Advanced Synthesis of Benzo Pyridylimidazo Isoindole Diketones for Commercial Pharmaceutical Applications
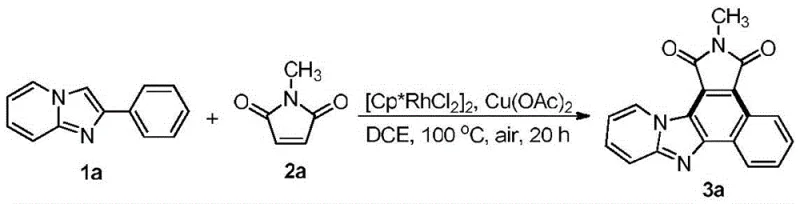
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex fused heterocyclic systems, which serve as critical scaffolds for next-generation therapeutics and advanced functional materials. Patent CN112174962A introduces a groundbreaking synthesis method for benzo[e]pyridylimidazo[4,5-g]isoindole-1,3(2H)-dione compounds, representing a significant leap forward in organic synthesis technology. This novel approach leverages a transition metal-catalyzed [4+2] oxidative cyclization reaction to directly fuse pyridoimidazole and maleimide structural units into a pentacyclic aromatic system. For R&D Directors and Procurement Managers alike, this technology offers a pathway to access high-value chemical entities that were previously difficult or costly to manufacture. The ability to construct such intricate nitrogen-containing fused heterocycles in a single operational step not only streamlines the synthetic route but also enhances the overall atom economy of the process. As a reliable pharmaceutical intermediates supplier, understanding the nuances of such patented technologies allows us to offer superior supply chain solutions that align with the rigorous demands of modern drug discovery and material science applications.
Furthermore, the strategic importance of this synthesis lies in its potential application across multiple high-growth sectors, including medicinal chemistry for antiviral and anticancer agents, as well as the development of fluorescent dyes and optoelectronic materials. The core innovation described in the patent eliminates the need for pre-functionalized starting materials that often require tedious preparation steps, thereby reducing the cumulative waste and energy consumption associated with traditional multi-step syntheses. By adopting this methodology, manufacturers can achieve cost reduction in fine chemical manufacturing through simplified processing and improved yield profiles. The direct formation of the pentacyclic core from readily available precursors underscores a shift towards more sustainable and efficient chemical production paradigms. This report delves deep into the mechanistic underpinnings and commercial implications of this technology, providing actionable insights for stakeholders aiming to secure a competitive advantage in the global market for specialized organic intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of complex fused heterocyclic systems like benzo[e]pyridylimidazo isoindole diketones has been plagued by significant synthetic challenges that hinder large-scale adoption. Conventional routes often rely on stepwise condensation reactions that require harsh conditions, such as strong acids or high temperatures, which can lead to the decomposition of sensitive functional groups and the formation of numerous by-products. These multi-step sequences typically necessitate the isolation and purification of unstable intermediates, resulting in substantial material loss and increased operational costs. Moreover, the use of stoichiometric amounts of hazardous reagents in older methodologies poses serious environmental and safety concerns, complicating waste disposal and regulatory compliance. For supply chain heads, these inefficiencies translate into longer lead times and higher volatility in raw material pricing, making it difficult to guarantee consistent supply for commercial scale-up of complex polymer additives or pharmaceutical ingredients. The lack of regioselectivity in some traditional cyclization methods further exacerbates the issue, requiring extensive chromatographic purification that is impractical for industrial tonnage production.
The Novel Approach
In stark contrast, the novel approach detailed in patent CN112174962A utilizes a sophisticated transition metal-catalyzed [4+2] oxidative cyclization strategy that fundamentally reshapes the synthetic landscape for these compounds. This method enables the direct coupling of 2-aryl-imidazo[1,2-a]pyridine derivatives with N-substituted maleimides under relatively mild conditions, typically ranging from 80°C to 120°C. The use of a rhodium catalyst facilitates precise C-H activation and bond formation, ensuring high regioselectivity and minimizing the generation of unwanted isomers. This one-pot transformation effectively bypasses the need for intermediate isolation, thereby drastically simplifying the workflow and reducing the overall process time. For a reliable agrochemical intermediate supplier or pharma partner, this efficiency is paramount, as it allows for rapid iteration during the development phase and smoother translation to manufacturing. The robustness of this catalytic system means that it can tolerate a wide array of substrates, providing the flexibility needed to explore diverse chemical space without redesigning the entire synthetic route for each new analog.
![General reaction scheme showing the Rh-catalyzed oxidative cyclization of 2-aryl-imidazo[1,2-a]pyridine and N-substituted maleimide to form the pentacyclic product](/insights/img/benzo-pyridylimidazo-isoindole-synthesis-pharma-supplier-20260303151603-01.webp)
Mechanistic Insights into Rhodium-Catalyzed Oxidative Cyclization
The heart of this technological breakthrough lies in the intricate catalytic cycle driven by the dichloro(pentamethylcyclopentadienyl)rhodium(III) dimer complex. Mechanistically, the reaction initiates with the coordination of the rhodium catalyst to the nitrogen atom of the imidazopyridine substrate, directing the metal center to the adjacent C-H bond for activation. This C-H metallation step is crucial as it generates a reactive organometallic intermediate that is poised for subsequent insertion into the maleimide double bond. The presence of the oxidant, such as copper acetate, plays a dual role: it regenerates the active Rh(III) species from the reduced Rh(I) state formed after reductive elimination, and it facilitates the oxidative aromatization required to finalize the fused ring system. Understanding this mechanism is vital for R&D teams aiming to optimize reaction parameters, as subtle changes in ligand environment or oxidant strength can significantly impact turnover numbers. The elegance of this mechanism ensures that the reaction proceeds with high fidelity, preserving the integrity of sensitive substituents on both the pyridine and maleimide rings while constructing the new carbon-carbon bonds necessary for the pentacyclic framework.
From an impurity control perspective, this catalytic pathway offers distinct advantages over radical-based or thermal cyclization methods. The directed nature of the C-H activation minimizes random polymerization or oligomerization side reactions that often plague free-radical processes. Furthermore, the mild reaction temperatures of 80-120°C prevent thermal degradation of the product, which is particularly important for maintaining high-purity OLED material or API intermediate standards. The choice of solvent, with 1,2-dichloroethane (DCE) demonstrating superior performance in the patent examples, further stabilizes the transition states involved in the cyclization. By controlling the stoichiometry of the oxidant and catalyst, manufacturers can fine-tune the reaction to maximize yield while minimizing metal residue in the final product. This level of control is essential for meeting the stringent purity specifications required by global regulatory bodies, ensuring that the final chemical entity is suitable for downstream applications in human health or advanced electronics without extensive remediation.
How to Synthesize Benzo Pyridylimidazo Isoindole Diketone Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to ensure reproducibility and high yield on a commercial scale. The patent outlines a generalized procedure where the key reactants are dissolved in a suitable solvent, followed by the sequential addition of the rhodium catalyst and the copper oxidant. It is imperative to maintain an inert atmosphere or controlled air exposure as specified, since the oxidative nature of the reaction relies on the precise balance of oxygen sources provided by the oxidant and the environment. The reaction mixture is then heated in a sealed pressure tube to maintain solvent integrity and pressure at elevated temperatures, typically around 100°C, for a duration of approximately 20 hours. Post-reaction workup involves standard filtration through celite to remove metal salts and catalyst residues, followed by concentration and purification via silica gel column chromatography.
- Dissolve 2-aryl-imidazo[1,2-a]pyridine and N-substituted maleimide in a solvent such as 1,2-dichloroethane.
- Add dichloro(pentamethylcyclopentadienyl)rhodium(III) dimer catalyst and copper acetate oxidant to the reaction mixture.
- Heat the sealed pressure tube at 80-120°C for approximately 20 hours, then isolate the product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented synthesis method translates into tangible strategic benefits that extend beyond mere technical feasibility. The primary advantage lies in the significant simplification of the supply chain, as the starting materials—2-aryl-imidazo[1,2-a]pyridines and N-substituted maleimides—are commercially available and relatively inexpensive compared to pre-functionalized precursors required by older methods. This accessibility reduces the risk of supply disruption and allows for more flexible sourcing strategies, enhancing the overall resilience of the manufacturing operation. Additionally, the one-pot nature of the reaction eliminates multiple unit operations such as intermediate drying, weighing, and transfer, which directly correlates to reduced labor costs and lower energy consumption per kilogram of product. By streamlining the process flow, companies can achieve substantial cost savings without compromising on the quality or purity of the final output, making it an attractive option for cost-sensitive markets.
- Cost Reduction in Manufacturing: The elimination of transition metal removal steps often required in other catalytic processes, combined with the high atom economy of the [4+2] cycloaddition, leads to a more economical production model. Since the reaction proceeds with high efficiency and minimal by-product formation, the burden on downstream purification is significantly lessened, reducing the volume of solvents and silica gel required for chromatography. This reduction in consumable usage directly impacts the bottom line, allowing for more competitive pricing structures in the global market. Furthermore, the ability to recycle or recover the solvent system adds another layer of financial efficiency, ensuring that the process remains viable even when raw material prices fluctuate. The overall process design prioritizes resource efficiency, aligning with modern green chemistry principles that are increasingly demanded by corporate sustainability goals.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions means that the process is less susceptible to minor variations in raw material quality or environmental factors, ensuring consistent batch-to-batch performance. This reliability is critical for maintaining continuous production schedules and meeting tight delivery deadlines for key clients in the pharmaceutical and electronic sectors. The use of stable catalysts and oxidants that can be stored and handled with standard safety protocols further reduces logistical complexities associated with hazardous material transport. By securing a synthesis route that is both chemically robust and operationally simple, supply chain heads can mitigate the risks associated with scaling up new chemistries, ensuring a steady flow of high-purity intermediates to support downstream formulation and manufacturing activities.
- Scalability and Environmental Compliance: Scaling this reaction from gram to kilogram or tonnage levels is facilitated by the mild temperature range and the absence of extremely hazardous reagents like strong Lewis acids or pyrophoric organometallics. The waste profile of this process is cleaner compared to traditional methods, as the primary by-products are metal salts that can be managed through established waste treatment protocols. This ease of waste management simplifies regulatory compliance and reduces the environmental footprint of the manufacturing facility. The scalability is further supported by the wide substrate scope, meaning that the same reactor setup can be utilized to produce a variety of analogs by simply changing the starting materials, maximizing asset utilization. This flexibility allows manufacturers to respond quickly to market demands for new derivatives without significant capital investment in new equipment or infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived from the specific details and advantageous effects outlined in the patent documentation. Understanding these aspects helps stakeholders make informed decisions about integrating this chemistry into their existing portfolios.
Q: What catalyst system is used for this oxidative cyclization?
A: The process utilizes a Rhodium(III) catalyst, specifically dichloro(pentamethylcyclopentadienyl)rhodium(III) dimer, paired with copper acetate as the oxidant to drive the [4+2] cycloaddition efficiently.
Q: What is the optimal temperature range for this synthesis?
A: Experimental data indicates that reacting at temperatures between 80°C and 120°C provides the best balance of conversion and yield, with 100°C often being the preferred setpoint for optimal kinetics.
Q: Can this method accommodate diverse substrate substituents?
A: Yes, the method demonstrates broad substrate scope, tolerating various groups on the aryl ring including halogens, methyl, methoxy, and trifluoromethyl groups, as well as different N-substituents on the maleimide.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzo Pyridylimidazo Isoindole Diketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic methodologies like the Rh-catalyzed oxidative cyclization described in CN112174962A. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to market availability is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of benzo[e]pyridylimidazo[4,5-g]isoindole-1,3(2H)-dione meets the highest industry standards. We understand that consistency and quality are non-negotiable for our partners in the pharmaceutical and fine chemical sectors, and our dedicated technical team is committed to optimizing every step of the process to maximize yield and minimize impurities.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our expertise can accelerate your development timeline and enhance your competitive position in the market. Let us be your trusted partner in turning complex chemical challenges into commercial successes.
