Advanced One-Step Synthesis of Indolizine Diarylmethane Derivatives for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to access complex heterocyclic scaffolds that serve as critical backbones for next-generation therapeutics. Patent CN114163436A introduces a groundbreaking advancement in this domain by disclosing a novel class of diarylmethane derivatives containing an indolizine core. These compounds are not merely structural curiosities; they represent a significant leap forward in medicinal chemistry, specifically designed to overcome the limitations of existing molecules which often suffer from narrow biological profiles. The invention provides a versatile platform where the indolizine moiety is fused with either benzofuran or indole structures, thereby endowing the resulting molecules with potent dual antibacterial and anticancer activities. This structural innovation addresses a critical gap in the current landscape of bioactive agents, where single-target efficacy is increasingly insufficient for treating complex diseases. Furthermore, the patent elucidates a highly efficient preparation method that transforms accessible starting materials into these high-value intermediates through a streamlined catalytic process.
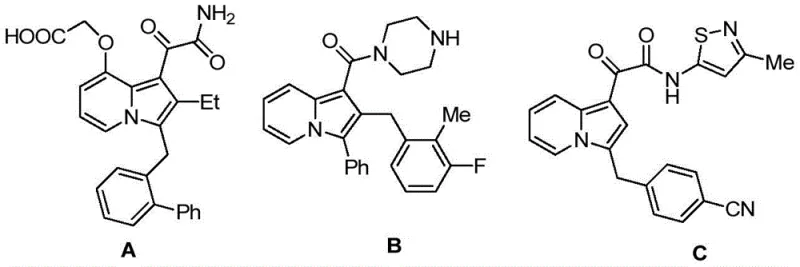
From a strategic sourcing perspective, the ability to produce such multifunctional intermediates reliably is paramount for maintaining a competitive edge in drug development pipelines. The disclosed technology enables the synthesis of compounds that can serve not only as final active pharmaceutical ingredients (APIs) but also as versatile intermediates for constructing other functional organic molecules. This dual utility enhances the value proposition for procurement teams looking to consolidate their supply chains. By adopting the methodologies outlined in this patent, manufacturers can secure a steady supply of high-purity precursors that are essential for the研发 of antiviral and anticancer drugs. The structural diversity allowed by the variable R groups ensures that a wide library of analogs can be generated to optimize pharmacokinetic properties without necessitating entirely new synthetic routes for each variant.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex diarylmethane structures incorporating indolizine rings has been fraught with significant challenges that hinder large-scale production and cost-effectiveness. Traditional routes often rely on multi-step sequences that require the isolation and purification of unstable intermediates, leading to cumulative yield losses and increased operational expenditures. These conventional methods frequently employ harsh reaction conditions, such as extreme temperatures or strongly acidic environments, which can degrade sensitive functional groups and generate difficult-to-remove impurities. Moreover, the starting materials for these older pathways are often expensive or difficult to source commercially, creating bottlenecks in the supply chain that can delay critical drug development timelines. The lack of atom economy in these processes also results in substantial chemical waste, posing environmental compliance issues that modern green chemistry initiatives strive to eliminate. Consequently, there has been a persistent industry demand for a more direct, efficient, and scalable approach to accessing these privileged scaffolds.
The Novel Approach
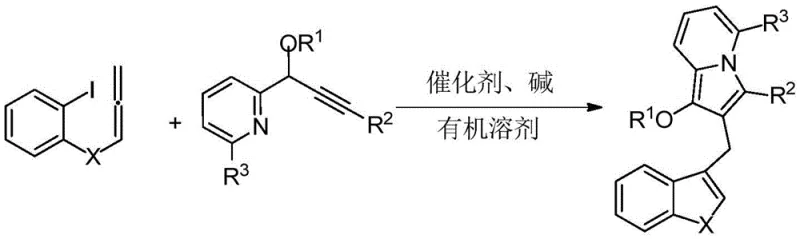
The methodology presented in patent CN114163436A offers a transformative solution by enabling the construction of the target indolizine-containing diarylmethane framework in a single operational step. This novel approach leverages a palladium-catalyzed cascade cyclization reaction that converges a 2-iodophenyl allene compound with a propargyl pyridine compound. As illustrated in the reaction scheme below, this tandem process efficiently builds multiple bonds and rings simultaneously, drastically reducing the number of unit operations required. The use of readily available and inexpensive raw materials, such as substituted 2-iodophenyl allenes and various propargyl pyridines, significantly lowers the entry barrier for production. The reaction proceeds under relatively mild conditions, typically requiring temperatures between 60-90°C, which preserves the integrity of sensitive substituents and minimizes energy consumption. This one-pot strategy not only simplifies the workflow but also enhances the overall throughput, making it an ideal candidate for commercial scale-up.
Mechanistic Insights into Palladium-Catalyzed Cascade Cyclization
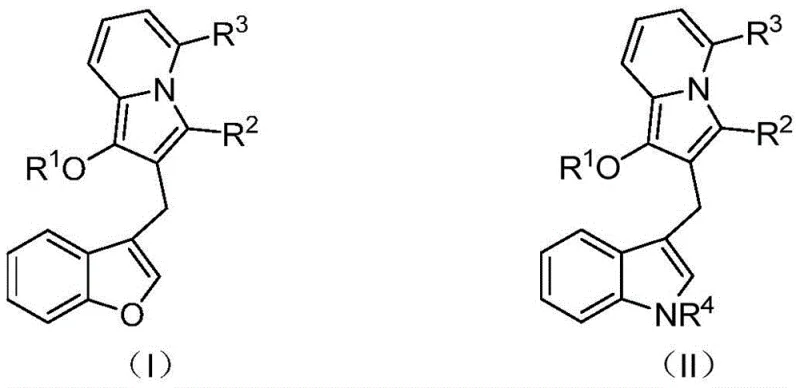
The core of this technological breakthrough lies in the sophisticated mechanistic pathway driven by the palladium catalyst. The reaction initiates with the oxidative addition of the palladium(0) species into the carbon-iodine bond of the 2-iodophenyl allene substrate, generating a reactive organopalladium intermediate. This species then undergoes a migratory insertion into the allene system, followed by a subsequent intramolecular carbopalladation involving the alkyne moiety of the propargyl pyridine partner. This sequence of events triggers a cascade of cyclization steps that ultimately forge the indolizine core while simultaneously establishing the diarylmethane linkage. The choice of ligand and catalyst loading is critical; the patent specifies the use of tetrakis(triphenylphosphine)palladium or similar Pd(0) sources, which provide the necessary electronic and steric environment to facilitate these transformations with high regioselectivity. The presence of a base, such as potassium carbonate or cesium carbonate, is essential to neutralize the acid byproducts and regenerate the active catalytic species, ensuring the cycle continues efficiently until completion.
Controlling the impurity profile is a major concern for R&D directors, and this mechanism offers inherent advantages in that regard. By conducting the reaction in polar aprotic solvents like N,N-dimethylformamide (DMF), the solubility of both organic substrates and inorganic bases is optimized, promoting homogeneous reaction kinetics that reduce the formation of side products. The mild thermal window of 60-90°C prevents thermal decomposition pathways that are common in high-temperature syntheses. Furthermore, the structural flexibility of the system allows for precise tuning of the electronic properties of the final product through the selection of R groups, as defined in the general formulas. For instance, varying the acyl group at the R1 position or the substituent on the phenyl ring at R2 allows chemists to modulate the lipophilicity and metabolic stability of the molecule without altering the core synthetic strategy. This modularity ensures that the process remains robust even when scaling up to produce diverse libraries of analogs for structure-activity relationship (SAR) studies.
How to Synthesize Indolizine Diarylmethane Derivatives Efficiently
To implement this synthesis in a laboratory or pilot plant setting, operators must adhere to strict procedural guidelines to maximize yield and purity. The process begins with the precise weighing of the 2-iodophenyl allene and the propargyl pyridine derivative, typically in a molar ratio ranging from 1:1 to 1:2 to ensure complete consumption of the limiting reagent. These substrates are combined with the palladium catalyst and the inorganic base in a dry reaction vessel purged with nitrogen to maintain an inert atmosphere, which is crucial for preventing catalyst deactivation by oxygen. The mixture is then heated to the optimal temperature, preferably around 80°C, and stirred for a duration of 4 to 12 hours, with 6 hours being the standard benchmark for high conversion. Following the reaction, a standardized workup procedure involving aqueous quenching, organic extraction, and chromatographic purification is employed to isolate the target compound.
- Mix 2-iodophenyl allene compound, propargyl pyridine compound, palladium catalyst (e.g., Pd(PPh3)4), alkali (e.g., K2CO3), and organic solvent (e.g., DMF) in a reaction vessel.
- Stir and react the mixture for 4-12 hours at a temperature range of 60-90°C under an inert nitrogen atmosphere to facilitate the cascade cyclization.
- Perform post-treatment including extraction with ethyl acetate, drying over anhydrous sodium sulfate, filtration, solvent removal, and purification via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers compelling economic and logistical benefits that extend beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the manufacturing process, which directly translates to reduced operational costs and improved margin potential. By consolidating what would traditionally be a multi-step synthesis into a single catalytic event, manufacturers can eliminate the need for intermediate isolation, drying, and storage, thereby reducing labor hours and facility occupancy time. The reliance on commodity chemicals such as potassium carbonate and DMF, rather than exotic or proprietary reagents, ensures that the raw material supply chain is resilient and less susceptible to market volatility. This stability is critical for long-term production planning and contract negotiations with downstream pharmaceutical clients who demand consistent pricing and availability.
- Cost Reduction in Manufacturing: The elimination of transition metal removal steps often required in other cross-coupling reactions is a significant cost driver here, as the workup is straightforward. The high atom economy of the cascade reaction means that a larger proportion of the starting mass ends up in the final product, minimizing waste disposal costs. Additionally, the ability to run the reaction at moderate temperatures reduces energy consumption compared to high-heat processes, contributing to lower utility bills. The use of standard column chromatography for purification, rather than complex crystallization or distillation setups, further lowers the capital expenditure required for equipment. Overall, the streamlined nature of this process allows for a leaner manufacturing footprint.
- Enhanced Supply Chain Reliability: The starting materials, specifically the 2-iodophenyl allene compounds and propargyl pyridines, are derived from widely available chemical feedstocks, reducing the risk of supply disruptions. The robustness of the reaction conditions means that the process is less sensitive to minor fluctuations in raw material quality, which can often cause batch failures in more sensitive chemistries. This tolerance enhances the predictability of production schedules, allowing supply chain managers to commit to tighter delivery windows with confidence. Furthermore, the scalability of the reaction from gram to kilogram scales has been demonstrated in the examples, indicating that technology transfer to larger reactors can be achieved with minimal re-optimization. This ease of scale-up ensures that supply can be ramped up quickly to meet surging demand.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste, aligning with increasingly stringent global environmental regulations regarding solvent use and heavy metal discharge. The solvents used, such as ethyl acetate for extraction, are relatively benign and can be recycled, supporting sustainability goals. The absence of stoichiometric toxic reagents reduces the burden on wastewater treatment facilities. From a scalability perspective, the exothermic profile of the reaction is manageable, allowing for safe operation in large-scale vessels without the need for specialized cooling infrastructure. This combination of safety, efficiency, and environmental friendliness makes the process highly attractive for contract development and manufacturing organizations (CDMOs) looking to expand their portfolio of green chemistry capabilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this indolizine synthesis technology. These answers are derived directly from the experimental data and specifications provided in the patent documentation to ensure accuracy and relevance for technical decision-makers. Understanding these nuances is essential for evaluating the fit of this technology within your specific production context.
Q: What are the biological activities of the new indolizine diarylmethane derivatives?
A: Unlike prior art compounds which often exhibit single biological activity, these novel derivatives simultaneously contain indolizine and benzofuran or indole moieties, demonstrating dual antibacterial and anticancer activities.
Q: What catalysts and conditions are required for this synthesis?
A: The process utilizes palladium salt catalysts such as tetrakis(triphenylphosphine)palladium, with alkali bases like potassium carbonate in solvents like DMF, typically reacting at 80°C for 6 hours.
Q: How does this method improve upon conventional synthetic routes?
A: This method achieves the construction of complex heterocyclic systems in a single step from cheap and easily obtained raw materials, avoiding multi-step sequences and harsh conditions associated with traditional methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indolizine Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the chemistry described in patent CN114163436A and are fully equipped to bring these advanced intermediates to the global market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. We understand that in the pharmaceutical sector, purity is non-negotiable; therefore, our stringent purity specifications and rigorous QC labs guarantee that every batch of indolizine derivative meets the highest international standards. Our state-of-the-art facilities are designed to handle palladium-catalyzed reactions safely and efficiently, mitigating the risks associated with heavy metal handling while maximizing yield.
We invite you to collaborate with us to leverage this cutting-edge synthesis for your drug development programs. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you accelerate your timeline to market with a reliable supply of high-quality indolizine-containing diarylmethane derivatives that drive innovation in anticancer and antibacterial therapies.
