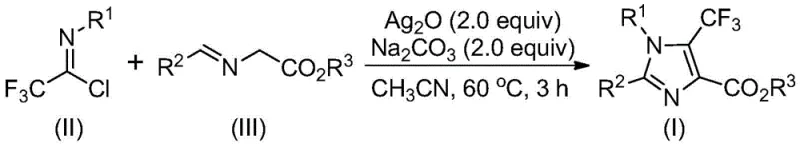
Mechanistic Insights into Silver-Promoted [3+2] Cycloaddition
To fully appreciate the elegance of this transformation, one must delve into the mechanistic intricacies that govern the reaction pathway. The process initiates with an alkali-promoted intermolecular carbon-carbon bond formation between the trifluoroethylimidoyl chloride and the imidate substrate. This initial coupling generates a bisimine intermediate, which serves as the precursor for the subsequent cyclization event. The presence of silver oxide is pivotal here; it does not merely act as a passive base but actively promotes the intramolecular cyclization of the bisimine species to form a 2-hydroimidazole compound. This step is critical for establishing the five-membered heterocyclic core with the correct regiochemistry, ensuring that the trifluoromethyl group is positioned precisely at the 5-position of the imidazole ring.
Following the cyclization, the reaction undergoes a final oxidative aromatization step, also facilitated by the silver oxide promoter. This oxidative dehydrogenation converts the dihydro-intermediate into the fully aromatic imidazole system, driving the reaction to completion and stabilizing the final product. From an impurity control perspective, this mechanism is highly advantageous because the silver-mediated pathway tends to be selective, minimizing the formation of side products that often plague radical-based or acid-catalyzed alternatives. The mild nature of the conditions preserves sensitive functional groups on the aryl rings, such as halogens or esters, allowing for the synthesis of complex derivatives like those shown in the patent examples without degradation. This high level of chemoselectivity is paramount for producing high-purity OLED material or pharmaceutical intermediates where trace impurities can have detrimental effects on downstream performance.
How to Synthesize 5-Trifluoromethyl Imidazoles Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to stoichiometry and reaction parameters to maximize yield and purity. The patent outlines a straightforward protocol where the molar ratio of the key components—trifluoroethylimidoyl chloride, imidate, and silver oxide—is optimized to ensure complete conversion while minimizing excess reagent waste. Typically, a slight excess of the imidate and silver oxide is employed to drive the equilibrium forward. The choice of solvent is also critical; while various aprotic solvents can support the reaction, acetonitrile has been identified as the superior medium due to its ability to effectively dissolve the starting materials and promote the reaction kinetics. The following guide summarizes the standardized operational procedure derived from the patent data.
- Combine trifluoroethylimidoyl chloride, imidate, silver oxide promoter, and sodium carbonate additive in an organic solvent such as acetonitrile.
- Heat the reaction mixture to a temperature between 40°C and 80°C and stir for 2 to 4 hours to ensure complete conversion.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the final 5-trifluoromethyl substituted imidazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this synthetic route offers compelling economic and operational benefits that extend beyond simple yield metrics. The fundamental shift towards using commodity chemicals as starting materials drastically simplifies the sourcing process, reducing dependency on niche suppliers who might dictate pricing or lead times. By eliminating the need for exotic or hazardous reagents, companies can also reduce their regulatory burden and insurance costs associated with handling dangerous goods. Furthermore, the simplified workup procedure, which involves basic filtration and standard chromatography, lowers the barrier for technology transfer from R&D to manufacturing, accelerating the time-to-market for new drug candidates.
- Cost Reduction in Manufacturing: The economic argument for this process is robust, primarily driven by the substitution of expensive trifluoro-acetaldehyde derivatives with cheap trifluoroethylimidoyl chloride. This raw material swap results in substantial cost savings per kilogram of produced intermediate. Additionally, the use of silver oxide, while a metal salt, is relatively inexpensive compared to noble metal catalysts like palladium or platinum, and it serves a dual purpose as both promoter and oxidant, effectively consolidating two reagent costs into one. The high reaction efficiency reported in the patent implies that less raw material is wasted on side reactions, further optimizing the atom economy and reducing the cost of goods sold (COGS) for the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the commercial availability of the requisite aldehydes, glycines, and amines used to prepare the precursors. Since these are bulk chemicals produced on a massive global scale, the risk of supply disruption is minimal compared to relying on custom-synthesized specialty reagents. The robustness of the reaction conditions also means that the process is less susceptible to variations in utility quality or minor fluctuations in environmental controls, ensuring consistent batch-to-batch quality. This reliability is crucial for maintaining the continuity of supply for critical medications, preventing stockouts that could impact patient health and company revenue.
- Scalability and Environmental Compliance: From an environmental and scalability standpoint, this method aligns well with modern sustainability goals. The reaction operates at moderate temperatures, reducing the energy footprint associated with heating and cooling large reactors. The post-treatment process is straightforward, involving filtration and silica gel mixing, which avoids the generation of complex aqueous waste streams that require expensive treatment. The ability to scale this reaction from gram-level experiments to multi-kilogram production without significant re-optimization demonstrates its maturity and readiness for industrial application. This scalability ensures that as demand for the final drug product grows, the supply of the intermediate can be ramped up seamlessly to meet market needs.
Frequently Asked Questions (FAQ)
Understanding the nuances of this technology is vital for stakeholders evaluating its potential integration into their existing portfolios. The following questions address common inquiries regarding the practical application, scope, and benefits of this patented synthesis method. These answers are derived directly from the technical disclosures and experimental data provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: What are the primary advantages of using silver oxide in this synthesis?
A: Silver oxide acts as both a promoter for the intermolecular carbon-carbon bond formation and an oxidant for the final aromatization step, eliminating the need for separate oxidation reagents and simplifying the workflow.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the method utilizes cheap and readily available starting materials like glycine derivatives and operates under mild conditions (40-80°C), making it highly amenable to scale-up from gram to multi-kilogram levels.
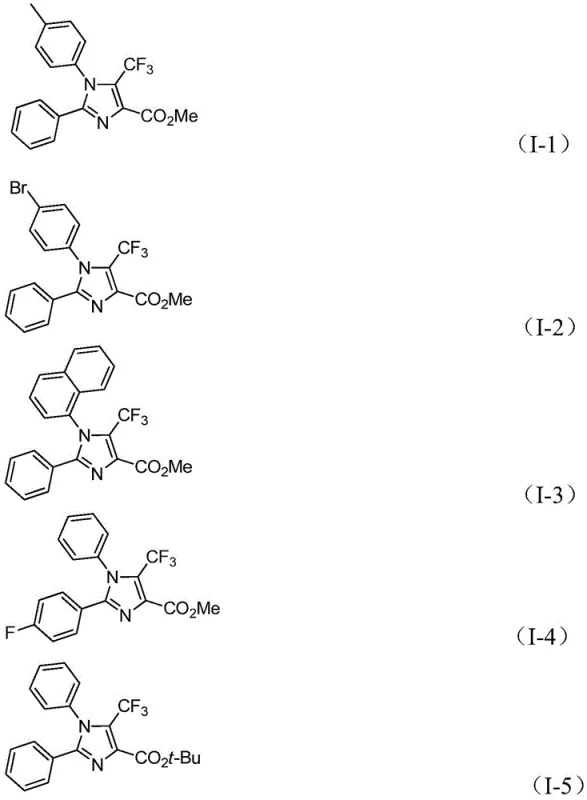
Q: What is the substrate scope for the R1 and R2 groups?
A: The process exhibits excellent functional group tolerance, accommodating various substituted aryl groups including those with methyl, tert-butyl, halogen, or trifluoromethyl substituents at ortho, meta, or para positions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl Imidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic methodologies like the one described in CN113735778A. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and high-quality supply of critical intermediates. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 5-trifluoromethyl imidazole delivered meets the exacting standards required for pharmaceutical applications. We are committed to leveraging our technical expertise to optimize these processes further, delivering value through efficiency and reliability.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this method for your supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that will drive your drug development programs forward with confidence and speed.
