Advancing Pharmaceutical Intermediate Synthesis with Regioselective Ruthenium-Catalyzed 7-Amide Indole Production
The pharmaceutical industry continuously seeks robust and efficient synthetic routes to access complex heterocyclic scaffolds, particularly indole derivatives which serve as privileged structures in numerous bioactive molecules. A significant technological breakthrough in this domain is documented in Chinese Patent CN111892528A, which discloses a novel preparation method for 7-amide indole compounds. This innovation addresses a long-standing challenge in organic synthesis: the direct, regioselective functionalization of the indole benzene ring, specifically at the sterically hindered C7 position. Traditional methods often suffer from poor selectivity, favoring the more reactive pyrrole ring (C2/C3 positions), or require multi-step sequences involving protection and deprotection strategies. The disclosed technology utilizes a ruthenium-catalyzed C-H activation strategy, employing readily available oxazolone compounds as efficient amidating agents. This approach not only streamlines the synthetic pathway but also operates under mild conditions, making it highly attractive for the production of high-purity pharmaceutical intermediates. For research and development teams focused on drug discovery, this method offers a powerful tool to rapidly diversify indole libraries, potentially accelerating the identification of new therapeutic candidates with improved pharmacological profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, achieving regioselective substitution at the C7 position of the indole nucleus has been a formidable obstacle for synthetic chemists. Early strategies, such as those reported by the Hartwig research group, relied on iridium-catalyzed borylation directed by silyl groups, followed by subsequent oxidation steps to install functionality. While effective, these multi-step protocols introduce significant inefficiencies, including increased material costs, longer processing times, and cumulative yield losses associated with each isolation step. Furthermore, alternative approaches involving the reduction of indoles to indolines, followed by functionalization and re-oxidation, add unnecessary complexity and redox steps that are undesirable for large-scale manufacturing. Other methods utilizing acyl azides with iridium catalysts have shown limited success, often reporting modest yields ranging from 26% to 55%, which is economically unviable for commercial production. These conventional pathways frequently demand stringent reaction conditions, such as elevated temperatures or strictly inert atmospheres, which complicate process safety and increase capital expenditure for specialized reactor equipment. The reliance on expensive noble metal catalysts like iridium, coupled with the need for specialized directing groups that must later be removed, creates a substantial bottleneck in the supply chain for complex indole-based active pharmaceutical ingredients.
The Novel Approach
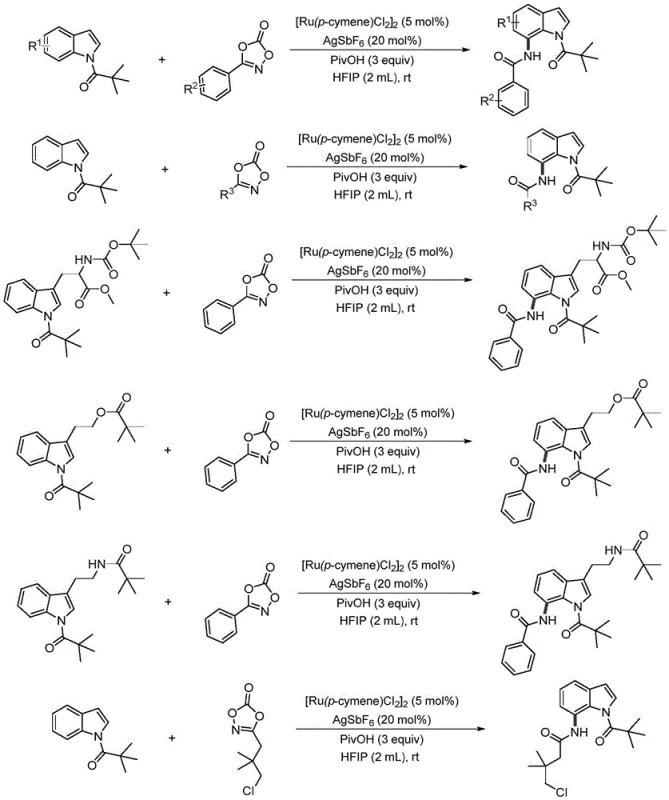
In stark contrast to these legacy methods, the technology described in patent CN111892528A introduces a direct, one-step amidation protocol that bypasses the need for pre-functionalization or redox manipulations. By leveraging a specific ruthenium catalyst system composed of [Ru(p-cymene)Cl2]2 and AgSbF6, the process achieves exceptional regioselectivity for the C7 position directly on the indole core. The use of oxazolones as the nitrogen source is particularly advantageous, as they act as safe and highly reactive amidating reagents that release carbon dioxide as the only byproduct, driving the reaction forward thermodynamically. This novel approach operates at room temperature in an air atmosphere, drastically simplifying the operational requirements compared to sensitive transition metal catalyses that demand glovebox techniques. The reaction demonstrates remarkable efficiency, with optimized examples yielding up to 70% of the desired 7-amide indole product after simple purification. This streamlined workflow significantly reduces the environmental footprint by minimizing solvent usage and waste generation, aligning with modern green chemistry principles. For procurement and supply chain managers, this translates to a more reliable and cost-effective sourcing strategy for critical intermediates, reducing the risk of production delays associated with complex multi-step syntheses.
Mechanistic Insights into Ru-Catalyzed C-H Amidation
The success of this transformation lies in the intricate interplay between the ruthenium catalyst and the substrate, facilitated by a well-defined catalytic cycle. The process initiates with the generation of an active cationic ruthenium species through the reaction of the dimeric precursor [Ru(p-cymene)Cl2]2 with the silver salt AgSbF6 in the presence of pivalic acid. This active cation coordinates with the carbonyl oxygen of the pivaloyl protecting group on the indole nitrogen, acting as a weakly coordinating directing group. This coordination is crucial as it positions the metal center in proximity to the C7-H bond, enabling selective carbon-hydrogen activation to form a stable six-membered ruthenacycle intermediate. Subsequently, the oxazolone substrate coordinates to the ruthenium center via its nitrogen atom. This coordination triggers the oxidative addition and subsequent ring-opening of the oxazolone, accompanied by the extrusion of carbon dioxide, to generate a high-valent ruthenium(IV) amide intermediate. The mechanistic elegance continues with a migratory insertion of the amide bond into the ruthenium-carbon bond, expanding the metallacycle to a seven-membered ring structure. Finally, protonolysis of this intermediate releases the final 7-amide indole product and regenerates the active ruthenium catalyst, allowing the cycle to continue. This detailed understanding of the mechanism allows for precise tuning of reaction parameters to maximize turnover numbers and minimize catalyst loading, ensuring consistent quality in the final product.
Controlling the impurity profile is paramount for pharmaceutical applications, and this catalytic system offers inherent advantages in this regard. The high regioselectivity imposed by the directing group mechanism effectively suppresses the formation of C2 or C3 substituted byproducts, which are common contaminants in non-directed indole functionalizations. Furthermore, the mild reaction conditions prevent the degradation of sensitive functional groups that might be present on complex drug-like molecules. The use of pivalic acid as an additive plays a dual role: it assists in the C-H activation step via a concerted metalation-deprotonation (CMD) pathway and helps maintain the solubility of the catalytic species. By avoiding harsh oxidants or strong bases, the process minimizes side reactions such as polymerization or over-oxidation of the indole core. This results in a cleaner crude reaction mixture, which simplifies downstream purification processes like column chromatography or crystallization. For quality control laboratories, this means fewer impurities to track and validate, reducing the analytical burden and accelerating the release of batches for clinical or commercial use. The robustness of the catalyst system against air and moisture further ensures batch-to-batch reproducibility, a critical factor for maintaining stringent purity specifications required by regulatory agencies.
How to Synthesize 7-Amide Indole Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires adherence to specific stoichiometric ratios and procedural steps to achieve the reported high yields. The standard protocol involves dissolving the N-pivaloyl indole substrate and the oxazolone coupling partner in hexafluoroisopropanol (HFIP), a solvent chosen for its unique ability to stabilize charged intermediates and enhance reaction rates. The catalyst system is then introduced, typically at a loading of 5 mol% for the ruthenium dimer and 20 mol% for the silver salt, along with three equivalents of pivalic acid to drive the activation cycle. The reaction proceeds under stirring at ambient temperature, with progress monitored by thin-layer chromatography (TLC) until the starting material is consumed, usually within 24 hours. Upon completion, the reaction mixture undergoes a straightforward aqueous workup involving extraction with dichloromethane, washing to remove metal residues, and drying over anhydrous sodium sulfate. The final product is isolated via concentration and purified by flash column chromatography. For a detailed, step-by-step breakdown of the standardized operating procedures and safety precautions, please refer to the guide below.
- Combine N-pivaloyl indole substrate and 3-phenyloxazolone in a reaction vessel with HFIP solvent.
- Add the catalyst system comprising [Ru(p-cymene)Cl2]2 and AgSbF6, along with pivalic acid as an additive.
- Stir the mixture at room temperature for 24 hours, then perform aqueous workup and column chromatography to isolate the pure 7-amide indole product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this ruthenium-catalyzed methodology offers substantial strategic benefits for organizations managing the supply of fine chemical intermediates. The shift from multi-step, low-yielding legacy processes to this direct one-pot transformation fundamentally alters the cost structure of manufacturing 7-amide indoles. By eliminating the need for separate borylation, oxidation, or reduction steps, the overall material throughput is significantly improved, leading to a drastic reduction in raw material consumption and waste disposal costs. The ability to run the reaction at room temperature without the need for cryogenic cooling or high-pressure reactors lowers energy consumption and reduces the capital intensity of the production facility. Moreover, the use of air-stable reagents and the tolerance of the catalyst to ambient conditions simplify logistics and storage requirements, mitigating risks associated with the handling of pyrophoric or moisture-sensitive materials. These factors collectively contribute to a more resilient supply chain capable of responding quickly to fluctuating market demands without compromising on product quality or delivery timelines.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the elimination of expensive stoichiometric reagents and the reduction of unit operations. Unlike previous methods requiring costly iridium catalysts or specialized directing groups that add molecular weight without value, this route uses relatively inexpensive ruthenium precursors and simple oxazolones. The high atom economy of the reaction, where the oxazolone ring opens to incorporate the amide group while releasing only gas, ensures that the majority of the reactant mass ends up in the final product. This efficiency translates directly into lower cost of goods sold (COGS), allowing for more competitive pricing in the global marketplace. Additionally, the simplified workup procedure reduces the volume of solvents required for purification, further decreasing operational expenditures related to solvent recovery and waste treatment.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the complexity of synthetic routes and the availability of niche reagents. This method utilizes commercially available indole derivatives and oxazolones, which are produced at scale by multiple suppliers, reducing the risk of single-source bottlenecks. The robustness of the reaction conditions means that production is less susceptible to interruptions caused by equipment failure or environmental fluctuations. The ability to synthesize a wide range of analogues using the same core protocol allows manufacturers to maintain a flexible inventory of intermediates that can be rapidly converted into different final products as customer needs evolve. This agility is crucial for supporting fast-paced drug development programs where timelines are critical and the ability to pivot quickly can provide a significant competitive advantage.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the plant often reveals hidden challenges, but this protocol is inherently designed for scalability. The exothermic nature of the reaction is manageable at room temperature, reducing the risk of thermal runaway incidents during large-batch production. The absence of toxic heavy metal oxidants or hazardous azide reagents improves the safety profile of the manufacturing site, facilitating easier compliance with increasingly stringent environmental, health, and safety (EHS) regulations. The use of HFIP, while a specialty solvent, can be recovered and recycled efficiently, aligning with sustainability goals. The streamlined process generates less chemical waste per kilogram of product, lowering the environmental impact and reducing the fees associated with hazardous waste disposal. This makes the technology not only economically sound but also environmentally responsible, appealing to partners committed to green chemistry initiatives.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology for their specific applications, we have compiled answers to common inquiries regarding the reaction scope, optimization, and practical implementation. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, ensuring accuracy and relevance for process development scientists. Understanding these nuances is essential for successfully transferring the methodology from literature to a working production line. We encourage stakeholders to review these points carefully to assess the alignment of this synthetic route with their project requirements.
Q: Why is the Ruthenium catalyst preferred over Iridium for this transformation?
A: According to the patent data, while Iridium catalysts have been used for indole functionalization, the specific Ruthenium system ([Ru(p-cymene)Cl2]2 with AgSbF6) demonstrated superior yields (up to 70%) compared to Iridium alternatives which showed significantly reduced yields or no product formation under identical conditions.
Q: What are the optimal reaction conditions for scaling this process?
A: The process operates efficiently at room temperature (20-30°C) in an air atmosphere using HFIP as the solvent. The use of ambient temperature and pressure eliminates the need for specialized heating or inert gas equipment, facilitating easier commercial scale-up.
Q: Can this method tolerate diverse functional groups on the indole ring?
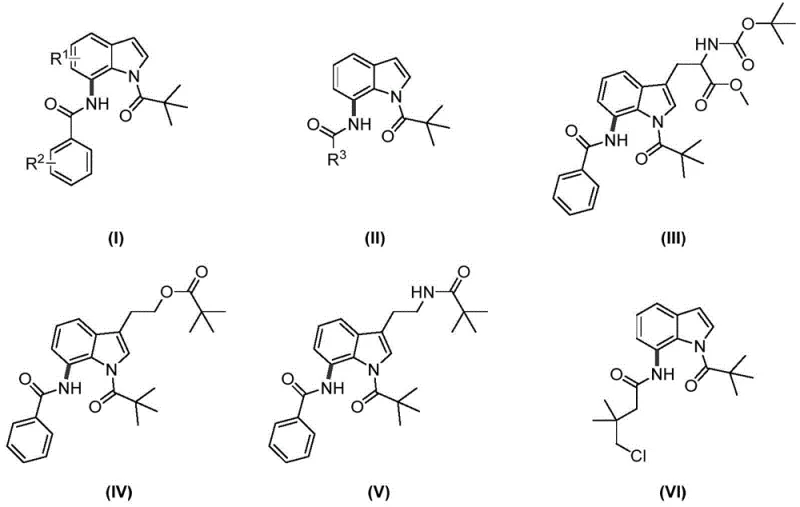
A: Yes, the methodology exhibits broad substrate scope. The patent examples demonstrate successful amidation on indoles substituted with methyl, methoxy, halogens, and even complex side chains like tryptophan derivatives, maintaining good regioselectivity at the C7 position.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 7-Amide Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug discovery and development. Our team of expert chemists has thoroughly analyzed the potential of the ruthenium-catalyzed C-H amidation technology and is fully equipped to leverage it for your custom synthesis needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory scale to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are designed to handle complex organometallic reactions with precision, supported by rigorous QC labs that enforce stringent purity specifications for every batch released. Whether you require gram-scale quantities for initial biological screening or metric tons for clinical trials, our infrastructure is ready to deliver consistent, high-quality 7-amide indole intermediates that meet your exacting standards.
We invite you to collaborate with us to optimize this synthetic route for your specific target molecules. Our technical procurement team is prepared to provide a Customized Cost-Saving Analysis that evaluates the economic benefits of switching to this novel methodology for your supply chain. Please contact us to request specific COA data for our reference standards and to discuss route feasibility assessments tailored to your project timeline. By partnering with NINGBO INNO PHARMCHEM, you gain access to cutting-edge chemical technology combined with the reliability of a trusted global supplier, empowering your organization to bring life-saving medicines to market faster and more efficiently.
