Scalable Manufacturing of Cinnoline Salts via Novel Copper-Catalyzed Cyclization for Global Markets
Scalable Manufacturing of Cinnoline Salts via Novel Copper-Catalyzed Cyclization for Global Markets
The landscape of organic synthesis for nitrogen-containing heterocycles is undergoing a significant transformation, driven by the urgent need for more sustainable and cost-effective manufacturing processes. A pivotal development in this field is detailed in patent CN112552242A, which discloses a robust method for synthesizing cinnoline salt compounds. This technology represents a major leap forward for the production of high-value pharmaceutical intermediates and fine chemicals. By utilizing a unique composite catalytic system comprising a copper catalyst and a specific auxiliary substance, this invention enables the efficient construction of the cinnoline skeleton through a catalytic cyclization reaction. For global procurement leaders and R&D directors, this patent offers a compelling alternative to traditional methods, promising enhanced supply chain stability and reduced production costs without compromising on product quality or purity standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cinnoline derivatives and related nitrogenous inner salt structures has relied heavily on transition metal catalysis involving precious metals such as Rhodium (Rh) and Palladium (Pd). While effective, these conventional pathways present substantial economic and logistical challenges for industrial-scale operations. The reliance on noble metal catalysts introduces high raw material costs and necessitates rigorous metal removal steps to meet stringent pharmaceutical purity specifications. Furthermore, many existing protocols require the use of strong oxidizers, expensive additives, and strictly anaerobic conditions, often involving inert gas protection like nitrogen or argon. These factors collectively increase the complexity of the manufacturing process, extend lead times, and elevate the overall cost of goods sold (COGS), making the supply of these critical intermediates vulnerable to market fluctuations in precious metal prices.
The Novel Approach
The methodology described in patent CN112552242A fundamentally disrupts these traditional constraints by introducing a base-metal catalytic system. This novel approach utilizes Cuprous Chloride (CuCl) in synergy with 4-Dimethylaminopyridine (DMAP) to facilitate the cyclization of 2-ethynylaniline derivatives with nitrosobenzene compounds. Unlike previous methods that demand expensive noble metals, this copper-catalyzed route leverages abundant and inexpensive materials. Crucially, the reaction operates efficiently under an air atmosphere, removing the need for complex inert gas setups. This shift not only drastically simplifies the operational requirements but also enhances the safety profile of the synthesis. The ability to achieve high yields under such mild and open conditions signifies a paradigm shift towards greener, more economical chemical manufacturing, directly addressing the pain points of cost and scalability faced by modern chemical enterprises.
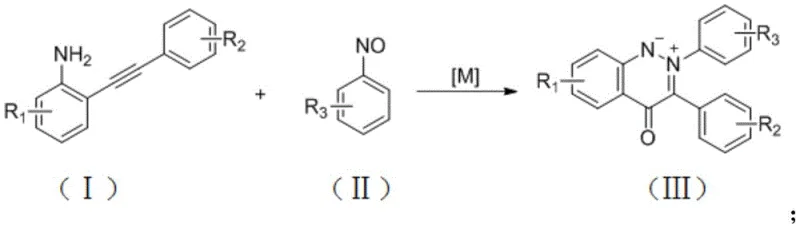
The core innovation lies in the specific interplay between the copper catalyst and the auxiliary amine base. As illustrated in the reaction scheme, the system effectively promotes the oxidative coupling and subsequent cyclization required to form the cinnoline ylide structure. This mechanistic efficiency allows for a broad substrate scope, accommodating various functional groups such as halogens, esters, cyano groups, and alkyl chains without significant loss in yield. For R&D teams, this versatility means a single, optimized platform can be adapted to synthesize a diverse library of analogues, accelerating the drug discovery pipeline and reducing the time-to-market for new therapeutic candidates.
Mechanistic Insights into CuCl-Catalyzed Cyclization
Understanding the mechanistic underpinnings of this transformation is vital for ensuring reproducibility and optimizing process parameters. The reaction likely proceeds through a copper-mediated activation of the alkyne moiety in the 2-ethynylaniline substrate. The Cu(I) species coordinates with the triple bond, increasing its electrophilicity and facilitating nucleophilic attack by the nitroso group of the nitrosobenzene. The presence of DMAP acts as a crucial promoter, potentially stabilizing intermediate copper species or assisting in proton transfer steps essential for the cyclization cascade. The use of air as the terminal oxidant is particularly noteworthy; molecular oxygen serves to regenerate the active copper catalyst, closing the catalytic cycle without the need for stoichiometric chemical oxidants that often generate hazardous waste. This atom-economical approach aligns perfectly with the principles of green chemistry, minimizing waste generation and improving the overall environmental footprint of the synthesis.
From an impurity control perspective, the mildness of the reaction conditions plays a pivotal role. High-temperature or strongly acidic/basic conditions often lead to decomposition or side reactions, generating difficult-to-remove impurities that complicate downstream purification. In contrast, the CuCl/DMAP system operates at a moderate temperature of 100°C in toluene, preserving the integrity of sensitive functional groups. The patent data indicates that the resulting crude products are relatively clean, allowing for straightforward purification via standard silica gel chromatography. This high level of chemoselectivity ensures that the final cinnoline salt compounds meet the rigorous purity standards required for pharmaceutical applications, thereby reducing the burden on quality control laboratories and ensuring a consistent supply of high-quality material.
How to Synthesize Cinnoline Salt Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting is straightforward and relies on standard chemical engineering practices. The process begins with the precise weighing of the starting materials: the 2-ethynylaniline derivative and the nitrosobenzene derivative. These are combined in a reaction vessel with the catalytic system consisting of CuCl (typically 10 mol%) and DMAP (40 mol%) dissolved in toluene. The simplicity of the reagent setup is a key advantage, as it avoids the handling of air-sensitive reagents that require glovebox techniques. Once the mixture is prepared, the vessel is sealed and heated to 100°C. The reaction is allowed to proceed for approximately 24 hours under an air atmosphere, during which time the oxidative cyclization takes place. Monitoring the reaction progress via TLC or HPLC is recommended to determine the optimal endpoint, although the patent suggests that 24 hours is generally sufficient for high conversion.
- Charge a reaction vessel with 2-ethynylaniline derivative, nitrosobenzene derivative, CuCl catalyst (10 mol%), and DMAP additive (40 mol%) in toluene solvent.
- Seal the vessel and heat the mixture to 100°C under an air atmosphere, stirring continuously for approximately 24 hours to ensure complete conversion.
- Upon completion, cool the mixture, wash with ethyl acetate, concentrate under reduced pressure, and purify the crude product via silica gel column chromatography.
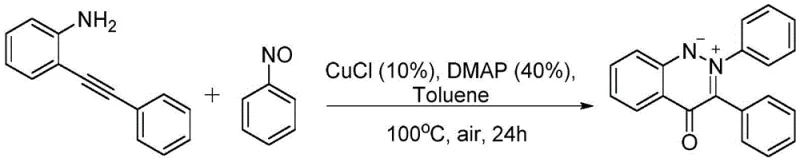
Following the reaction period, the workup procedure is designed for maximum efficiency and minimal waste. The reaction mixture is cooled to room temperature and transferred, where it is washed with ethyl acetate to extract the organic components. The combined organic layers are then concentrated under reduced pressure to remove the solvent. The resulting crude residue is subjected to silica gel column chromatography, using a mixture of dichloromethane and ethyl acetate as the eluent, to isolate the pure cinnoline salt product. As demonstrated in Example 1 of the patent, this protocol reliably yields 2,3-diphenyl-4-oxocinnoline ylide with a 74% isolated yield, confirming the practical viability of the method.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this copper-catalyzed technology offers tangible strategic benefits that extend beyond simple chemistry. The shift from noble metals to base metals fundamentally alters the cost structure of the manufacturing process. Copper chloride is orders of magnitude cheaper than rhodium or palladium catalysts, leading to a direct reduction in raw material expenses. Furthermore, the elimination of inert gas requirements simplifies the infrastructure needed for production, allowing for the use of standard reactors rather than specialized pressure vessels equipped for anaerobic conditions. These factors combine to create a more resilient supply chain that is less susceptible to the volatility of precious metal markets and geopolitical supply disruptions.
- Cost Reduction in Manufacturing: The substitution of expensive noble metal catalysts with inexpensive copper salts results in substantial cost savings per kilogram of product. Additionally, the use of air as the oxidant eliminates the need for purchasing and storing hazardous chemical oxidizers, further lowering operational expenditures. The simplified workup and purification processes also reduce labor and solvent consumption, contributing to a leaner and more cost-effective manufacturing model that enhances overall profit margins.
- Enhanced Supply Chain Reliability: By utilizing widely available and stable reagents like CuCl and toluene, the risk of supply bottlenecks is significantly minimized. The robustness of the reaction under air atmosphere means that production can continue uninterrupted even if there are temporary shortages of specialty gases. This reliability ensures consistent delivery schedules for downstream customers, fostering stronger long-term partnerships and trust within the global pharmaceutical supply network.
- Scalability and Environmental Compliance: The mild reaction conditions and lack of toxic heavy metals make this process inherently safer and easier to scale from gram to ton quantities. The reduced generation of hazardous waste aligns with increasingly strict environmental regulations, lowering the costs associated with waste disposal and compliance reporting. This sustainability advantage not only protects the company's reputation but also future-proofs the manufacturing process against tightening global environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of cinnoline salts using this novel methodology. These insights are derived directly from the experimental data and technical disclosures found in the patent literature, providing a transparent view of the technology's capabilities and limitations for potential partners and stakeholders. Understanding these details is crucial for making informed decisions about process adoption and integration into existing manufacturing workflows.
Q: What are the primary advantages of this CuCl catalytic system over traditional noble metal methods?
A: The primary advantage is the replacement of expensive noble metal catalysts like Rhodium or Palladium with inexpensive and abundant Copper(I) Chloride. Additionally, the reaction proceeds efficiently under an air atmosphere, eliminating the need for costly inert gas protection and complex degassing procedures, which significantly simplifies the operational workflow and reduces raw material costs.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method is highly suitable for scale-up. The use of common solvents like toluene, mild reaction temperatures around 100°C, and the tolerance to air oxidation make the process robust and safe for kilogram-to-ton scale manufacturing without requiring specialized high-pressure or anaerobic equipment.
Q: What is the typical yield range for this cinnoline salt synthesis?
A: According to the patent data, the reaction demonstrates good efficiency with isolated yields typically ranging from 60% to 86% across various substrate derivatives, indicating a reliable and consistent process for generating high-purity intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cinnoline Salt Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic route described in patent CN112552242A for the production of high-performance cinnoline salt intermediates. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate this innovative laboratory chemistry into robust, commercial-scale manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities.
We invite you to collaborate with us to leverage this cost-effective and scalable technology for your next project. Whether you require custom synthesis of specific cinnoline derivatives or optimization of existing routes, our technical procurement team is ready to assist. Please contact us today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are eager to provide you with specific COA data and comprehensive route feasibility assessments to demonstrate how we can add value to your supply chain and accelerate your time to market.
