Advanced Catalytic Asymmetric Synthesis of 2'-Hydroxychalcone Diels-Alder Adducts for Pharmaceutical Applications
Advanced Catalytic Asymmetric Synthesis of 2'-Hydroxychalcone Diels-Alder Adducts for Pharmaceutical Applications
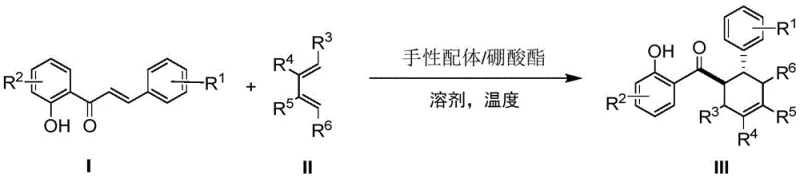
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for complex natural product scaffolds, particularly those exhibiting potent biological activities such as anticancer and anti-inflammatory properties. Patent CN107032972B introduces a significant technological breakthrough in the preparation of Diels-Alder products featuring a 2'-hydroxychalcone structure, which serve as critical precursors for flavonoid-based drug candidates. This innovation addresses the longstanding challenge of achieving high enantioselectivity in Diels-Alder cycloadditions without relying on expensive stoichiometric chiral auxiliaries. By utilizing a novel chiral ligand and triphenyl borate complex system, the disclosed method enables the efficient construction of six-membered rings with precise stereochemical control. For R&D directors and procurement specialists, this represents a pivotal shift towards more cost-effective and environmentally sustainable manufacturing processes for high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of flavonoid Diels-Alder type natural products has been fraught with significant chemical and economic inefficiencies. Traditional bionic synthesis routes often relied on thermal heating methods that produced racemic mixtures, necessitating difficult and yield-lossing resolution steps to obtain the desired enantiomer. Furthermore, earlier asymmetric approaches, such as those reported by Lexuan et al., required the use of chiral ligand-boric acid complexes in stoichiometric quantities. This requirement not only drastically inflated the raw material costs due to the high price of chiral ligands but also complicated the downstream purification process, as removing large amounts of chiral additives from the final product proved labor-intensive. Additionally, alternative methods involving single electron transfer or nano-silver particle catalysis often suffered from operational complexity and inconsistent reproducibility, making them unsuitable for large-scale commercial production where reliability is paramount.
The Novel Approach
The methodology outlined in patent CN107032972B fundamentally transforms this landscape by introducing a truly catalytic asymmetric system. Instead of consuming the chiral source, the new protocol employs a chiral ligand, such as (R)-VANOL or (R)-BINOL, in conjunction with triphenyl borate in merely catalytic amounts (typically 10 mol%). This drastic reduction in catalyst loading directly translates to substantial cost savings and simplifies the reaction mixture. The process operates under relatively mild reflux conditions in trifluorotoluene at 100°C, demonstrating remarkable tolerance for various substituents on both the chalcone and diene components. Crucially, the patent highlights that the chiral ligand can be quantitatively recovered after the reaction, creating a closed-loop system that minimizes waste and maximizes atom economy. This approach not only achieves high yields ranging from 85% to 98% but also delivers excellent enantiomeric excess values, reaching up to 90.8% ee in optimized examples, thereby eliminating the need for subsequent chiral resolution.
Mechanistic Insights into Chiral Ligand/Borate Catalyzed Diels-Alder Reaction
The core of this technological advancement lies in the formation of an active chiral Lewis acid complex in situ. When the chiral ligand, such as (R)-3,3'-diphenyl-[2,2'-binaphthalene]-1,1'-diol ((R)-VANOL), interacts with triphenyl borate, it generates a highly organized chiral environment capable of activating the dienophile. The boron center acts as a Lewis acid, coordinating with the carbonyl oxygen of the 2'-hydroxychalcone substrate. This coordination lowers the LUMO energy of the dienophile, facilitating the [4+2] cycloaddition with the diene. The bulky substituents on the binaphthyl backbone of the ligand create a steric shield that directs the approach of the diene, ensuring that the reaction proceeds through a specific transition state that favors one enantiomer over the other. This precise stereocontrol is essential for producing pharmaceutical intermediates where biological activity is often dependent on absolute configuration.
Furthermore, the reaction conditions have been meticulously optimized to balance reaction rate and selectivity. Research indicates that temperature plays a critical role, with 100°C identified as the optimal point for maximizing both yield and enantioselectivity. At this temperature, the kinetic energy is sufficient to overcome the activation barrier for the cycloaddition without compromising the integrity of the chiral catalyst or promoting non-selective background reactions. The mechanism also accounts for the broad substrate scope observed in the examples; electron-donating groups like methoxy and electron-withdrawing groups like bromine on the chalcone ring are well-tolerated. Similarly, various dienes, from simple isoprene to complex terpenes like myrcene, participate effectively. This mechanistic robustness ensures that impurity profiles remain clean, as the catalytic cycle is highly specific, reducing the formation of regioisomers or polymerization byproducts that often plague uncatalyzed thermal Diels-Alder reactions.
How to Synthesize 2'-Hydroxychalcone Cyclohexene Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires strict adherence to the activation and addition protocols described in the patent to ensure optimal catalyst performance. The process begins with the pre-formation of the active catalytic species by refluxing the chalcone, ligand, and borate source, followed by the controlled addition of the diene. This sequential addition is vital to prevent diene polymerization and ensure the catalyst is fully engaged with the dienophile before the cycloaddition commences. The following guide outlines the standardized operational procedure derived from the patent examples, providing a clear roadmap for technical teams to replicate these high-efficiency results.
- Prepare the catalytic system by dissolving substituted 2'-hydroxychalcone, a chiral ligand (such as R-VANOL), and triphenyl borate in trifluorotoluene, then reflux at 100°C for 1 hour to activate the complex.
- Cool the mixture to room temperature, add the diene substrate (e.g., isoprene or myrcene), and continue refluxing for 2 to 39 hours while monitoring progress via TLC.
- Quench the reaction with distilled water, extract with ethyl acetate, wash with brine, dry the organic phase, and purify via silica gel chromatography to recover the chiral ligand and isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from stoichiometric to catalytic asymmetric synthesis offers profound strategic benefits beyond mere technical elegance. The primary driver for adoption is the significant reduction in manufacturing costs associated with raw material consumption. By reducing the loading of expensive chiral ligands from stoichiometric equivalents to catalytic levels, the direct material cost per kilogram of the final intermediate is drastically lowered. Moreover, the ability to recover and reuse the chiral ligand quantitatively further amortizes this cost over multiple batches, creating a compounding financial advantage that improves gross margins for the final API. This economic efficiency makes the commercial production of complex flavonoid derivatives viable for broader therapeutic applications where cost sensitivity is high.
- Cost Reduction in Manufacturing: The elimination of stoichiometric chiral reagents removes a major cost bottleneck typically associated with asymmetric synthesis. In traditional methods, the chiral auxiliary often constitutes a significant portion of the total batch cost and generates substantial waste. By shifting to a catalytic regime with recoverable ligands, the process inherently reduces the cost of goods sold (COGS). Additionally, the use of triphenyl borate, a relatively inexpensive and stable reagent, avoids the need for exotic or hazardous metal catalysts that require specialized handling and expensive disposal protocols. This simplification of the reagent profile streamlines the supply chain, reducing dependency on scarce or volatile raw material markets.
- Enhanced Supply Chain Reliability: The robustness of this catalytic system contributes directly to supply chain stability. The reaction tolerates a wide range of substrates and operates under standard reflux conditions, minimizing the risk of batch failures due to sensitive reaction parameters. The starting materials, such as substituted chalcones and common dienes like isoprene or myrcene, are commercially available commodities with established supply lines, reducing the risk of raw material shortages. Furthermore, the simplified workup procedure involving standard aqueous quenching and extraction reduces the processing time per batch, allowing for faster turnaround times and increased throughput capacity to meet fluctuating market demands without requiring massive capital investment in new equipment.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this method aligns perfectly with modern green chemistry principles. The absence of heavy transition metals simplifies the purification process and ensures that the final product meets stringent residual metal specifications required for pharmaceutical ingredients. The quantitative recovery of the chiral ligand minimizes chemical waste generation, lowering the environmental footprint and disposal costs associated with hazardous waste treatment. The scalability of the process is evidenced by its straightforward operational steps—dissolution, reflux, and extraction—which are easily transferable from gram-scale laboratory experiments to multi-ton industrial reactors, ensuring a seamless path from process development to commercial manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric Diels-Alder technology. These insights are derived directly from the experimental data and technical specifications provided in patent CN107032972B, offering clarity on the practical application of this synthesis route for pharmaceutical intermediate production.
Q: What is the primary advantage of this catalytic method over traditional stoichiometric approaches?
A: Unlike previous methods requiring stoichiometric amounts of expensive chiral ligands, this patent describes a catalytic system using only 10 mol% of the chiral ligand/borate complex, significantly reducing raw material costs while maintaining high enantioselectivity (up to 90.8% ee).
Q: Is the chiral ligand recoverable in this process?
A: Yes, the process allows for the quantitative recovery of the chiral ligand after the reaction workup and purification steps, which further enhances the economic viability and sustainability of the manufacturing process.
Q: What range of substrates is compatible with this Diels-Alder protocol?
A: The method demonstrates broad substrate scope, accommodating various substituted 2'-hydroxychalcones (including bromo and methoxy derivatives) and diverse dienes such as isoprene, 2,3-dimethyl-1,3-butadiene, and myrcene, yielding products with 85-98% isolated yields.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2'-Hydroxychalcone Derivative Supplier
The technological potential of the catalytic asymmetric Diels-Alder reaction described in CN107032972B positions it as a cornerstone for the next generation of flavonoid-based therapeutics. NINGBO INNO PHARMCHEM stands ready to leverage this advanced chemistry to support your drug development pipeline. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from clinical trials to market launch is seamless. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of 2'-hydroxychalcone derivatives meets the highest standards of quality and enantiomeric purity required by global regulatory bodies.
We invite you to collaborate with our technical team to explore how this cost-effective synthesis route can optimize your specific project requirements. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating exactly how this catalytic method can reduce your overall manufacturing expenses. We encourage you to contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss custom route feasibility assessments for your proprietary targets, ensuring a secure and efficient supply chain for your critical pharmaceutical intermediates.
