Advanced Synthesis of 2-(4-Chlorophenoxy)-Propoxyamine for Scalable Pharmaceutical Production
Advanced Synthesis of 2-(4-Chlorophenoxy)-Propoxyamine for Scalable Pharmaceutical Production
The pharmaceutical industry continuously seeks robust synthetic pathways that balance high purity with operational safety, particularly for critical intermediates like 2-(4-chlorophenoxy)-propoxyamine. Patent CN112544621B introduces a transformative methodology that addresses longstanding inefficiencies in the production of this key building block, which is essential for various bioactive molecules. This innovative approach replaces hazardous traditional reagents with safer alternatives while simultaneously boosting overall process yield and stereochemical control. By leveraging a unique hydroxamic acid coupling strategy, the technology offers a compelling solution for manufacturers aiming to optimize their supply chains for complex ether-amine derivatives. The following analysis details the technical merits and commercial implications of this advanced synthesis route.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-(4-chlorophenoxy)-propoxyamine has relied heavily on protection-deprotection strategies involving N-hydroxyphthalimide or similar bulky groups. These conventional routes typically require the use of hydrazine hydrate or other aggressive nucleophiles for the final deprotection step, introducing significant safety hazards due to the explosive nature of hydrazine derivatives. Furthermore, the phthalimide protecting group is sterically demanding, often leading to sluggish reaction kinetics and requiring harsh conditions that can degrade sensitive chiral centers. The cumulative effect of these inefficiencies is a lower overall yield, often hovering around 65%, and a more complex purification burden to remove phthalhydrazide byproducts. Such drawbacks not only inflate manufacturing costs but also create substantial environmental and safety compliance challenges for production facilities.
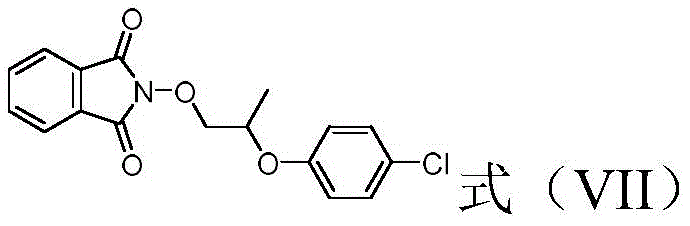
The Novel Approach
In stark contrast, the method disclosed in CN112544621B utilizes a streamlined three-step sequence that bypasses the need for hazardous hydrazine deprotection entirely. The process initiates with the activation of the alcohol precursor via mesylation, followed by a highly efficient nucleophilic substitution using acetohydroxamic acid. This key transformation installs the nitrogen functionality directly without the steric bulk associated with phthalimide groups, allowing for milder reaction temperatures and faster kinetics. The final step involves a simple acid hydrolysis to cleave the acetamide bond, releasing the free amine under controlled conditions that preserve stereochemical integrity. This strategic shift results in a marked improvement in process safety and a significant increase in isolated yield, reaching approximately 79% in optimized examples.
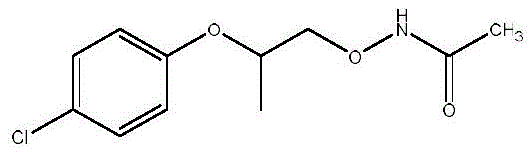
Mechanistic Insights into Hydroxamic Acid-Mediated Etherification
The core innovation of this pathway lies in the nucleophilic displacement of the mesylate leaving group by the deprotonated acetohydroxamic acid species. Under basic conditions, typically employing sodium hydroxide in polar aprotic solvents like DMSO or NMP, the hydroxamic acid forms a reactive anion that attacks the electrophilic carbon of the mesylate. This SN2-type mechanism proceeds with inversion of configuration, yet the overall process is designed to maintain high enantiomeric excess by minimizing racemization side reactions through precise temperature control between 30°C and 60°C. The choice of acetohydroxamic acid is particularly advantageous as the resulting acetamide intermediate is stable enough to be isolated yet labile enough to be hydrolyzed under mild acidic conditions. This balance ensures that the nitrogen atom is introduced efficiently without requiring extreme thermal energy that could compromise the chiral center adjacent to the ether linkage.
Following the coupling step, the hydrolysis mechanism is carefully managed to ensure high chemical purity of the final amine. The use of hydrochloric acid at concentrations of 30 wt% or higher facilitates the cleavage of the N-O bond in the acetohydroxamic derivative, regenerating the free hydroxylamine functionality which tautomerizes or rearranges to the stable amino-ether form. Crucially, the inventors found that hydrochloric acid outperforms other mineral acids like sulfuric acid in terms of both yield and chiral purity preservation. This specificity suggests that the counter-ion and acidity strength play a vital role in suppressing side reactions such as ether cleavage or elimination, thereby ensuring that the final product meets stringent pharmaceutical specifications with R-configuration purity exceeding 93%.
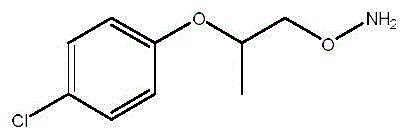
How to Synthesize 2-(4-Chlorophenoxy)-Propoxyamine Efficiently
Implementing this synthesis requires careful attention to stoichiometry and solvent selection to maximize the efficiency of each transformation. The process begins with the activation of the chiral alcohol, followed by the critical coupling with the hydroxamic acid, and concludes with the hydrolytic workup. Each stage has been optimized to minimize waste and maximize throughput, making it an ideal candidate for technology transfer. For a detailed breakdown of the specific operational parameters, reagent ratios, and workup procedures validated in the patent examples, please refer to the standardized synthesis guide below.
- Activate (R)-2-(4-chlorophenoxy)-propanol by reacting with methanesulfonyl chloride and triethylamine in toluene at 35°C to form the mesylate intermediate.
- Perform nucleophilic substitution by reacting the mesylate with acetohydroxamic acid and sodium hydroxide in DMSO at 60°C to generate N-[2-(4-chloro-phenoxy)-propoxy]acetamide.
- Conduct acid hydrolysis using 30 wt% hydrochloric acid in ethanol at 30°C, followed by pH adjustment and extraction to isolate the final amine product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this novel synthetic route offers profound advantages by fundamentally altering the cost and risk profile of producing 2-(4-chlorophenoxy)-propoxyamine. The elimination of expensive and dangerous hydrazine reagents removes a major bottleneck in raw material sourcing and storage, while the improved yield directly translates to lower cost of goods sold. Additionally, the use of commodity chemicals like methanesulfonyl chloride and acetohydroxamic acid ensures a stable and resilient supply chain that is less susceptible to market volatility compared to specialized protecting group reagents. These factors combine to create a manufacturing process that is not only economically superior but also operationally robust for long-term commercial partnerships.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substantial increase in reaction yield from approximately 65% to nearly 80%, which drastically reduces the amount of starting material required per kilogram of final product. Furthermore, the replacement of costly phthalimide derivatives with inexpensive acetohydroxamic acid significantly lowers the raw material bill, while the simplified purification steps reduce solvent consumption and energy usage during distillation. By avoiding the need for specialized hazard mitigation systems required for hydrazine handling, facilities can also realize significant savings in capital expenditure and operational safety compliance costs.
- Enhanced Supply Chain Reliability: The reliance on widely available bulk chemicals such as toluene, ethanol, and hydrochloric acid ensures that production is not dependent on single-source suppliers for exotic reagents. This diversification of the supply base mitigates the risk of production stoppages due to raw material shortages, providing a secure and continuous flow of intermediates for downstream API synthesis. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in reagent quality, further stabilizing the supply chain against upstream fluctuations.
- Scalability and Environmental Compliance: The mild reaction temperatures and absence of heavy metal catalysts make this process inherently scalable from pilot plant to multi-ton commercial production without significant engineering hurdles. The waste stream is significantly cleaner due to the avoidance of phthalhydrazide byproducts, simplifying wastewater treatment and reducing the environmental footprint of the manufacturing site. This alignment with green chemistry principles facilitates easier regulatory approval and supports corporate sustainability goals, making it a preferred choice for environmentally conscious pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and comparative examples provided in the patent documentation to ensure accuracy and relevance for potential licensees or buyers. Understanding these nuances is critical for evaluating the feasibility of integrating this route into existing manufacturing portfolios.
Q: How does the new hydroxamic acid route improve safety compared to traditional phthalimide methods?
A: The novel process eliminates the use of hazardous hydrazine compounds required for deprotection in traditional phthalimide routes, significantly reducing explosion risks and improving operational safety profiles for industrial scale-up.
Q: What yields and purity levels can be expected from this synthesis method?
A: Experimental data indicates consistent yields around 79% with chemical purity exceeding 94% and chiral purity (R-configuration) greater than 93%, outperforming conventional methods which typically yield around 65%.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the method utilizes mild reaction conditions (30-60°C), common solvents like toluene and ethanol, and avoids expensive transition metal catalysts, making it highly adaptable for cost-effective commercial production from 100 kgs to multi-ton scales.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(4-Chlorophenoxy)-Propoxyamine Supplier
NINGBO INNO PHARMCHEM stands at the forefront of adopting such advanced synthetic methodologies to deliver superior value to our global clientele. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practical, large-scale operations. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the critical chiral purity metrics required for high-performance pharmaceutical applications.
We invite forward-thinking organizations to collaborate with us to leverage this cost-effective and safe production technology. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized supply chain can support your long-term strategic goals.
