Advanced Catalytic Synthesis of 2-Amino-3-Cyano-4H-Pyran Compounds for Commercial Pharmaceutical Applications
Introduction to Green Catalytic Innovation in Heterocyclic Synthesis
The pharmaceutical and fine chemical industries are constantly seeking more efficient, sustainable, and cost-effective pathways to synthesize complex heterocyclic scaffolds. Patent CN111229311B introduces a groundbreaking methodology for the synthesis of 2-amino-3-cyano-4H-pyran compounds, utilizing a novel KIT-6 supported imidazole ionic liquid catalyst. This technology represents a significant leap forward in green chemistry, addressing the longstanding challenges of catalyst recovery and environmental impact associated with traditional homogeneous catalysis. The 2-amino-3-cyano-4H-pyran core is a privileged structure in medicinal chemistry, serving as a vital intermediate for constructing bioactive molecules with potential anticancer, antiviral, and anti-inflammatory properties. By leveraging the unique mesoporous structure of KIT-6 silica combined with the tunable acidity and basicity of imidazolium ionic liquids, this invention offers a robust platform for the reliable production of high-purity pharmaceutical intermediates. The process operates under mild conditions, typically between 20°C and 90°C, utilizing ethanol as a benign solvent, which aligns perfectly with modern regulatory demands for safer manufacturing practices.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-amino-3-cyano-4H-pyran derivatives has relied on a variety of catalytic systems, many of which suffer from significant drawbacks that hinder large-scale commercial adoption. Traditional methods often employ homogeneous catalysts that, while effective, pose severe challenges in terms of separation and recycling. The inability to easily recover these catalysts leads to increased production costs due to the loss of expensive catalytic materials and the generation of substantial chemical waste. Furthermore, many conventional protocols require harsh reaction conditions, including high temperatures and the use of toxic organic solvents, which raise serious safety and environmental concerns. The presence of residual metal contaminants in the final product is another critical issue, particularly for pharmaceutical applications where strict purity specifications must be met. These factors collectively contribute to low atomic utilization rates and complicated downstream processing, making the conventional synthesis of these valuable heterocycles less attractive for cost-sensitive supply chains.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN111229311B utilizes a heterogeneous KIT-6 supported imidazole ionic liquid catalyst that effectively bridges the gap between homogeneous activity and heterogeneous recoverability. This innovative approach facilitates a one-pot condensation and Michael addition reaction between malononitrile, aryl aldehydes, and ethyl acetoacetate with exceptional efficiency. The KIT-6 support provides a high surface area and ordered mesoporous structure that ensures excellent dispersion of the active ionic liquid sites, thereby minimizing mass transfer resistance and maximizing contact between the substrate and the catalyst. 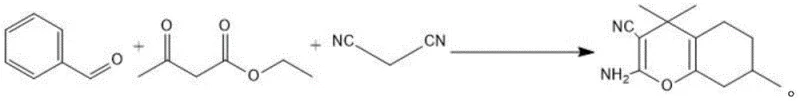 As illustrated in the reaction scheme, the process proceeds smoothly in ethanol, a green solvent, yielding the target pyran derivatives with high conversion rates. The solid nature of the catalyst allows for simple filtration post-reaction, enabling the filtrate to be directly processed for product isolation while the catalyst is recovered for reuse. This streamlined workflow not only simplifies the operation but also significantly enhances the overall economic viability of the manufacturing process.
As illustrated in the reaction scheme, the process proceeds smoothly in ethanol, a green solvent, yielding the target pyran derivatives with high conversion rates. The solid nature of the catalyst allows for simple filtration post-reaction, enabling the filtrate to be directly processed for product isolation while the catalyst is recovered for reuse. This streamlined workflow not only simplifies the operation but also significantly enhances the overall economic viability of the manufacturing process.
Mechanistic Insights into KIT-6 Supported Ionic Liquid Catalysis
The efficacy of this catalytic system lies in the synergistic interaction between the mesoporous silica support and the functionalized ionic liquid. The KIT-6 material serves as a stable scaffold that prevents the aggregation of ionic liquid species, maintaining a high density of accessible active sites throughout the reaction. The imidazolium cation acts as a phase-transfer agent and stabilizes the transition states, while the exchangeable anions—such as vanadate (VO4 3-), molybdate (MOO4 2-), ethoxide (CH3CH2O-), or hydroxide (OH-)—play a pivotal role in activating the nucleophiles. Specifically, the basic anions deprotonate the active methylene group of malononitrile, generating a carbanion that readily attacks the carbonyl carbon of the aryl aldehyde. This initial Knoevenagel condensation forms an arylmethylene malononitrile intermediate, which subsequently undergoes a Michael addition with ethyl acetoacetate. 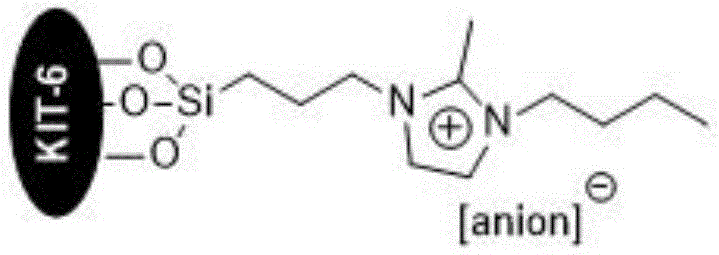 The structural integrity of the catalyst, as shown in the diagram, ensures that these active species remain anchored to the solid support, preventing leaching into the reaction medium. This immobilization is crucial for maintaining product purity and enabling the catalyst's recyclability, as the active components do not contaminate the final API intermediate.
The structural integrity of the catalyst, as shown in the diagram, ensures that these active species remain anchored to the solid support, preventing leaching into the reaction medium. This immobilization is crucial for maintaining product purity and enabling the catalyst's recyclability, as the active components do not contaminate the final API intermediate.
Furthermore, the mechanistic pathway inherently favors the formation of the desired 4H-pyran ring through an intramolecular cyclization step that is facilitated by the proximity of reactive groups on the catalyst surface. The ordered pore structure of KIT-6 may also exert a shape-selective effect, potentially suppressing the formation of bulky byproducts and enhancing the selectivity towards the target 2-amino-3-cyano-4H-pyran skeleton. This level of control over the reaction trajectory is essential for minimizing impurity profiles, a key concern for R&D directors focused on regulatory compliance. The ability to tune the anion component of the ionic liquid provides an additional layer of optimization, allowing chemists to adjust the basicity and nucleophilicity of the catalyst to suit specific substrate electronic properties. Such mechanistic flexibility ensures that the process remains robust across a diverse range of aryl aldehyde inputs, from electron-rich to electron-deficient variants.
How to Synthesize 2-Amino-3-Cyano-4H-Pyran Efficiently
Implementing this catalytic technology in a laboratory or pilot plant setting involves a straightforward protocol that emphasizes simplicity and reproducibility. The synthesis begins with the preparation of the KIT-6 supported ionic liquid catalyst, which involves grafting the functionalized imidazolium salt onto the silica surface followed by anion exchange. Once the catalyst is prepared, the reaction is conducted by simply mixing the three starting materials—malononitrile, the chosen aryl aldehyde, and ethyl acetoacetate—in ethanol with a catalytic amount of the solid material. The mixture is heated to a moderate temperature, typically around 80°C, and stirred for a few hours until completion. The detailed standardized synthesis steps, including precise molar ratios, reaction times, and workup procedures, are outlined in the guide below to ensure consistent results.
- React N-methylimidazole with sodium ethoxide in ethanol at 60-100°C for 7-10 hours to form Intermediate 1.
- Condense Intermediate 1 with 3-chloropropyltriethoxysilane in acetonitrile at 60-100°C for 10-20 hours to yield Intermediate 2.
- Quaternize Intermediate 2 with n-butyl bromide in toluene at 80-120°C for 24-48 hours to obtain the imidazolium salt precursor.
- Perform anion exchange using sodium vanadate, molybdate, ethoxide, or hydroxide in methanol to generate the active ionic liquid species.
- Impregnate the functionalized ionic liquid onto KIT-6 mesoporous silica in ethanol at 60-100°C to finalize the supported catalyst.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this KIT-6 supported ionic liquid technology offers compelling strategic advantages that extend beyond mere technical performance. The shift towards this greener, heterogeneous catalytic system addresses several critical pain points in the current supply chain for fine chemical intermediates. By eliminating the need for stoichiometric amounts of hazardous reagents and simplifying the purification process, manufacturers can achieve significant reductions in raw material consumption and waste disposal costs. The robustness of the catalyst also translates to greater supply chain reliability, as the process is less susceptible to variations in raw material quality and operating conditions. This stability ensures a consistent flow of high-quality intermediates, reducing the risk of production delays that can ripple through the entire pharmaceutical value chain.
- Cost Reduction in Manufacturing: The implementation of this catalytic route drives cost reduction in fine chemical manufacturing primarily through the elimination of expensive transition metal catalysts and the associated removal steps. Traditional methods often require costly scavengers or chromatography to remove metal residues, whereas this heterogeneous system allows for simple filtration. Additionally, the reusability of the catalyst for multiple cycles without significant loss of activity means that the effective cost per kilogram of product is drastically lowered over time. The use of ethanol as a solvent further contributes to cost savings, as it is cheaper, safer, and easier to recover than many chlorinated or aromatic solvents used in legacy processes.
- Enhanced Supply Chain Reliability: Supply chain reliability is significantly enhanced by the simplicity and robustness of the reaction conditions. The process operates at atmospheric pressure and moderate temperatures, reducing the need for specialized high-pressure equipment and lowering energy consumption. The catalyst's stability and ease of recovery mean that production schedules are less likely to be disrupted by catalyst degradation or complex regeneration protocols. Furthermore, the broad substrate scope allows for the flexible production of various derivatives from a single catalytic platform, enabling suppliers to respond quickly to changing market demands for different API intermediates without retooling entire production lines.
- Scalability and Environmental Compliance: Scalability is a inherent strength of this heterogeneous catalytic system, making it ideal for commercial scale-up of complex heterocyclic intermediates. The solid catalyst can be easily handled in large-scale reactors, and the filtration step scales linearly with batch size. From an environmental compliance perspective, the process aligns with green chemistry principles by minimizing waste generation and avoiding toxic heavy metals. This reduces the regulatory burden on manufacturers and lowers the costs associated with environmental permits and waste treatment, ensuring long-term sustainability and compliance with increasingly stringent global environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented catalytic technology. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on performance metrics and operational parameters. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this method into their existing manufacturing workflows.
Q: What are the primary advantages of using KIT-6 supported ionic liquids over traditional homogeneous catalysts?
A: The KIT-6 supported ionic liquid catalyst combines the high activity of homogeneous ionic liquids with the ease of separation associated with heterogeneous catalysts. This hybrid structure allows for simple filtration and reuse of the catalyst for multiple cycles without significant loss of activity, drastically reducing waste generation and operational costs compared to traditional methods that require complex workup procedures.
Q: Can this catalytic system accommodate diverse aryl aldehyde substrates?
A: Yes, the patent data demonstrates excellent substrate tolerance. The catalytic system effectively processes aryl aldehydes bearing various substituents such as hydrogen, hydroxyl, nitro, methyl, chloro, and trifluoromethyl groups at different positions on the aromatic ring. This versatility makes it a robust choice for synthesizing a wide library of 2-amino-3-cyano-4H-pyran derivatives for drug discovery.
Q: How does the anion structure influence the catalytic efficiency?
A: The anion plays a critical role in activating the active methylene group of malononitrile. The patent highlights that anions like vanadate, molybdate, ethoxide, and hydroxide enhance the nucleophilicity of the reactants, facilitating the Knoevenagel condensation and subsequent Michael addition. This tunability allows chemists to optimize reaction rates and yields by selecting the appropriate metal salt anion for specific substrate requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino-3-Cyano-4H-Pyran Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the KIT-6 supported ionic liquid system in driving the next generation of pharmaceutical synthesis. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are seamlessly translated into industrial reality. Our commitment to quality is unwavering, with stringent purity specifications and rigorous QC labs dedicated to verifying the identity and purity of every batch of 2-amino-3-cyano-4H-pyran compounds we produce. We understand that consistency is key in the pharmaceutical supply chain, and our state-of-the-art facilities are designed to deliver high-purity intermediates that meet the exacting standards of global regulatory bodies.
We invite you to collaborate with us to leverage this cutting-edge synthesis method for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this green catalytic route can optimize your bill of materials. Please contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary molecules. Together, we can build a more efficient, sustainable, and reliable supply chain for the future of medicine.
