Advanced Synthesis of Aggregation-Induced Luminescence Nitroxide Radicals for Commercial Scale-Up
Advanced Synthesis of Aggregation-Induced Luminescence Nitroxide Radicals for Commercial Scale-Up
The chemical landscape for functional materials is evolving rapidly, driven by the demand for multifunctional compounds that bridge the gap between electronic performance and biological compatibility. Patent CN110818614A introduces a groundbreaking methodology for synthesizing nitrogen-oxygen stable free radicals that possess aggregation-induced luminescence (AIE) functions. This innovation represents a significant leap forward for manufacturers seeking reliable electronic chemical suppliers who can deliver complex, dual-functionality intermediates. By integrating the robust paramagnetic properties of TEMPO-like radicals with the unique photophysical characteristics of tetraphenylethylene (TPE) derivatives, this technology opens new avenues for the development of advanced fluorescent probes, OLED materials, and smart sensors. The strategic value of this patent lies not only in the novel molecular architecture but also in the practical, scalable synthetic route that avoids the pitfalls of traditional high-energy processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of functionalized free radicals has been plagued by stability issues and complex purification requirements. Traditional methods often rely on harsh oxidative conditions that can degrade sensitive fluorophores or require extreme temperatures that compromise the integrity of the radical center. Furthermore, conventional AIE materials frequently lack the spin-labeling capabilities necessary for advanced biological tracking or controlled polymerization applications. When manufacturers attempt to combine these two distinct functional groups, they often encounter low yields due to incompatible reaction conditions, leading to substantial waste and increased cost reduction challenges in display material manufacturing. The separation of paramagnetic and fluorescent moieties typically results in mixtures that are difficult to purify to the stringent standards required for high-purity optoelectronic material applications, creating bottlenecks in the supply chain for downstream users.
The Novel Approach
The methodology disclosed in CN110818614A offers a transformative solution by decoupling the synthesis of the radical core and the AIE generator before joining them under mild conditions. This modular approach allows for the independent optimization of the tetraphenylethylene scaffold and the nitroxide radical precursor. The key breakthrough is the final nucleophilic substitution step, which proceeds efficiently at room temperature, thereby preserving the delicate balance between the radical stability and the luminescent quantum yield. This strategy effectively eliminates the need for high-thermal stress during the final assembly, drastically simplifying the process control and enhancing the overall reproducibility of the commercial scale-up of complex polymer additives and electronic chemicals. By utilizing a benzyl bromide intermediate for the coupling, the reaction kinetics are favorable, ensuring high conversion rates without the need for exotic catalysts that could contaminate the final product.
Mechanistic Insights into the Modular Synthesis Strategy
The synthetic pathway is elegantly designed to maximize yield while minimizing impurity formation through a four-step sequence. Initially, tetramethylpiperidinol undergoes oxidation using potassium hydrogen persulfate in a biphasic system to generate the hydroxyl-functionalized nitroxide radical (Intermediate 1). Concurrently, the AIE core is constructed via a palladium-catalyzed Suzuki coupling between 2-bromo-1,1,2-triphenylethylene and 4-methylphenylboronic acid, yielding the methyl-substituted tetraphenylethylene (Intermediate 2). This intermediate is then subjected to radical bromination using N-bromosuccinimide (NBS) to install the reactive benzyl bromide handle (Intermediate 3). The culmination of the process involves the nucleophilic attack of the hydroxyl-radical on the benzyl bromide species in the presence of a base like triethylamine. This specific mechanistic pathway ensures that the radical center remains intact while forming a stable ether linkage to the bulky TPE group, which is essential for inducing the aggregation-dependent emission properties.
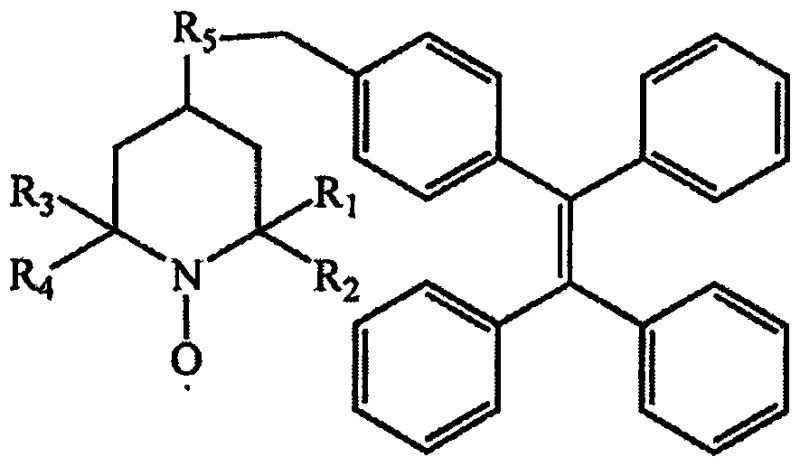
Impurity control is inherently built into this mechanism through the choice of reagents and separation techniques. The use of column chromatography after the Suzuki coupling and bromination steps effectively removes palladium residues and unreacted halides, which are critical contaminants for electronic applications. The final room-temperature substitution minimizes side reactions such as elimination or radical dimerization, which are common failure modes in high-temperature radical chemistry. The resulting product exhibits strong absorption in the UV region and distinct fluorescence emission in the blue-green spectrum when aggregated, confirming the successful integration of the AIE motif. This level of mechanistic precision allows for the production of high-purity OLED material precursors that meet the rigorous specifications demanded by top-tier research and development teams globally.
How to Synthesize Nitroxide Stable Free Radical Efficiently
The operational protocol for producing this advanced material is designed for scalability and safety, leveraging standard organic synthesis unit operations. The process begins with the careful preparation of the radical precursor, followed by the construction of the hydrophobic AIE core, and concludes with their convergence. Detailed standard operating procedures regarding exact stoichiometric ratios, solvent volumes, and workup parameters are critical for maintaining batch-to-batch consistency. For a comprehensive breakdown of the specific reaction conditions, including temperature profiles and purification solvents, please refer to the standardized guide below which outlines the critical process parameters derived from the patent data.
- Oxidize tetramethylpiperidinol using potassium hydrogen persulfate to form the hydroxyl-functionalized nitroxide radical intermediate.
- Perform Suzuki coupling between 2-bromo-1,1,2-triphenylethylene and 4-methylphenylboronic acid using a palladium catalyst to generate the tetraphenylethylene core.
- Conduct radical bromination on the tetraphenylethylene intermediate using N-bromosuccinimide to introduce the reactive benzyl bromide group.
- Execute a room-temperature nucleophilic substitution between the brominated tetraphenylethylene and the hydroxyl-nitroxide radical to finalize the AIE-active product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this synthetic route offers compelling advantages that directly address cost and supply chain volatility. The reliance on commercially available starting materials such as tetramethylpiperidinol and simple boronic acids ensures a robust supply chain that is not dependent on scarce or geopolitically sensitive resources. The elimination of cryogenic conditions or ultra-high pressure reactors in the final steps significantly lowers the capital expenditure required for manufacturing infrastructure. Furthermore, the ability to perform the final coupling at ambient temperature translates to drastic energy savings and reduced cooling loads, contributing to a lower carbon footprint and enhanced sustainability metrics for the end product. These factors collectively position this technology as a highly attractive option for cost reduction in electronic chemical manufacturing.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive transition metal catalysts in the final coupling step, relying instead on simple base-mediated nucleophilic substitution. This removal of precious metals not only lowers raw material costs but also simplifies the downstream purification process, as there is no need for specialized metal scavenging resins or complex chelation treatments. The high atom economy of the substitution reaction further minimizes waste generation, leading to substantial cost savings in waste disposal and solvent recovery operations. Additionally, the mild reaction conditions extend the lifespan of reactor vessels and reduce maintenance downtime, contributing to long-term operational efficiency.
- Enhanced Supply Chain Reliability: By utilizing a modular synthesis strategy, the production of key intermediates can be decoupled, allowing for flexible inventory management and risk mitigation. If one supply line for a specific precursor faces disruption, the other intermediate can still be stockpiled, ensuring continuity of operations. The use of common organic solvents like tetrahydrofuran and ethyl acetate, rather than specialized or hazardous reagents, simplifies logistics and storage requirements. This flexibility ensures reducing lead time for high-purity optoelectronic materials, allowing manufacturers to respond swiftly to fluctuating market demands without compromising on quality or delivery schedules.
- Scalability and Environmental Compliance: The synthetic route is inherently scalable, moving seamlessly from gram-scale laboratory synthesis to multi-kilogram pilot production without significant re-engineering. The absence of highly toxic reagents in the final steps aligns with increasingly stringent environmental regulations, facilitating easier permitting and compliance auditing. The straightforward workup procedures, involving standard liquid-liquid extractions and crystallizations, are easily adaptable to continuous flow chemistry or large-scale batch reactors. This scalability ensures that the commercial scale-up of complex polymer additives and functional materials can be achieved with minimal technical risk and maximum environmental stewardship.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these novel AIE-active radicals. The answers are derived directly from the technical specifications and experimental data provided in the source patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these nuances is critical for evaluating the feasibility of integrating this material into your existing product pipelines or research workflows.
Q: What is the primary advantage of combining nitroxide radicals with tetraphenylethylene?
A: The combination merges the stable paramagnetic properties of nitroxide radicals with the aggregation-induced emission (AIE) characteristics of tetraphenylethylene, enabling dual-functionality for bio-imaging and sensing applications without fluorescence quenching in aggregated states.
Q: Does the final synthesis step require high temperatures or harsh conditions?
A: No, the critical final coupling step is performed at room temperature, which significantly reduces energy consumption and minimizes the risk of thermal decomposition of the sensitive radical species.
Q: How is product purity ensured for electronic or biological applications?
A: The process utilizes column chromatography purification at multiple stages, specifically removing palladium residues from the Suzuki coupling and unreacted starting materials, ensuring high purity suitable for sensitive optoelectronic or biomedical uses.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nitroxide Stable Free Radical Supplier
The technological potential of aggregation-induced luminescence radicals is immense, yet realizing this potential requires a manufacturing partner with deep expertise in complex organic synthesis. NINGBO INNO PHARMCHEM stands ready to support your development goals, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of material meets the exacting standards required for advanced electronic and biomedical applications. We understand the critical nature of supply continuity and are committed to delivering consistent quality that empowers your innovation.
We invite you to engage with our technical team to explore how this novel synthesis route can be optimized for your specific needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of adopting this technology. We encourage you to contact our technical procurement team today to索取 specific COA data and route feasibility assessments, ensuring that your transition to these advanced materials is seamless, efficient, and commercially viable.
