Scalable Synthesis of 2,4,5-Triaryl Imidazoles Using Biodegradable Acidic Ionic Liquids for Commercial Production
The pharmaceutical and fine chemical industries are constantly seeking more sustainable and efficient pathways for synthesizing heterocyclic scaffolds, particularly imidazole derivatives which serve as critical building blocks for numerous bioactive molecules. Patent CN103880755A introduces a significant technological breakthrough in this domain by disclosing a method for preparing 2,4,5-triaryl substituted imidazoles utilizing a novel degradable acidic ionic liquid catalyst. This innovation addresses the longstanding challenges associated with traditional synthesis routes, such as harsh reaction conditions, difficult product purification, and the environmental persistence of conventional catalysts. By employing a Brönsted acidic ionic liquid that is both highly active and biodegradable, this technology offers a compelling solution for the commercial scale-up of complex pharmaceutical intermediates. The process operates under mild reflux conditions in ethanol, achieving high conversion rates while minimizing the ecological footprint, thereby positioning it as an ideal candidate for modern green manufacturing protocols.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
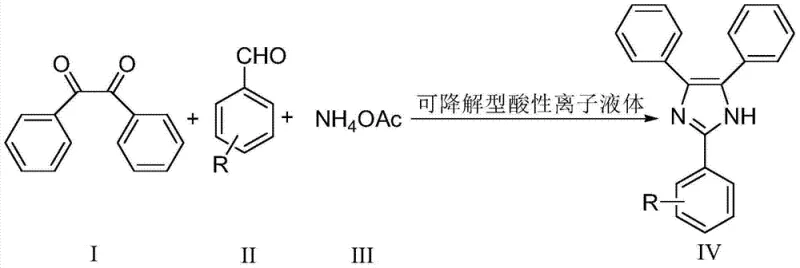
Historically, the synthesis of aryl-substituted imidazoles has relied heavily on the condensation of benzil, aromatic aldehydes, and ammonium acetate under reflux, often requiring strong mineral acids or non-degradable ionic liquids as promoters. Traditional methods frequently suffer from prolonged reaction times, sometimes extending over many hours, which leads to energy inefficiencies and potential thermal degradation of sensitive functional groups. Furthermore, the use of conventional acidic ionic liquids, such as those based on imidazolium or pyridinium cations, presents a severe environmental liability due to their poor biodegradability and toxicity profiles. These persistent organic pollutants accumulate in wastewater streams, necessitating expensive and complex treatment processes to meet regulatory discharge standards. Additionally, many existing catalytic systems require stoichiometric amounts or very high loadings to achieve acceptable yields, drastically inflating the raw material costs and complicating the downstream isolation of the pure product from the reaction mixture.
The Novel Approach
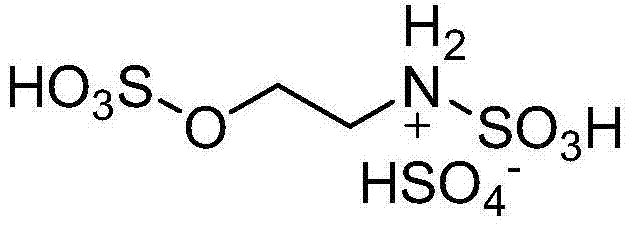
In stark contrast, the methodology outlined in CN103880755A utilizes a specifically designed degradable acidic ionic liquid that functions as a highly efficient Brönsted acid catalyst. This novel catalyst features a unique molecular architecture that combines high acidity with a linear structure conducive to biological degradation, effectively solving the environmental persistence issue plaguing earlier generations of ionic liquids. The reaction proceeds rapidly, typically completing within 0.5 to 2 hours, which represents a substantial improvement in throughput and energy consumption compared to legacy processes. Moreover, the catalyst loading is remarkably low, ranging from only 3% to 5% molar ratio relative to the aromatic aldehyde, which significantly reduces the cost of goods sold (COGS). The use of ethanol as a solvent further enhances the safety profile and ease of operation, making this approach highly attractive for cost reduction in pharmaceutical intermediate manufacturing where safety and efficiency are paramount.
Mechanistic Insights into Brönsted Acidic Ionic Liquid Catalysis
The exceptional catalytic performance observed in this system can be attributed to the specific structural features of the ionic liquid, which acts as a dual-acidic site promoter. As illustrated in the catalyst structure, the molecule possesses multiple sulfonic acid (-SO3H) groups that provide strong Brönsted acidity, facilitating the activation of the carbonyl groups in both the benzil and the aromatic aldehyde. This dual activation mechanism lowers the energy barrier for the nucleophilic attack by ammonia (generated in situ from ammonium acetate), thereby accelerating the cyclization process that forms the imidazole ring. The uniform distribution of these acidic sites within the ionic liquid medium ensures consistent reaction kinetics, preventing the formation of localized hot spots that could lead to side reactions or polymerization. This precise control over the reaction environment is crucial for maintaining high selectivity and minimizing the generation of impurities that are difficult to remove in later purification stages.
From an impurity control perspective, the mild nature of this catalytic system plays a pivotal role in ensuring the quality of the final high-purity pharmaceutical intermediates. Unlike strong mineral acids which can promote dehydration or rearrangement side reactions, the buffered acidity of the ionic liquid creates a controlled environment that favors the desired condensation pathway. The solubility characteristics of the ionic liquid also aid in keeping the reaction intermediates in solution during the critical cyclization phase, preventing premature precipitation that could trap unreacted starting materials. Upon completion, the addition of water triggers the precipitation of the product while the catalyst remains largely in the aqueous phase or is easily separated, allowing for a clean isolation process. This inherent selectivity reduces the burden on downstream purification units, such as chromatography columns, leading to a more streamlined and cost-effective production workflow.
How to Synthesize 2,4,5-Triaryl Imidazoles Efficiently
The operational simplicity of this synthesis route makes it highly accessible for laboratory scale-up and subsequent industrial implementation. The process begins with the precise weighing of benzil, the chosen aromatic aldehyde, and ammonium acetate, which are then introduced into a reaction vessel containing ethanol and the degradable acidic ionic liquid catalyst. The mixture is subjected to vigorous stirring and heated to reflux, a condition that is maintained for a short duration of 0.5 to 2 hours depending on the electronic nature of the substituents on the aldehyde. Monitoring the reaction progress via Thin Layer Chromatography (TLC) allows operators to determine the exact endpoint, ensuring maximum conversion without unnecessary energy expenditure. Once the reaction is complete, the solvent is removed via rotary evaporation, and the crude product is induced to precipitate by the addition of water, followed by filtration, washing, and recrystallization to yield the pure target compound.
- Mix benzil, aromatic aldehyde, and ammonium acetate in a molar ratio of 1: 1:2~4 with 3-5% mol of degradable acidic ionic liquid catalyst in ethanol.
- Reflux the mixture under vigorous stirring for 0.5 to 2 hours until TLC indicates the disappearance of raw materials.
- Remove solvent via rotary evaporation, add water to precipitate solids, filter, wash, dry, and recrystallize from ethanol to obtain pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this catalytic technology translates into tangible strategic benefits that extend beyond mere chemical efficiency. The primary advantage lies in the drastic simplification of the supply chain for catalysts; since the ionic liquid can be recovered and reused multiple times without significant loss of activity, the dependency on continuous fresh catalyst purchases is greatly diminished. This recyclability feature not only stabilizes the supply of critical reagents but also insulates the production process from volatility in raw material pricing. Furthermore, the biodegradable nature of the catalyst eliminates the need for specialized hazardous waste disposal services, resulting in substantial cost savings related to environmental compliance and waste management. The use of ethanol, a commodity solvent with a stable global supply, further mitigates supply chain risks associated with exotic or regulated solvents.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the extremely low catalyst loading of 3-5% and its ability to be recycled at least five times, which dramatically lowers the per-batch cost of catalytic materials. By eliminating the need for expensive transition metal catalysts or stoichiometric amounts of corrosive acids, manufacturers can achieve significant reductions in raw material expenditures. Additionally, the shortened reaction time of 0.5 to 2 hours increases reactor turnover rates, allowing facilities to produce more batches within the same timeframe and effectively spreading fixed operational costs over a larger volume of output. The simplified workup procedure, which avoids complex extraction or neutralization steps, further reduces labor and utility costs associated with the manufacturing process.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures a consistent and reliable supply of 2,4,5-triaryl imidazoles, which is critical for maintaining uninterrupted production schedules for downstream API manufacturing. The catalyst's stability and reusability mean that production is less susceptible to disruptions caused by catalyst supply shortages or quality variations between batches. Moreover, the use of widely available starting materials like benzil and common aromatic aldehydes ensures that the entire supply chain is resilient and less prone to geopolitical or logistical bottlenecks. This reliability is essential for long-term contracts with pharmaceutical clients who demand strict adherence to delivery timelines and consistent product quality.
- Scalability and Environmental Compliance: Scaling this process from laboratory to industrial production is straightforward due to the use of standard equipment and safe solvents like ethanol, which do not require specialized pressure vessels or exotic metallurgy. The biodegradable nature of the catalyst aligns perfectly with increasingly stringent global environmental regulations, reducing the risk of fines or shutdowns due to non-compliance. The aqueous workup generates minimal hazardous waste, simplifying the permitting process for new manufacturing lines and enhancing the company's sustainability profile. This environmental stewardship is becoming a key differentiator in the B2B chemical market, appealing to multinational corporations with aggressive carbon reduction and green chemistry goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this degradable ionic liquid catalytic system. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the technology's capabilities and limitations. Understanding these details is crucial for R&D teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios and for procurement teams assessing the total cost of ownership.
Q: What are the environmental advantages of this ionic liquid catalyst compared to traditional imidazolium salts?
A: Unlike conventional imidazolium-based ionic liquids which are resistant to biodegradation, the catalyst used in patent CN103880755A features a linear structure with sulfonic acid groups that is easily biodegradable, aligning with green chemistry policies and reducing wastewater treatment costs.
Q: Can the catalyst be recycled without significant loss of activity?
A: Yes, the patent data demonstrates that the filtrate containing the catalyst can be reused at least 5 times after simple water removal at 110°C, with no significant decrease in product yield, ensuring high atom economy and reduced material costs.
Q: What yields can be expected for various substituted aromatic aldehydes?
A: The method is robust across various substrates; for instance, unsubstituted benzaldehyde yields 95%, while electron-withdrawing groups like dichloro-substituents yield 91%, and electron-donating methoxy groups yield 92%, demonstrating broad substrate tolerance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,4,5-Triaryl Imidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of green catalytic technologies like the one described in CN103880755A for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel ionic liquid method are fully realized in a practical industrial setting. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2,4,5-triaryl imidazole delivered meets the highest international standards for pharmaceutical applications. We are committed to leveraging our technical expertise to optimize this green synthesis route for your specific needs, ensuring both economic viability and environmental sustainability.
We invite you to collaborate with us to explore how this advanced catalytic method can enhance your supply chain efficiency and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us today to request specific COA data for our imidazole derivatives and to discuss detailed route feasibility assessments for your upcoming projects. Let us be your trusted partner in navigating the complexities of modern chemical synthesis and delivering superior value to your organization.
