Advanced Ionic Liquid Catalysis for High-Purity Methyl Indole-1-Carboxylate Production
The pharmaceutical and agrochemical industries rely heavily on the precise manipulation of heterocyclic scaffolds, with the indole ring system standing as a cornerstone structure for countless bioactive molecules. A critical step in the synthesis of complex indole-based active pharmaceutical ingredients (APIs) is the temporary protection of the nitrogen atom to prevent unwanted side reactions during subsequent functionalization steps. Patent CN101157650B introduces a groundbreaking preparation method for methyl indole-1-carboxylate and its derivatives, addressing long-standing safety and environmental challenges in this domain. This technology leverages the unique properties of ionic liquids combined with dimethyl carbonate (DMC) to achieve high-yield N-carbomethoxylation under mild conditions. By shifting away from hazardous traditional reagents, this process offers a reliable pharmaceutical intermediate supplier pathway that aligns with modern green chemistry principles. The innovation lies not just in the yield, but in the fundamental restructuring of the reaction environment to minimize toxicity and maximize operational safety, making it an ideal candidate for large-scale commercial adoption.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the N-protection of indoles has been plagued by significant safety and environmental drawbacks that pose severe risks to both personnel and production facilities. Traditional methodologies predominantly utilize highly toxic reagents such as phosgene, methyl chloroformate, or acetyl chloride, which require stringent containment measures due to their lethal nature and potential for causing catastrophic industrial accidents. Furthermore, these processes typically necessitate the use of strong, corrosive bases like sodium hydride (NaH) or sodium amide, which are prone to violent exothermic reactions and pose substantial explosion hazards upon contact with moisture. The reliance on volatile organic solvents such as dichloromethane, DMF, or THF exacerbates the environmental burden, generating large volumes of hazardous waste that require costly disposal protocols. Additionally, the generation of waste acid as a byproduct complicates the downstream purification process, often leading to lower overall recovery rates and increased contamination risks in the final product. These cumulative factors render conventional methods increasingly untenable for modern, sustainability-focused manufacturing environments.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN101157650B revolutionizes the synthesis landscape by employing dimethyl carbonate (DMC) as a dual-function solvent and methylating agent, coupled with task-specific ionic liquids as catalysts. This approach completely eliminates the need for toxic phosgene derivatives and dangerous strong bases, replacing them with a benign, "green" reagent system that significantly enhances operational safety. The use of ionic liquids, such as 1-butyl-3-methylimidazolium hydroxide ([Bmim]OH), provides a tunable reaction medium that facilitates high conversion rates without the corrosion issues associated with inorganic alkalis. As demonstrated in comparative studies, switching from traditional sodium hydroxide to ionic liquid catalysis results in a dramatic improvement in product yield, skyrocketing from a negligible 3% to an impressive 96% under optimized conditions. This paradigm shift not only mitigates environmental impact by preventing waste acid formation but also streamlines the workflow, allowing for easier product isolation and catalyst recycling, thereby establishing a new standard for cost reduction in pharmaceutical intermediate manufacturing.
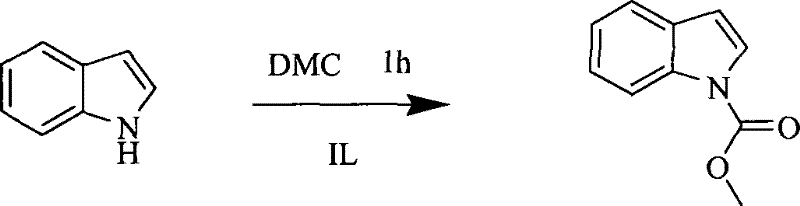
Mechanistic Insights into Ionic Liquid-Catalyzed Carbomethoxylation
The efficacy of this novel synthetic route is rooted in the unique physicochemical properties of the ionic liquid catalyst, which acts as a sophisticated promoter for the nucleophilic attack of the indole nitrogen on the carbonyl carbon of dimethyl carbonate. Unlike traditional bases that deprotonate the indole nitrogen aggressively, potentially leading to decomposition or polymerization, the basic ionic liquid [Bmim]OH creates a localized alkaline environment that gently activates the nucleophile while stabilizing the transition state. The ionic nature of the catalyst ensures excellent solubility for both the organic substrate and the polar intermediates, facilitating a homogeneous reaction phase that maximizes molecular collisions and reaction kinetics. Furthermore, the thermal stability of ionic liquids allows the reaction to proceed at elevated temperatures, up to the boiling point of DMC, without catalyst degradation, which is crucial for overcoming the activation energy barrier of the esterification process. This mechanistic advantage ensures that the reaction proceeds cleanly to the desired methyl indole-1-carboxylate with minimal formation of byproducts, thereby simplifying the impurity profile and reducing the burden on downstream purification units.
From an impurity control perspective, the absence of halogenated reagents and strong inorganic bases fundamentally alters the impurity spectrum of the final product. Conventional methods often leave traces of chloride ions or metal contaminants that are difficult to remove and can interfere with subsequent coupling reactions in API synthesis. The ionic liquid-mediated process, however, generates methanol and carbon dioxide as the primary byproducts, both of which are easily removed or are benign. The general reaction scheme illustrates the clean transformation of the indole scaffold into the protected ester, highlighting the atom economy of the process where the methyl group is transferred efficiently without extraneous leaving groups. This high level of chemical fidelity is paramount for R&D teams focused on developing robust, scalable processes for high-purity OLED material or pharmaceutical intermediates where trace metal limits are strictly regulated. The ability to tune the cation and anion of the ionic liquid further allows chemists to optimize the reaction for specific substituted indoles, ensuring consistent quality across a diverse range of substrates.
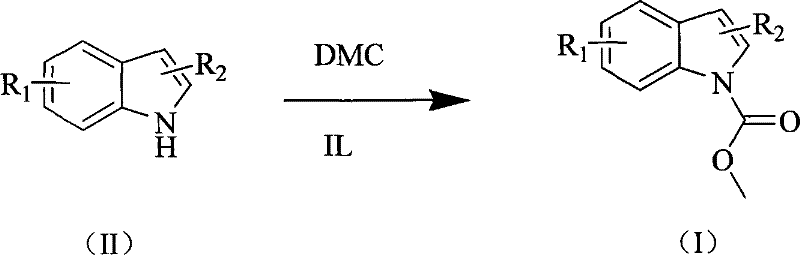
How to Synthesize Methyl Indole-1-Carboxylate Efficiently
The practical implementation of this technology involves a straightforward procedure that dissolves the indole substrate directly in dimethyl carbonate, followed by the addition of a catalytic quantity of the ionic liquid. The reaction mixture is then heated to a preferred temperature range of 80°C to 90°C, where it is maintained for a duration sufficient to achieve complete conversion, typically around one hour for unsubstituted indoles. This operational simplicity belies the sophisticated chemistry occurring within the reactor, where the ionic liquid orchestrates the transfer of the carbomethoxy group with high precision. For those seeking to implement this protocol in a pilot or production setting, the detailed standardized synthesis steps provided below outline the precise stoichiometry, temperature controls, and workup procedures necessary to replicate the high yields reported in the patent literature.
- Dissolve indole or substituted indole compounds in dimethyl carbonate (DMC) acting as both solvent and reagent.
- Add a catalytic amount of ionic liquid, preferably [Bmim]OH, to the reaction mixture.
- Heat the mixture to 90°C for 1 hour, then cool, extract with ether, and purify to obtain the target ester.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this ionic liquid-catalyzed process represents a strategic opportunity to optimize both cost structures and supply reliability. By transitioning to a reagent system based on dimethyl carbonate, organizations can eliminate the procurement complexities and security risks associated with controlled substances like phosgene, thereby reducing administrative overhead and insurance costs. The elimination of hazardous waste streams, specifically waste acid and heavy metal residues, translates directly into substantial cost savings related to waste treatment and environmental compliance, enhancing the overall economic viability of the manufacturing process. Moreover, the recyclability of the ionic liquid catalyst means that the effective consumption of this high-value additive is minimized over multiple batches, further driving down the variable cost per kilogram of the final product.
- Cost Reduction in Manufacturing: The replacement of expensive and dangerous reagents with commodity-grade dimethyl carbonate significantly lowers raw material costs while simultaneously reducing the capital expenditure required for specialized corrosion-resistant equipment. The process avoids the need for cryogenic conditions often required for sensitive organometallic reactions, allowing for energy savings through the use of standard heating systems. Furthermore, the high selectivity of the reaction minimizes the loss of valuable starting materials to side products, ensuring that the theoretical yield is closely approached in practice, which is a critical factor in maximizing return on investment for high-value intermediates.
- Enhanced Supply Chain Reliability: Dimethyl carbonate is a widely available bulk chemical with a stable global supply chain, reducing the risk of production stoppages due to reagent shortages that often plague specialty chemical markets. The robustness of the ionic liquid catalyst against moisture and air exposure simplifies storage and handling requirements, ensuring that the production line remains resilient even under less-than-ideal logistical conditions. This reliability is essential for maintaining continuous supply to downstream API manufacturers, preventing bottlenecks that could delay drug development timelines or market launches.
- Scalability and Environmental Compliance: The inherent safety of the process, characterized by the absence of explosion hazards and toxic gas evolution, facilitates easier scale-up from laboratory to commercial production scales without the need for extensive re-engineering of safety systems. The green nature of the chemistry aligns perfectly with increasingly stringent global environmental regulations, future-proofing the manufacturing asset against potential regulatory crackdowns on volatile organic compounds and hazardous waste. This compliance advantage not only protects the company's reputation but also ensures uninterrupted operations in regions with strict environmental oversight.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this ionic liquid-catalyzed synthesis method. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on reaction conditions, substrate compatibility, and expected outcomes. Understanding these nuances is critical for process chemists evaluating the feasibility of this route for their specific target molecules.
Q: Why is dimethyl carbonate (DMC) preferred over phosgene for indole protection?
A: DMC is a non-toxic, green reagent that eliminates the severe safety hazards and waste acid generation associated with highly toxic phosgene and corrosive methyl chloroformate.
Q: What is the advantage of using ionic liquids like [Bmim]OH in this synthesis?
A: Ionic liquids provide superior catalytic activity with yields exceeding 96%, are recyclable, and avoid the explosion risks and corrosion associated with traditional strong bases like sodium hydride.
Q: Can this method be applied to sterically hindered indole derivatives?
A: Yes, while unsubstituted indoles react rapidly, sterically hindered substrates like 2-methylindole can still be converted effectively by extending reaction times, demonstrating the method's robustness.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Methyl Indole-1-Carboxylate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of green chemistry technologies like the one described in patent CN101157650B to redefine the standards of quality and efficiency in fine chemical manufacturing. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this innovative ionic liquid process can be fully realized at an industrial level. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications for every batch of methyl indole-1-carboxylate and its derivatives we produce. We are committed to delivering high-purity pharmaceutical intermediates that meet the exacting demands of the global healthcare industry.
We invite forward-thinking partners to collaborate with us to leverage this advanced synthesis technology for their next-generation drug candidates. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and project timelines. We encourage you to reach out today to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your supply chain is built on a foundation of safety, sustainability, and scientific excellence.
