Advanced Palladium-Catalyzed Asymmetric Hydrogenation for High-Purity Chiral Gamma-Sultam Manufacturing
Advanced Palladium-Catalyzed Asymmetric Hydrogenation for High-Purity Chiral Gamma-Sultam Manufacturing
The pharmaceutical and agrochemical industries are constantly seeking more efficient, sustainable, and cost-effective routes to access high-value chiral intermediates. A pivotal advancement in this domain is detailed in patent CN101260085A, which discloses a highly enantioselective method for synthesizing chiral gamma-sultams via palladium-catalyzed asymmetric hydrogenation. This technology addresses critical bottlenecks in the production of sulfonamide-containing heterocycles, which are privileged structures found in numerous bioactive molecules. By leveraging a homogeneous palladium system with chiral diphosphine ligands, this method achieves exceptional stereocontrol, offering a robust alternative to legacy synthetic strategies. For R&D directors and procurement specialists, understanding this catalytic breakthrough is essential for optimizing supply chains and reducing the cost of goods for complex heterocyclic APIs.
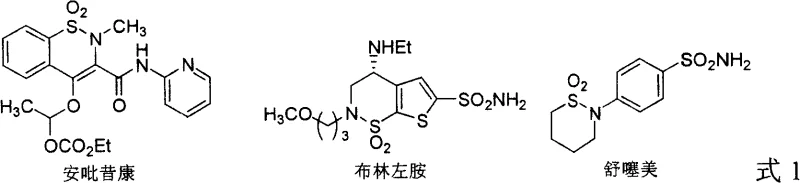
The significance of the gamma-sultam scaffold cannot be overstated, as it serves as a core structural unit in a wide array of therapeutic agents and agricultural reagents. As illustrated in the chemical structures provided, compounds like Anitazepine and Brinzolamide rely on this specific heterocyclic motif for their physiological activity. Beyond their direct application as active pharmaceutical ingredients (APIs), these chiral sultams function effectively as chiral auxiliaries and reagents in asymmetric synthesis, further amplifying their commercial value. The ability to produce these scaffolds with high optical purity is therefore a strategic imperative for any organization aiming to secure a reliable pharmaceutical intermediate supplier partnership. The patent highlights that while many compounds containing cyclic sulfonamide functional groups possess significant biological potential, the stereoselective synthesis of these molecules has historically remained a formidable challenge for process chemists.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral sultams has been plagued by reliance on the "chiral pool" or stoichiometric chiral auxiliaries, which impose severe limitations on structural diversity and economic efficiency. For instance, earlier methodologies reported by researchers such as Lee et al. utilized beta-chiral amino alcohols as starting materials, requiring multi-step sequences including intramolecular alkylation to close the ring. The fundamental drawback of this approach is that the stereochemical outcome of the final product is strictly dictated by the chirality of the expensive natural starting material. This dependency restricts the accessible chemical space to only those derivatives that can be sourced from limited natural amino alcohols, thereby hindering the exploration of novel analogs. Furthermore, other approaches involving nucleophilic addition to carbon-nitrogen double bonds, as seen in Enders' work, often require the installation and subsequent removal of chiral auxiliaries. These extra steps not only increase the overall process mass intensity (PMI) but also introduce additional opportunities for yield loss and impurity generation, making them less attractive for cost reduction in pharmaceutical intermediate manufacturing.
The Novel Approach
In stark contrast, the method disclosed in patent CN101260085A represents a paradigm shift by employing catalytic asymmetric hydrogenation, a technique renowned for its atom economy and operational simplicity. Instead of relying on stoichiometric chiral sources, this novel approach utilizes a catalytic amount of a chiral palladium-diphosphine complex to induce asymmetry directly onto the prochiral cyclic sulfonimide substrate. This strategy decouples the source of chirality from the substrate structure, allowing for the flexible synthesis of various substituted gamma-sultams simply by changing the configuration of the ligand. The reaction proceeds under relatively mild conditions, utilizing hydrogen gas as the reductant, which generates no toxic byproducts other than the desired saturated product. This green chemistry profile, combined with the ability to achieve high enantiomeric excess values ranging from 79% to 93%, positions this technology as a superior choice for the commercial scale-up of complex pharmaceutical intermediates. The elimination of auxiliary removal steps significantly streamlines the workflow, enhancing both throughput and sustainability.
Mechanistic Insights into Pd-Catalyzed Asymmetric Hydrogenation
The core of this technological breakthrough lies in the sophisticated interplay between the palladium metal center and the chiral diphosphine ligand within a homogeneous catalytic cycle. The reaction initiates with the formation of an active palladium-hydride species, generated in situ from a palladium precursor such as palladium trifluoroacetate and the chiral ligand in a solvent like acetone. This active catalyst then coordinates with the cyclic sulfonimide substrate, specifically interacting with the imine double bond. The chiral environment created by the bulky diphosphine ligand, such as (S)-SegPhos or (S)-SYNPHOS, dictates the facial selectivity of the hydrogen addition. As the hydrogen molecule adds across the carbon-nitrogen double bond, the steric and electronic properties of the ligand ensure that one enantiomer is formed preferentially over the other. The use of 2,2,2-trifluoroethanol (TFE) as the solvent plays a crucial role, likely stabilizing the transition state through hydrogen bonding interactions and enhancing the solubility of the polar intermediates. This precise control over the stereochemical outcome allows for the production of chiral gamma-sultams with defined absolute configurations, a critical requirement for regulatory compliance in drug development.
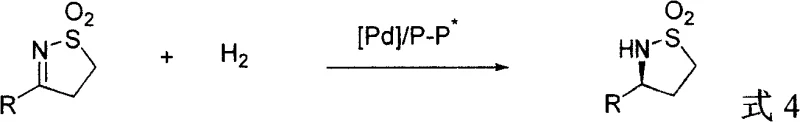
Furthermore, the mechanistic robustness of this system contributes significantly to impurity control, a primary concern for R&D directors overseeing process validation. The high specificity of the catalytic hydrogenation minimizes side reactions such as over-reduction or ring-opening, which are common pitfalls in non-selective hydrogenation processes. The patent data indicates that proton NMR analysis of the crude reaction mixtures often reveals no detectable side products, underscoring the cleanliness of the transformation. This high level of chemoselectivity simplifies downstream processing, as the crude product can often be purified directly via column chromatography or crystallization without extensive remediation. The ability to tune the absolute configuration of the product merely by switching the enantiomer of the chiral ligand provides a powerful tool for generating both enantiomers of a drug candidate for toxicological studies. Such flexibility is invaluable during the early stages of drug discovery and process optimization, ensuring that the synthetic route remains viable regardless of the final stereochemical requirement of the API.
How to Synthesize Chiral Gamma-Sultam Efficiently
Implementing this catalytic asymmetric hydrogenation protocol requires careful attention to catalyst preparation and reaction parameters to maximize both yield and enantioselectivity. The process begins with the in situ generation of the active catalyst, where a palladium metal precursor and a chiral diphosphine ligand are stirred in acetone at room temperature, followed by vacuum concentration to remove the solvent. This pre-formed catalyst is then introduced to a solution of the cyclic sulfonimide substrate in 2,2,2-trifluoroethanol under an inert atmosphere. The reaction mixture is subsequently pressurized with hydrogen gas and heated to facilitate the transformation. The detailed standardized synthesis steps, including specific molar ratios, temperature ranges, and workup procedures, are outlined in the guide below to ensure reproducibility and safety during scale-up operations.
- Prepare the catalyst by stirring palladium trifluoroacetate and a chiral diphosphine ligand (e.g., (S)-SegPhos) in acetone at room temperature for 0.5-1 hour, followed by vacuum concentration.
- Dissolve the cyclic sulfonimide substrate in 2,2,2-trifluoroethanol (TFE) within a reaction vessel and add the prepared catalyst solution under a nitrogen atmosphere.
- Pressurize the reactor with hydrogen gas to 35-40 atmospheres and maintain the reaction at 25-75°C for 10-12 hours to achieve high conversion and enantioselectivity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this palladium-catalyzed hydrogenation technology offers substantial strategic benefits that extend beyond mere technical feasibility. The shift from stoichiometric chiral auxiliaries to a catalytic system fundamentally alters the cost structure of the manufacturing process. By eliminating the need for expensive, single-use chiral starting materials and the associated waste disposal costs, the overall cost of goods sold (COGS) can be significantly reduced. Additionally, the simplified workflow, which avoids multiple protection and deprotection steps, shortens the production cycle time, thereby enhancing the responsiveness of the supply chain to market demands. The use of hydrogen gas as a clean reductant aligns with increasingly stringent environmental regulations, reducing the burden of waste treatment and facilitating smoother regulatory approvals for new manufacturing sites. These factors collectively contribute to a more resilient and cost-efficient supply chain for high-value heterocyclic intermediates.
- Cost Reduction in Manufacturing: The transition to a catalytic asymmetric hydrogenation process eliminates the reliance on costly stoichiometric chiral pools, which traditionally account for a significant portion of raw material expenses. By using a small amount of recyclable or highly active palladium catalyst to generate large quantities of chiral product, the material cost per kilogram of the intermediate is drastically lowered. Furthermore, the high atom economy of the hydrogenation reaction means that fewer atoms are wasted as byproducts, reducing the load on purification systems and minimizing solvent consumption. This lean manufacturing approach ensures that the production of chiral gamma-sultams remains economically viable even at large scales, providing a competitive edge in pricing for downstream API manufacturers.
- Enhanced Supply Chain Reliability: Dependence on natural chiral sources often introduces volatility into the supply chain due to agricultural fluctuations or geopolitical constraints on raw material sourcing. This synthetic method circumvents such risks by utilizing readily available achiral precursors and commercially accessible chiral ligands. The robustness of the reaction conditions, which tolerate a range of temperatures and pressures, ensures consistent batch-to-batch quality and reduces the likelihood of production delays caused by sensitive process parameters. Consequently, partners can rely on a stable and continuous supply of high-purity intermediates, safeguarding their own production schedules against upstream disruptions and ensuring timely delivery to global markets.
- Scalability and Environmental Compliance: The green chemistry principles embedded in this process, such as the use of molecular hydrogen and the generation of minimal waste, make it highly scalable from laboratory to industrial production. The absence of heavy metal waste streams associated with stoichiometric reagents simplifies effluent treatment and lowers environmental compliance costs. Moreover, the mild reaction conditions reduce energy consumption compared to high-temperature or cryogenic alternatives, further enhancing the sustainability profile of the manufacturing operation. This alignment with global sustainability goals not only mitigates regulatory risk but also enhances the brand reputation of companies adopting this technology as responsible chemical manufacturers committed to eco-friendly practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric hydrogenation technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of the process capabilities. By addressing concerns related to stereocontrol, reaction conditions, and scalability, we aim to facilitate informed decision-making for technical teams evaluating this route for their specific project needs. The answers reflect the proven performance of the catalyst system in generating diverse chiral sultam derivatives with high fidelity.
Q: What are the key advantages of this Pd-catalyzed method over traditional chiral auxiliary routes?
A: Unlike traditional methods that rely on stoichiometric chiral starting materials which limit structural diversity, this catalytic asymmetric hydrogenation uses a tunable chiral ligand to induce chirality. This allows for the synthesis of various substituted gamma-sultams with high enantiomeric excess (up to 93% ee) without being restricted by the availability of natural chiral pools.
Q: What are the optimal reaction conditions for maximizing yield and selectivity?
A: The patent specifies that optimal results are achieved using 2,2,2-trifluoroethanol as the solvent under a hydrogen pressure of 35-40 atmospheres. The reaction temperature should be maintained between 25-75°C, typically running for 10-12 hours, with a substrate-to-catalyst molar ratio of approximately 40-50:1 to ensure high conversion rates.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for scalability. It features mild reaction conditions, simple operation, and easy product separation via column chromatography or crystallization. The use of a robust palladium catalyst system and the absence of complex multi-step chiral induction sequences make it highly amenable to commercial scale-up for pharmaceutical intermediate manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Gamma-Sultam Supplier
The technological potential of palladium-catalyzed asymmetric hydrogenation for synthesizing chiral gamma-sultams is immense, offering a pathway to high-value intermediates with superior purity and efficiency. At NINGBO INNO PHARMCHEM, we pride ourselves on our capability to translate such innovative academic and patent technologies into robust commercial processes. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can seamlessly transition from pilot scale to full manufacturing. We adhere to stringent purity specifications and operate rigorous QC labs equipped with advanced chiral HPLC and NMR instrumentation to guarantee that every batch meets the exacting standards required by the global pharmaceutical industry. Our commitment to quality and scalability makes us the ideal partner for your complex heterocycle synthesis needs.
We invite you to engage with our technical procurement team to discuss how this advanced hydrogenation route can be tailored to your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits of switching to this catalytic method for your specific target molecule. We encourage potential partners to reach out for specific COA data and comprehensive route feasibility assessments to validate the suitability of this technology for your upcoming projects. Let us collaborate to optimize your synthesis strategy and secure a competitive advantage in the marketplace through superior chemical innovation and reliable supply.
