Scalable Manufacturing of 3-Aminopyrrolidine Hydrochloride via Novel Aspartic Acid Route
Scalable Manufacturing of 3-Aminopyrrolidine Hydrochloride via Novel Aspartic Acid Route
The pharmaceutical industry constantly seeks robust, scalable, and cost-effective pathways for critical heterocyclic intermediates, particularly those serving as the backbone for next-generation antibiotics. Patent CN112574087A introduces a transformative synthetic methodology for 3-aminopyrrolidine hydrochloride, a pivotal building block in the manufacture of fluoroquinolone antibacterial drugs such as norfloxacin and clinafloxacin. This innovation addresses long-standing inefficiencies in legacy production routes by leveraging a strategic sulfonyl protection mechanism starting from inexpensive aspartic acid diesters. By shifting away from hazardous high-pressure hydrogenation and expensive metal catalysts, this technology offers a streamlined, five-step sequence that operates under remarkably mild conditions. For R&D directors and procurement specialists alike, this represents a significant opportunity to optimize the supply chain for quinolone derivatives, ensuring higher purity standards while drastically reducing the operational risks associated with traditional pyrophoric reagents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 3-aminopyrrolidine has been plagued by complex multi-step sequences that rely on dangerous and costly reagents. One prevalent legacy route involves the addition of ethyl acrylate to benzylamine, followed by Dieckmann cyclization and subsequent reduction using Raney nickel. As illustrated in the reaction scheme below, this pathway is not only lengthy but also necessitates the use of flammable Raney nickel and high-pressure hydrogenation equipment, creating substantial safety hazards and capital expenditure burdens for manufacturing facilities.
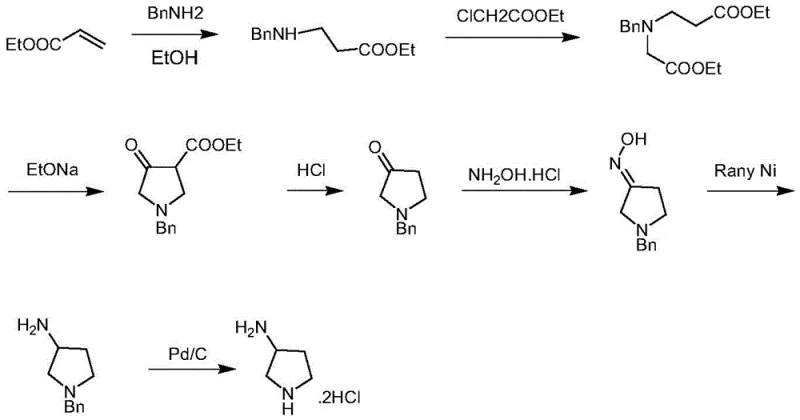
Furthermore, alternative historical methods utilizing 1,4-dichlorobutene or malic acid derivatives often require harsh reducing agents like lithium aluminum hydride or borane complexes. These reagents are moisture-sensitive, difficult to handle on a multi-ton scale, and generate significant volumes of hazardous waste. The reliance on noble metal catalysts for debenzylation in these older routes further inflates the cost of goods sold (COGS) and introduces potential heavy metal contamination risks that complicate regulatory compliance for API manufacturing. Consequently, these conventional approaches struggle to meet the modern demands for green chemistry and economic efficiency in the production of high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast, the novel methodology disclosed in CN112574087A utilizes a clever protection-deprotection strategy centered around aspartic acid diesters. This approach bypasses the need for high-pressure hydrogenation entirely by employing sulfonyl groups to mask the amine functionality during the critical ring-closing steps. The process begins with the straightforward protection of the amino group, followed by a controlled reduction of the ester moieties to alcohols using standard reducing agents like sodium borohydride. This is succeeded by a halogenation step to activate the chain for cyclization. The elegance of this route lies in its modularity; each step proceeds with high conversion rates under ambient or near-ambient pressures, eliminating the need for specialized autoclaves. By replacing expensive benzyl protecting groups with easily removable sulfonyl groups, the process achieves a cleaner deprotection profile in the final step, yielding the target dihydrochloride salt with exceptional purity.
Mechanistic Insights into Sulfonyl-Protection Cyclization
The core of this technological breakthrough is the precise manipulation of the aspartic acid backbone through a series of well-defined chemical transformations. The general reaction pathway, depicted below, highlights the conversion of the linear diester into the cyclic pyrrolidine structure. The initial sulfonylation (Step S1) creates a robust intermediate (Compound I) that withstands the subsequent reduction conditions without premature deprotection. This stability is crucial for maintaining the integrity of the nitrogen atom throughout the synthesis, preventing the formation of polymeric byproducts that often plague direct cyclization attempts.
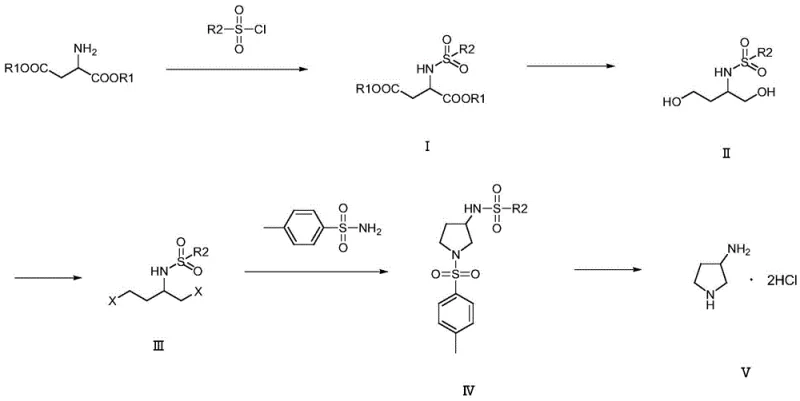
Following the reduction to the diol (Compound II), the halogenation step (Step S3) activates the terminal carbons for nucleophilic attack. The subsequent cyclization (Step S4) involves a double displacement reaction where the sulfonamide nitrogen attacks the halogenated carbons, closing the five-membered ring to form Compound IV. The structural specificity of these intermediates, shown in detail below, ensures that the amino group ends up at the 3-position of the pyrrolidine ring with high regioselectivity. Finally, the acidic hydrolysis in Step S5 cleaves the sulfonyl protecting groups and simultaneously forms the stable hydrochloride salt. This mechanism effectively decouples the ring formation from the amine exposure, allowing for rigorous control over impurity profiles and ensuring that the final product meets the stringent specifications required for antibiotic synthesis.
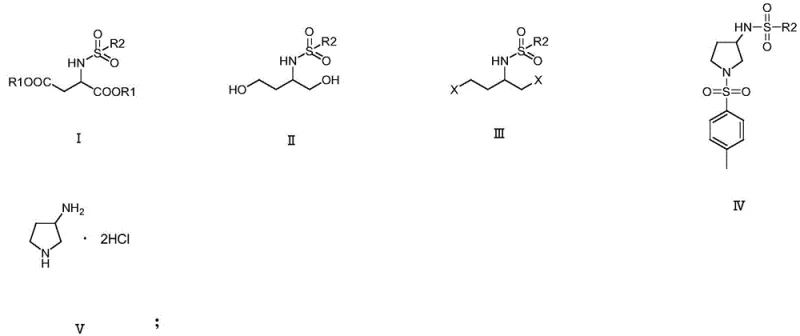
How to Synthesize 3-Aminopyrrolidine Hydrochloride Efficiently
Implementing this synthesis requires careful attention to stoichiometry and temperature control, particularly during the halogenation and cyclization phases. The patent outlines a robust protocol that balances reaction kinetics with safety, utilizing common organic solvents like dichloromethane and ethanol to facilitate easy workup and solvent recovery. The following guide summarizes the critical operational parameters derived from the patent examples, providing a roadmap for process chemists to replicate these high-yield results in a pilot or production setting.
- Protect the amino group of aspartic acid diester using substituted sulfonyl chloride to form Compound I.
- Reduce the ester groups of Compound I to hydroxyl groups using a reducing agent to obtain Compound II.
- Convert the hydroxyl groups of Compound II into halogens using a halogenating reagent to yield Compound III.
- Perform substitution and cyclization with p-toluenesulfonamide to generate the protected pyrrolidine ring (Compound IV).
- Deprotect the sulfonyl groups and salt with hydrochloric acid to obtain the final 3-aminopyrrolidine hydrochloride product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel synthetic route translates directly into enhanced operational resilience and cost predictability. By eliminating the dependency on volatile noble metal markets for palladium or rhodium catalysts, manufacturers can stabilize their raw material costs and avoid the price fluctuations associated with precious metals. Furthermore, the removal of high-pressure hydrogenation steps reduces the need for specialized, high-maintenance reactor infrastructure, allowing for production in standard glass-lined or stainless steel vessels. This flexibility significantly lowers the barrier to entry for scaling production, enabling faster response times to market demand surges for quinolone antibiotics without the lead time associated with commissioning high-pressure units.
- Cost Reduction in Manufacturing: The substitution of expensive reagents like lithium aluminum hydride and Raney nickel with cost-effective alternatives such as sodium borohydride and thionyl chloride drives down the variable cost per kilogram. Additionally, the high yields reported in the patent examples minimize raw material waste, ensuring that a greater proportion of the input aspartic acid is converted into saleable product. This efficiency gain is compounded by the simplified purification processes, which reduce solvent consumption and energy usage during distillation and crystallization, leading to substantial overall savings in utility and waste disposal costs.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals like aspartic acid diesters and p-toluenesulfonamide ensures a secure and diversified supply base. Unlike specialized catalysts that may have single-source suppliers and long lead times, the key inputs for this process are widely available from multiple global vendors. This diversification mitigates the risk of supply disruptions and allows procurement teams to negotiate more favorable terms. Moreover, the mild reaction conditions reduce the likelihood of batch failures due to equipment malfunction or thermal runaway, ensuring consistent on-time delivery of critical intermediates to downstream API manufacturers.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route offers a significantly greener profile. The avoidance of pyrophoric reagents and high-pressure hydrogen gas reduces the facility's safety risk rating, potentially lowering insurance premiums and regulatory scrutiny. The aqueous workups described in the patent facilitate easier separation of organic and inorganic waste streams, simplifying wastewater treatment protocols. As regulatory bodies increasingly demand sustainable manufacturing practices, adopting this lower-impact synthesis method positions companies favorably for future audits and aligns with corporate sustainability goals regarding waste reduction and energy efficiency.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this new synthesis is vital for stakeholders evaluating its integration into existing production lines. The following questions address common concerns regarding scalability, purity, and regulatory compliance, drawing directly from the experimental data and technical disclosures within the patent documentation. These insights are intended to clarify the practical implications of switching from legacy methods to this advanced aspartic acid-based protocol.
Q: What are the primary advantages of the aspartic acid route over traditional methods?
A: The aspartic acid route avoids hazardous reagents like Raney nickel and lithium aluminum hydride, operates under mild conditions, and utilizes cheap, readily available raw materials, significantly lowering production costs and safety risks.
Q: How does this synthesis method impact impurity profiles?
A: By employing specific sulfonyl protection and controlled cyclization steps, the method minimizes side reactions and byproduct formation, resulting in a cleaner crude product that simplifies downstream purification.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process eliminates the need for high-pressure equipment and expensive noble metal catalysts, making it highly scalable and economically viable for commercial production of quinolone intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Aminopyrrolidine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to innovative synthetic routes requires a partner with deep technical expertise and proven scale-up capabilities. Our team of process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN112574087A are fully realized in a GMP-compliant manufacturing environment. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 3-aminopyrrolidine hydrochloride meets the exacting standards required for fluoroquinolone antibiotic synthesis.
We invite pharmaceutical and agrochemical companies to collaborate with us to leverage this cost-effective technology for their supply chains. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to discuss route feasibility assessments and to obtain specific COA data for our pilot batches, ensuring that your transition to this superior manufacturing method is seamless, compliant, and commercially advantageous.
