Advanced Solvent-Free Levetiracetam Manufacturing for Global Pharmaceutical Supply Chains
Advanced Solvent-Free Levetiracetam Manufacturing for Global Pharmaceutical Supply Chains
The global demand for high-purity antiepileptic medications continues to surge, driving the need for more sustainable and cost-effective manufacturing processes. Patent CN102675181B introduces a groundbreaking preparation method for Levetiracetam that fundamentally alters the traditional synthetic landscape by eliminating organic solvents entirely. This technical insight report analyzes the proprietary two-step sequence, which utilizes aqueous acylation followed by a novel solvent-free mechanochemical cyclization. For R&D Directors and Supply Chain Heads, this patent represents a critical opportunity to optimize production efficiency while adhering to increasingly stringent environmental regulations. The method achieves high yields through mild reaction conditions, specifically operating at room temperature, which contrasts sharply with the energy-intensive cooling requirements of legacy protocols. By integrating this green chemistry approach, manufacturers can significantly reduce their environmental footprint while maintaining the rigorous quality standards required for API production.
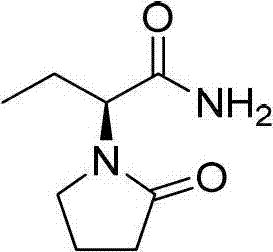
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Levetiracetam has relied heavily on protocols that are both economically and environmentally burdensome. Traditional methods, such as those described in USP4696943A, typically require the use of volatile organic compounds like acetonitrile and dichloromethane in substantial volumes. These solvents not only escalate raw material costs but also necessitate complex recovery and waste treatment systems to comply with environmental safety standards. Furthermore, conventional cyclization steps often demand low-temperature conditions, frequently around 0°C, which imposes a significant energy load on the manufacturing facility. The reliance on phase transfer catalysts in these older routes adds another layer of complexity, introducing potential impurities that require additional purification steps to remove. Consequently, the overall process mass intensity is high, leading to increased operational expenditures and a larger carbon footprint per kilogram of active pharmaceutical ingredient produced.
The Novel Approach
In stark contrast, the novel approach detailed in CN102675181B offers a streamlined pathway that addresses these inefficiencies through innovative solvent management. The first step utilizes water as the sole reaction medium for the acylation of (S)-2-aminobutyramide hydrochloride, completely bypassing the need for expensive organic solvents. This aqueous system operates effectively at room temperature, eliminating the energy costs associated with cryogenic cooling. The second step is even more revolutionary, employing a solvent-free grinding technique where the intermediate is mixed with an inorganic carrier and base to induce intramolecular cyclization. This mechanochemical activation removes the need for dichloromethane and phase transfer catalysts entirely. The result is a process that is not only greener but also operationally simpler, reducing the number of unit operations and minimizing the risk of solvent-related safety incidents in the plant.
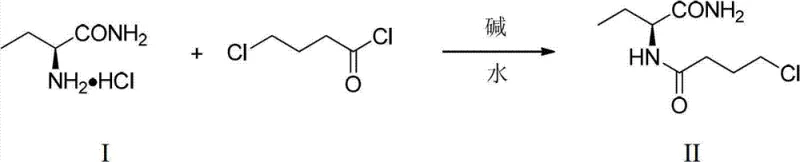
Mechanistic Insights into Aqueous Acylation and Mechanochemical Cyclization
The chemical mechanism underpinning this synthesis is designed to maximize atomic economy while minimizing side reactions. In the initial acylation phase, the nucleophilic attack of the amine group on the acid chloride is facilitated in an aqueous environment through careful pH control using inorganic bases like sodium carbonate or potassium carbonate. The use of water as a solvent creates a unique microenvironment that can enhance reaction selectivity, reducing the formation of hydrolysis byproducts often seen in organic media. The stoichiometry is tightly controlled, with a molar ratio of base to raw material optimized between 2:1 and 5:1 to ensure complete conversion without excessive waste. This precise control over reaction conditions is critical for maintaining the integrity of the chiral center, ensuring that the resulting intermediate retains the necessary stereochemistry for the final API.
The subsequent cyclization step leverages the principles of mechanochemistry to drive the ring-closing reaction. By grinding the intermediate amide with a solid base such as sodium hydroxide and an inorganic carrier like anhydrous sodium sulfate, the reaction proceeds through direct solid-state contact. This method avoids the solvation effects that can sometimes hinder cyclization or promote polymerization in solution-phase reactions. The mechanical energy input during grinding activates the reactants, allowing the nucleophilic nitrogen to attack the terminal chloride and form the pyrrolidone ring efficiently. This solvent-free environment inherently limits the mobility of potential impurities, leading to a cleaner reaction profile. The absence of liquid solvents also simplifies the work-up process, as the product can be directly extracted and recrystallized, streamlining the path to high-purity Levetiracetam.

How to Synthesize Levetiracetam Efficiently
Implementing this synthesis route requires a shift from traditional solution-phase processing to a hybrid aqueous and solid-state workflow. The process begins with the dissolution of the chiral starting material in water, followed by the controlled addition of the acylating agent under basic conditions. Once the intermediate is isolated, the focus shifts to solid-state processing, where mixing efficiency and grinding time become critical process parameters. Detailed standardized synthesis steps are essential to ensure reproducibility and consistent quality across different batch sizes. Operators must be trained to monitor the reaction progress via TLC during the grinding phase to prevent over-processing or incomplete conversion. The following guide outlines the critical operational parameters derived from the patent data to assist technical teams in adopting this methodology.
- Perform acylation of (S)-2-aminobutyramide hydrochloride with 4-chlorobutyryl chloride in water using inorganic bases at room temperature.
- Isolate the intermediate amide and mix with an inorganic carrier and solid base for solvent-free grinding.
- Complete the intramolecular cyclization via mechanochemical activation and purify via ethyl acetate recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this green synthesis route offers substantial strategic advantages beyond mere regulatory compliance. The elimination of organic solvents directly translates to a reduction in raw material procurement costs, as water and inorganic salts are significantly cheaper than acetonitrile or dichloromethane. Furthermore, the removal of phase transfer catalysts simplifies the bill of materials, reducing the dependency on specialized chemical suppliers. The simplified operational workflow also enhances supply chain reliability by reducing the number of potential failure points in the manufacturing process. With fewer unit operations and less complex waste management requirements, production lead times can be optimized, ensuring a more consistent flow of high-purity intermediates to downstream API manufacturing facilities.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the complete removal of volatile organic solvents from the reaction sequence. By substituting expensive organic media with water and utilizing solvent-free grinding, the facility eliminates the costs associated with solvent purchase, storage, recovery, and disposal. Additionally, the ability to run reactions at room temperature removes the energy burden of maintaining low-temperature reactors, leading to significant utility savings. The simplified purification process, which avoids complex chromatographic separations often needed to remove catalyst residues, further reduces operational expenditures. These cumulative efficiencies result in a lower cost of goods sold, providing a competitive edge in the pricing of the final pharmaceutical product.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly improved by reducing the reliance on hazardous and regulated solvents. Organic solvents often face supply volatility due to environmental regulations and transportation restrictions, whereas water and inorganic bases are universally available and stable commodities. The simplified process flow also reduces the risk of production delays caused by equipment failures associated with complex solvent recovery systems. By minimizing the number of chemical inputs and processing steps, the manufacturing timeline becomes more predictable and robust. This stability is crucial for meeting the rigorous delivery schedules demanded by global pharmaceutical clients, ensuring reducing lead time for high-purity APIs without compromising on quality or safety standards.
- Scalability and Environmental Compliance: Scaling this process from pilot to commercial production is inherently safer and more straightforward due to the absence of large volumes of flammable solvents. The solvent-free grinding step can be adapted to industrial-scale milling equipment, facilitating the commercial scale-up of complex pharmaceutical intermediates without the need for massive reactor vessels. From an environmental perspective, the drastic reduction in waste solvent generation simplifies compliance with increasingly strict environmental protection laws. This green profile enhances the corporate sustainability image, which is becoming a key factor in vendor selection for major multinational pharmaceutical companies. The process aligns with green chemistry principles, ensuring long-term viability in a regulatory landscape that favors sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this solvent-free synthesis method. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation. They are designed to provide clarity on process feasibility, quality control, and the specific advantages this route offers over traditional methods. Understanding these details is essential for technical teams evaluating the integration of this technology into existing production lines.
Q: How does this method eliminate organic solvent waste?
A: The process replaces acetonitrile and dichloromethane with water in the first step and utilizes a solvent-free grinding technique for the cyclization step, drastically reducing VOC emissions.
Q: Is the chiral integrity maintained during grinding?
A: Yes, the mechanochemical cyclization occurs under mild room temperature conditions without harsh acidic or basic solutions that typically risk racemization, preserving optical purity.
Q: What are the scalability advantages of this route?
A: Eliminating large volumes of organic solvents reduces reactor volume requirements and safety hazards, allowing for more efficient commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Levetiracetam Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of green chemistry in modern pharmaceutical manufacturing. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative lab-scale methods like this solvent-free route can be successfully translated to industrial reality. Our rigorous QC labs and commitment to stringent purity specifications guarantee that every batch meets the highest international standards. We understand the complexities of chiral synthesis and the critical importance of maintaining optical purity throughout the manufacturing process. Our technical team is ready to collaborate with your R&D department to optimize this pathway for your specific capacity needs.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can enhance your supply chain efficiency. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the potential economic benefits specific to your operation. We encourage you to contact us for specific COA data and route feasibility assessments to verify the compatibility of this method with your current infrastructure. Partnering with us ensures access to cutting-edge technology and a reliable supply of high-quality intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
