Scalable Green Synthesis of Bioactive Lignan Intermediates Using Heterogeneous Photocatalysis
Introduction to Advanced Lignan Synthesis Technology
The pharmaceutical and fine chemical industries are constantly seeking more efficient and sustainable routes to access bioactive scaffolds, particularly lignans, which serve as critical precursors for anticancer and antiviral agents. Patent CN113603578A introduces a groundbreaking methodology for the synthesis of lignan compounds, utilizing a heterogeneous Ag/Ag3PO4 photocatalyst under visible light irradiation. This technology addresses the long-standing challenges associated with the total synthesis of complex tetrahydronaphthalene cyclolignans, such as podophyllotoxin analogues, which are renowned for their potent biological activities including anti-tumor and anti-hepatitis virus properties. 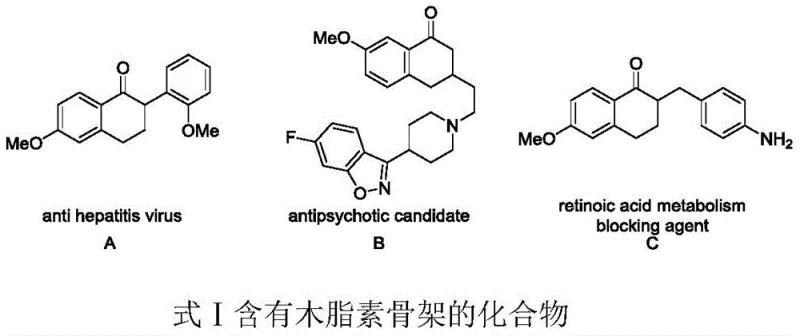 By leveraging oxidative cyclization of electron-rich aromatic vinyl compounds, this process achieves high yields and exceptional regioselectivity without the need for toxic additives or extreme reaction conditions, marking a significant leap forward for reliable lignan intermediate suppliers aiming to modernize their production capabilities.
By leveraging oxidative cyclization of electron-rich aromatic vinyl compounds, this process achieves high yields and exceptional regioselectivity without the need for toxic additives or extreme reaction conditions, marking a significant leap forward for reliable lignan intermediate suppliers aiming to modernize their production capabilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the lignan skeleton has relied heavily on classical Friedel-Crafts cyclization or stoichiometric oxidation methods, both of which suffer from severe drawbacks in a modern manufacturing context. Traditional Friedel-Crafts approaches often necessitate the use of large quantities of corrosive Lewis acids and specialized, difficult-to-synthesize starting materials, leading to complex workup procedures and significant environmental hazards due to acidic waste streams. Furthermore, existing photocatalytic methods utilizing homogeneous transition metal complexes, such as ruthenium or iridium salts, present substantial economic and operational barriers; these catalysts are prohibitively expensive, often costing over 1000 yuan per gram, and their removal from the final product requires rigorous and costly purification steps to meet stringent pharmaceutical purity standards. 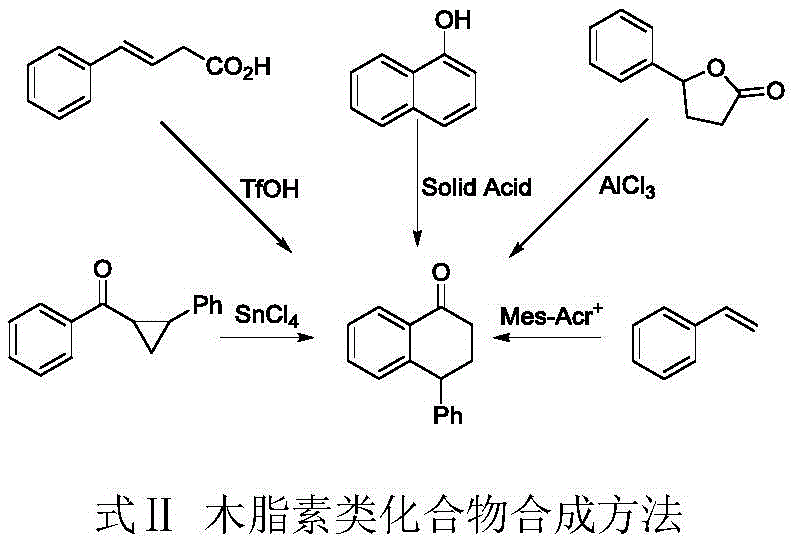 Additionally, some prior art methods require high-energy ultraviolet light or elevated temperatures, which not only increases energy consumption but also poses risks of substrate decomposition and poor selectivity, ultimately limiting the commercial viability of these routes for cost reduction in pharmaceutical intermediates manufacturing.
Additionally, some prior art methods require high-energy ultraviolet light or elevated temperatures, which not only increases energy consumption but also poses risks of substrate decomposition and poor selectivity, ultimately limiting the commercial viability of these routes for cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a robust heterogeneous Ag/Ag3PO4 catalyst that operates efficiently under mild visible light irradiation, typically at wavelengths around 475 nm and temperatures as low as 0°C. This method eliminates the need for expensive homogeneous metal complexes and stoichiometric oxidants, relying instead on molecular oxygen from the air as the terminal oxidant, which is both atom-economical and environmentally benign. The heterogeneous nature of the catalyst allows for straightforward separation via simple filtration, enabling catalyst recycling and significantly reducing the overall process mass intensity (PMI). This shift from homogeneous to heterogeneous photocatalysis represents a paradigm shift for the commercial scale-up of complex polymer additives and pharmaceutical intermediates, offering a pathway to high-purity products with minimal metal contamination and simplified downstream processing.
Mechanistic Insights into Ag/Ag3PO4 Catalyzed Oxidative Cyclization
The core of this technological advancement lies in the unique mechanistic pathway facilitated by the Ag/Ag3PO4 heterojunction under visible light excitation. Upon irradiation, the semiconductor component generates electron-hole pairs, where the photogenerated holes possess sufficient oxidative potential to activate the electron-rich styrene substrates. This activation leads to the formation of radical cation intermediates, which subsequently undergo a highly selective [4+2] cycloaddition or oxidative dimerization to construct the tetralone core. The presence of silver nanoparticles on the phosphate surface enhances charge separation efficiency and extends the lifetime of the reactive species, thereby driving the reaction to completion with superior conversion rates compared to bare semiconductor materials. 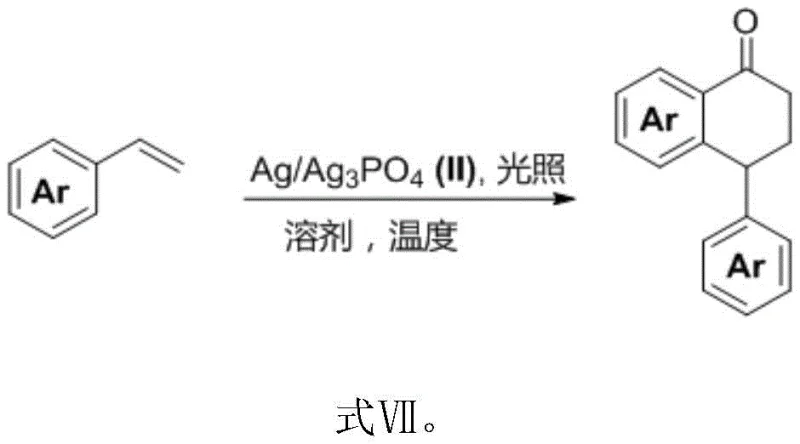 This precise control over the radical mechanism ensures excellent regioselectivity, favoring the formation of the desired alpha-aryl-tetralone isomers while suppressing the formation of unwanted oligomers or polymeric byproducts that often plague free-radical reactions.
This precise control over the radical mechanism ensures excellent regioselectivity, favoring the formation of the desired alpha-aryl-tetralone isomers while suppressing the formation of unwanted oligomers or polymeric byproducts that often plague free-radical reactions.
From an impurity control perspective, the mild reaction conditions play a pivotal role in maintaining product integrity. Unlike thermal methods that can induce rearrangement or degradation of sensitive functional groups, this photochemical process preserves delicate substituents such as ethers and esters on the aromatic rings. The absence of strong Lewis acids prevents side reactions like demethylation or skeletal rearrangements, resulting in a cleaner crude reaction profile. This inherent selectivity reduces the burden on purification teams, allowing for simpler chromatographic separations or even crystallization-based purification strategies, which is essential for achieving the high-purity lignan intermediates required for clinical applications.
How to Synthesize Lignan Compounds Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing high-value lignan derivatives with consistent quality. The process begins with the preparation of the electron-rich styrene precursor, which can be readily accessed via standard Wittig olefination of corresponding aromatic aldehydes. The key innovation lies in the subsequent photocatalytic step, where the precursor is subjected to visible light irradiation in the presence of the recyclable Ag/Ag3PO4 catalyst and oxygen. This standardized approach minimizes variability and ensures reproducibility across different batches, making it highly suitable for industrial adoption. For detailed operational parameters and safety guidelines, please refer to the standardized synthesis steps provided below.
- Prepare the reaction mixture by mixing electron-rich aromatic vinyl compounds (Compound A) with the Ag/Ag3PO4 catalyst in acetonitrile solvent under an oxygen atmosphere.
- Cool the reaction system to 0°C and irradiate with a visible light source (preferably 475 nm LED) while maintaining oxygen supplementation for 8 to 20 hours.
- Upon completion, filter the heterogeneous catalyst, concentrate the filtrate, and purify the crude product via column chromatography to obtain high-purity lignan derivatives.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this Ag/Ag3PO4 photocatalytic technology offers transformative benefits that directly impact the bottom line and operational resilience. By replacing expensive homogeneous catalysts with a reusable heterogeneous system, manufacturers can achieve significant cost savings in raw material procurement and waste disposal. The elimination of toxic Lewis acids and stoichiometric oxidants not only reduces the cost of reagents but also simplifies regulatory compliance regarding hazardous waste management, thereby lowering the total cost of ownership for the production facility.
- Cost Reduction in Manufacturing: The transition to a heterogeneous photocatalyst fundamentally alters the cost structure of lignan production. Since the Ag/Ag3PO4 catalyst can be recovered and reused multiple times without significant loss of activity, the effective cost per kilogram of catalyst consumed drops dramatically compared to single-use ruthenium complexes. Furthermore, the reaction proceeds under ambient pressure and low temperature, eliminating the need for specialized high-pressure reactors or energy-intensive heating systems, which translates to substantially reduced utility costs and capital expenditure on equipment.
- Enhanced Supply Chain Reliability: The reliance on commercially available, electron-rich styrene derivatives as starting materials ensures a stable and diversified supply base, mitigating the risk of bottlenecks associated with exotic or custom-synthesized precursors. The robustness of the reaction conditions means that production is less susceptible to fluctuations in environmental controls, ensuring consistent output and reliable delivery schedules for downstream API manufacturers. This stability is crucial for maintaining continuous supply chains for critical pharmaceutical intermediates.
- Scalability and Environmental Compliance: The green chemistry principles embedded in this process, such as the use of oxygen as an oxidant and the generation of minimal waste, align perfectly with increasingly strict global environmental regulations. The scalability of flow chemistry adaptations for photocatalytic reactions further enhances the potential for large-scale production, allowing manufacturers to ramp up from pilot to commercial scale with confidence. This environmental compatibility reduces the risk of regulatory shutdowns and enhances the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic synthesis route. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: What are the advantages of using Ag/Ag3PO4 over traditional Lewis acid catalysts for lignan synthesis?
A: Unlike traditional Friedel-Crafts reactions that require harsh conditions, large amounts of corrosive Lewis acids, and high temperatures, the Ag/Ag3PO4 photocatalytic method operates at mild temperatures (0°C) under visible light. It eliminates the need for stoichiometric additives, significantly reducing waste generation and simplifying post-reaction purification.
Q: Can the Ag/Ag3PO4 catalyst be recycled for industrial scale-up?
A: Yes, a key advantage of this heterogeneous photocatalyst is its recyclability. The solid catalyst can be easily separated from the reaction mixture via simple filtration, washed, and reused, which drastically lowers the cost of goods sold (COGS) compared to expensive homogeneous ruthenium or iridium complexes that are difficult to recover.
Q: What is the regioselectivity of this oxidative cyclization reaction?
A: The method exhibits excellent regioselectivity, specifically favoring the formation of alpha-aryl-tetralone structures. This precision minimizes the formation of structural isomers and byproducts, ensuring a cleaner impurity profile which is critical for pharmaceutical intermediate manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lignan Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of this photocatalytic technology to revolutionize the supply of high-value lignan intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of materials. Our state-of-the-art facilities are equipped with advanced photocatalytic reactors and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of lignan compound delivered meets the highest industry standards for pharmaceutical applications.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this greener methodology. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, and let us demonstrate how our expertise can accelerate your drug development timeline while optimizing your manufacturing costs.
