Scalable Synthesis of 5-Trifluoromethyl-1,2,4-Triazoles for Advanced Pharmaceutical Intermediates
Scalable Synthesis of 5-Trifluoromethyl-1,2,4-Triazoles for Advanced Pharmaceutical Intermediates
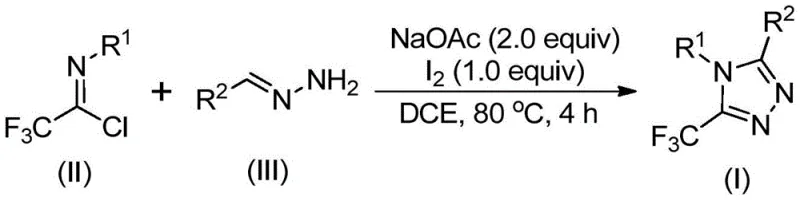
The pharmaceutical and agrochemical industries are constantly seeking robust, scalable, and cost-effective methodologies for constructing nitrogen-containing heterocycles, particularly those bearing trifluoromethyl groups which enhance metabolic stability and lipophilicity. Patent CN110467579B, published in August 2020, discloses a groundbreaking preparation method for 5-trifluoromethyl substituted 1,2,4-triazole compounds that addresses many of the historical pain points in heterocyclic synthesis. This technology leverages a non-metallic iodine-promoted cyclization strategy, utilizing inexpensive hydrazones and trifluoroethylimidoyl chlorides as starting materials. The significance of this development cannot be overstated for R&D directors and procurement managers alike, as 1,2,4-triazole scaffolds are ubiquitous in high-value active pharmaceutical ingredients (APIs) such as antifungal agents and aromatase inhibitors. By eliminating the need for complex pre-functionalization or dangerous diazo reagents, this patent offers a streamlined pathway to high-purity intermediates.
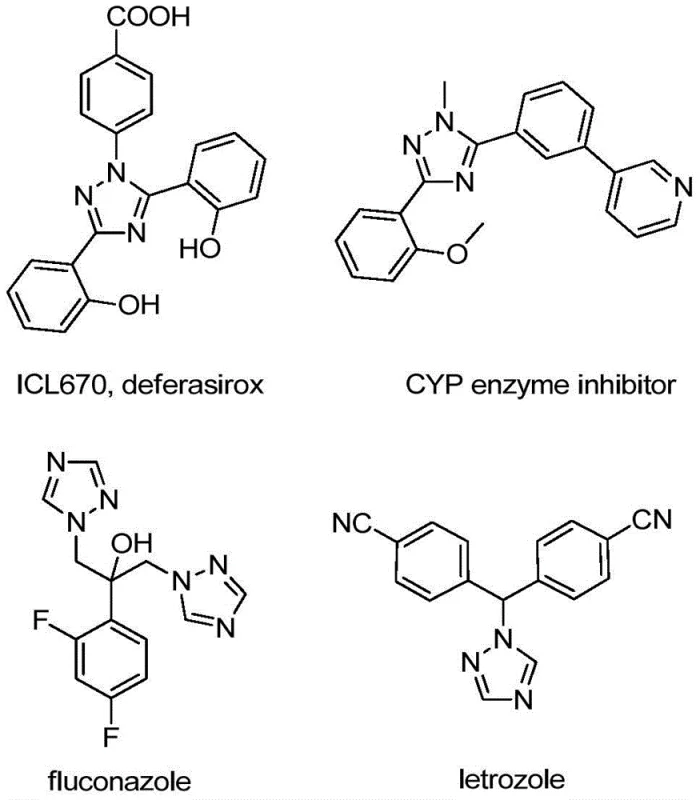
The structural versatility of the 1,2,4-triazole ring makes it a cornerstone in medicinal chemistry, appearing in diverse therapeutic areas ranging from oncology to infectious disease control. As illustrated in the reference structures, compounds like fluconazole and letrozole rely on this heterocyclic core for their biological efficacy. The introduction of a trifluoromethyl group at the 5-position further augments these properties by modulating electron density and improving binding affinity to target enzymes. However, traditional synthetic routes to access these specific motifs have often been plagued by harsh conditions, low atom economy, or the requirement for specialized equipment. The methodology outlined in CN110467579B represents a paradigm shift, offering a reliable pharmaceutical intermediate supplier with a tool to access these valuable chemical spaces with unprecedented operational simplicity and safety.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted nitrogen heterocycles has relied heavily on two primary strategies, both of which present significant logistical and economic challenges for large-scale manufacturing. The first approach involves the direct trifluoromethylation of pre-synthesized 1,2,4-triazole rings. While conceptually straightforward, this method necessitates the use of specialized and often hazardous trifluoromethylating reagents, such as Togni reagents or Umemoto reagents, which are prohibitively expensive for bulk production. Furthermore, these reactions frequently require strict anhydrous and anaerobic conditions to prevent reagent decomposition, imposing heavy burdens on facility infrastructure and operational costs. The second conventional pathway utilizes trifluoromethyl synthons like trifluorodiazoethane to react with coupling substrates. Although effective in academic settings, trifluorodiazoethane is inherently unstable and potentially explosive, posing severe safety risks that complicate regulatory approval for commercial scale-up. Additionally, these traditional methods often suffer from narrow substrate scope and poor functional group tolerance, limiting their utility in the synthesis of complex drug candidates.
The Novel Approach
In stark contrast to these legacy techniques, the novel approach detailed in the patent utilizes a metal-free, iodine-promoted cyclization that operates under remarkably mild and practical conditions. The core innovation lies in the condensation of trifluoroethylimidoyl chloride with readily available hydrazones in the presence of sodium acetate and elemental iodine. This reaction proceeds efficiently in common organic solvents like 1,2-dichloroethane (DCE) at moderate temperatures of 80°C to 100°C. Crucially, the process does not demand inert atmosphere protection or rigorous drying of solvents, drastically reducing the barrier to entry for implementation. The use of elemental iodine as a promoter rather than a stoichiometric oxidant or heavy metal catalyst ensures that the final product is free from toxic metal residues, a critical quality attribute for pharmaceutical intermediates. This method not only simplifies the synthetic route but also broadens the applicability to various substituted aryl and heteroaryl groups, enabling the rapid generation of diverse chemical libraries for drug discovery.
Mechanistic Insights into Iodine-Promoted Cyclization
Understanding the mechanistic underpinnings of this transformation is vital for R&D teams aiming to optimize the process for specific substrates. The reaction is believed to initiate with a base-promoted intermolecular carbon-nitrogen bond formation between the hydrazone nitrogen and the imidoyl chloride carbon, generating a trifluoroacetamidine intermediate. This step is facilitated by sodium acetate, which acts as a mild base to scavenge the generated hydrochloric acid without inducing unwanted side reactions. Following this condensation, the system undergoes an isomerization to align the reactive centers for cyclization. The pivotal role of elemental iodine then comes into play; it promotes an oxidative iodination process that activates the intermediate towards intramolecular electrophilic substitution. This activation lowers the energy barrier for ring closure, allowing the formation of the five-membered triazole ring under thermal conditions that would otherwise be insufficient. The final step involves aromatization, driven by the elimination of hydrogen iodide, to yield the stable 5-trifluoromethyl-1,2,4-triazole scaffold. This elegant cascade avoids the high-energy intermediates associated with radical trifluoromethylation, resulting in a cleaner reaction profile.
From an impurity control perspective, this mechanism offers distinct advantages over transition-metal catalyzed alternatives. The absence of palladium or copper eliminates the risk of metal leaching, which is a persistent challenge in API manufacturing that often requires additional scavenging steps and rigorous analytical testing. Furthermore, the mild basicity of sodium acetate minimizes the hydrolysis of sensitive functional groups on the aromatic rings, such as esters or nitriles, which might be degraded under stronger basic conditions. The oxidative nature of the iodine promotion is sufficiently selective to target the specific C-H bonds required for cyclization without over-oxidizing electron-rich substituents on the phenyl rings. This high level of chemoselectivity ensures that the crude reaction mixture contains fewer by-products, thereby simplifying downstream purification and enhancing the overall yield of the desired high-purity heterocyclic compounds.
How to Synthesize 5-Trifluoromethyl-1,2,4-Triazoles Efficiently
The operational simplicity of this synthesis makes it an ideal candidate for technology transfer from laboratory to pilot plant. The procedure begins with the charging of sodium acetate, trifluoroethylimidoyl chloride, and the specific hydrazone derivative into a reaction vessel containing an aprotic organic solvent. The mixture is agitated and heated to the optimal temperature range, allowing the initial condensation to reach completion before the introduction of the iodine promoter. This staged addition helps manage the exotherm and ensures the correct sequence of mechanistic events. Detailed standardized synthesis steps see the guide below.
- Mix sodium acetate, trifluoroethylimidoyl chloride, and hydrazone in an organic solvent such as dichloroethane (DCE).
- Heat the reaction mixture to 80°C and stir for 2 to 4 hours to facilitate initial condensation.
- Add elemental iodine to the system and continue heating for an additional 1 to 2 hours to promote oxidative cyclization.
- Perform post-treatment including filtration and column chromatography to isolate the pure 5-trifluoromethyl-1,2,4-triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route translates into tangible strategic benefits that extend beyond mere chemical yield. The primary advantage lies in the drastic simplification of the raw material supply chain. Unlike methods requiring custom-synthesized trifluoromethylating agents or unstable diazo compounds, this process relies on commodity chemicals. Trifluoroethylimidoyl chlorides and hydrazones are widely available from multiple global suppliers, ensuring supply continuity and mitigating the risk of single-source bottlenecks. The robustness of the reaction conditions means that production does not require specialized glass-lined reactors equipped for high-pressure or cryogenic operations, allowing for utilization of existing general-purpose manufacturing assets. This flexibility significantly reduces capital expenditure requirements for scaling up production capacity.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts represents a major driver for cost optimization in API manufacturing. Traditional cross-coupling or C-H activation methods often rely on palladium or rhodium complexes, which not only carry high upfront costs but also necessitate expensive recovery systems to meet residual metal specifications. By replacing these with elemental iodine and sodium acetate, the direct material costs are significantly reduced. Furthermore, the simplified workup procedure, which avoids complex metal scavenging steps, reduces the consumption of auxiliary materials and shortens the overall cycle time. The ability to run the reaction without strict anhydrous conditions also lowers utility costs associated with solvent drying and nitrogen purging, contributing to substantial overall cost savings in the production budget.
- Enhanced Supply Chain Reliability: The reliance on stable, shelf-stable starting materials enhances the reliability of the supply chain. Hydrazones and imidoyl chlorides can be stored for extended periods without significant degradation, allowing manufacturers to maintain strategic inventory buffers against market fluctuations. In contrast, methods relying on transient reagents like trifluorodiazoethane require just-in-time generation, creating a fragile supply chain vulnerable to equipment failure or reagent instability. The operational robustness of the iodine-promoted method also means that batch-to-batch variability is minimized, ensuring consistent quality and delivery schedules for downstream customers. This predictability is crucial for long-term supply agreements with multinational pharmaceutical companies.
- Scalability and Environmental Compliance: From an environmental and safety standpoint, this method aligns well with modern green chemistry principles and regulatory expectations. The avoidance of heavy metals simplifies waste stream management, as the effluent does not require specialized treatment for toxic metal removal. Elemental iodine, while requiring careful handling, is less hazardous than many alternative oxidants and can be recovered or neutralized using standard protocols. The use of common solvents like DCE, while requiring appropriate containment, is well-understood in industrial hygiene contexts. The scalability of the process is evidenced by its successful demonstration at the gram level with high conversion rates, suggesting a smooth path to kilogram and tonne-scale production without the need for extensive process re-engineering or safety re-validation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for potential partners evaluating this synthetic route for their own pipelines. Understanding these nuances is essential for assessing the feasibility of integrating this method into existing manufacturing workflows.
Q: Does this synthesis require expensive heavy metal catalysts?
A: No, the method described in patent CN110467579B utilizes elemental iodine and sodium acetate as promoters, completely avoiding the need for costly transition metal catalysts like palladium or copper.
Q: What are the reaction conditions regarding moisture and oxygen?
A: The process is highly robust and does not require strict anhydrous or anaerobic conditions, making it significantly easier to operate compared to traditional trifluoromethylation methods.
Q: Can this method be scaled for industrial production?
A: Yes, the patent explicitly states that the method can be easily expanded to the gram level and beyond, utilizing cheap and readily available starting materials suitable for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient heterocyclic synthesis plays in accelerating drug development timelines. Our technical team has thoroughly analyzed the methodology disclosed in CN110467579B and validated its potential for producing high-quality pharmaceutical intermediates. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to industrial supply is seamless. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of detecting trace impurities down to ppm levels, guaranteeing that every batch of 5-trifluoromethyl-1,2,4-triazole meets the exacting standards required for GMP manufacturing.
We invite R&D directors and procurement specialists to collaborate with us to leverage this advanced synthetic technology for your next project. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage you to contact our technical procurement team today to request specific COA data for our triazole intermediates and to discuss route feasibility assessments for your target molecules. Let us help you secure a competitive advantage through superior chemistry and reliable supply chain execution.
